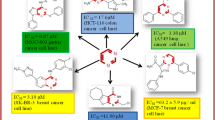
A series of arylsubstituted tetrazolocyclanodihydropyrimidines was prepared and their cytotoxic actions on SPEV-2 cells were studied. The activity of these substances was found to depend on the nature and position of the substitution groups in the benzene ring and the size of the alicycle. The most active compounds were o-chloro(methyl)phenyltetrazolohexahydroquinazolines.



Similar content being viewed by others
References
I. S. Khazhieva, T. V. Glukhareva, O. S. El’tsov, et al., Khim.-Farm. Zh., 49(5), 12 – 15 (2015); Pharm. Chem. J., 49(5), 296 – 300 (2015).
R. A. Al-Salahi, A. M. Gamal-Eldeen, A. M. Alanazi, et al., Molecules, 18, 1434 – 1446 (2013).
N. O. Vasil’kova and A. P. Kriven’ko, Proceedings of the International Scientific Publication SWorld [in Russian], Odessa (2015), pp. 4 – 7.
N. O. Vasil’kova, V. N. Filimonova, and A. P. Kriven’ko, Proceedings of the X All-Russian Conference of Young Scientists with International Participation [in Russian], Saratov (2015), pp. 11 – 12.
A. A. Matveeva, N. V. Poplevina, N. O. Borisova, and A. P. Kriven’ko, Khim. Geterotsikl. Soedin., 12, 2000 – 2002 (2012).
Guidelines for Clinical Trials of Medicines [in Russian], A. N. Mironov (ed.), Grif i K., Moscow (2012), Part 1, pp. 217 – 225.
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 51, No. 9, pp. 8 – 11, September, 2017.
Rights and permissions
About this article
Cite this article
Vasil’kova, N.O., Ivonin, M.A., Burygin, G.L. et al. Synthesis and Cytotoxic Activity of Arylsubstituted Tetrazolocyclanopyrimidines. Pharm Chem J 51, 756–759 (2017). https://doi.org/10.1007/s11094-017-1687-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-017-1687-z