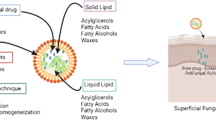
Voriconazole is a triazole antifungal drug used to treat invasive fungal infections including candidiasis, aspergillosis, and certain emerging fungal infections. Voriconazole demonstrates low water solubility. Therefore, drug solubilization is necessary for effective topical delivery. Nano-strategy may overcome its poor aqueous solubility as well as controlled drug release at action site, reduction of total dose, avoidance of systemic absorption and dosage form retention on skin. In topical antifungal administration, drug should pass outermost layer of skin; stratum corneum to reach lower layers, particularly into follicular epithelium. Nano-formulations including colloidal systems, vesicular carriers, and nanoparticles may play a major role for enhancing antifungals permeation profile. Present review surveys key experiments with voriconazole nano-based formulations used in superficial fungal infections treatment.

Similar content being viewed by others
References
R. Herbrecht, Expert. Rev. Anti Infect. Ther., 2, 485 – 497 (2004).
P. Hegener, P. Troke, G. Fätkenheuer, et al., AIDS, 12, 2227 – 2228 (1998).
P. Muñoz, M. Marín, P. Tornero, et al., Clin. Infect. Dis., 31, 1499 – 1501 (2000).
M. Ghannoum and D. Kuhn, Eur. J. Med. Res., 7, 242 – 256 (2002).
L. B. Johnson and C. A. Kauffman, Clin. Infect. Dis., 36, 630 – 637 (2003).
K. C. Klein and R. A. Blackwood, Pediatrics,118, e506 – e508 (2006).
C. H. Prats, F. L. Tello, A. B. San Jose, et al., Ann. Pharmacother., 38, 414 – 417 (2004).
Y. B. Vatinee, M. H. Kristin, J. R. Christopher, et al., Am. J. Ophthalmol., 143, 151 – 153 (2007).
Voriconazole AUPMV, available at http: // www.usp.org / sites / default / files / usp pdf / EN / USPNF / pendingStandards / m2761.pdf
D. H. Harding, Pharmaceutical formulations containing voriconazole, US Patent no. 6632803, B1 (2003).
M. Glujoy, C. Salerno, C. Bregni, et al., Int. J. Pharm. Pharm. Sci., 6, 8 – 16 (2014).
S. Güngör, M. S. Erdal, and B. Aksu, J. Cosmet. Dermatol. Sci. Appl., 3, 56 – 65 (2013).
Jr. Kumar, S. Muralidharan, and S. Parasuraman, J. Pharm. Sci. Res., 6, 229 – 235 (2014).
W. Yang, N. P. Wiederhold, and R. O. Williams III, Expert Opin Drug Deliv., 5, 1199 – 1216 (2008).
H. C. Korting and M. Schäfer-Korting, Handb. Exp. Pharmacol., 197, 435 – 468 (2010).
G. N. El-Hadidy, H. K. Ibrahim, M. I. Mohamed, et al., Drug Dev. Ind. Pharm., 38, 64 – 72 (2012).
Y.-H. Kim, C. K. Song, E. Jung, et al., J. Pharm. Investig., 44, 517 – 524 (2014).
R. H. Muller, M. Radtke, and S. A. Wissing, Adv. Drug. Deliv. Rev., 54, 131 – 155 (2002).
E. B. Souto, S. A. Wissing, C. M. Barbosa, et al., Int. J. Pharm., 278, 71 – 77 (2004).
P. G. Kakadia and B. R. Conway, Am. J. Pharmacol. Sci., 2, 1 – 7 (2014).
P. Schlupp, T. Blaschke, and K. D. Kramer, Skin. Pharmacol. Physiol., 24, 199 – 209 (2011).
S. H. Song, K. M. Lee, J. B. Kang, et al., Chem. Pharm. Bull., 62, 793 – 798(2014).
M. Gupta, U. Agrawal, and S. P. Vyas, Expert Opin Drug Deliv., 9, 783 – 804 (2012).
C. K. Song, P. Balakrishnan, C.-K. Shim, et al., Colloids. Surf. B: Biointerf., 92, 299 – 304 (2012).
Acknowledgements
The authors appreciate Center for Research and Training in Skin Diseases and Leprosy, Tehran University of Medical Sciences and Janus Knowledge Based Company, Tehran, Iran for support of this work.
Disclosure Statement
No competing financial interests exist.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nassiri-Kashani, M., Namdar, R., Nafisi, S. et al. Improved Voriconazole Topical Delivery by Nanoparticles (Minireview). Pharm Chem J 50, 76–79 (2016). https://doi.org/10.1007/s11094-016-1401-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-016-1401-6