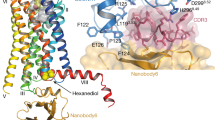
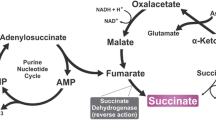
A considerable number of reports have been published in recent years on G protein-coupled receptors, their distribution in the body, mechanism of activation, and potential pathways for pharmacological actions. Intermediates in carbohydrate, fat, and protein metabolism and the tricarboxylic acid cycle, operating as endogenous ligands for a large group of ex-orphan receptors, have active roles in regulating metabolic processes, while their synthetic analogs, operating as both agonists and antagonists, may have potential for the development of new pharmaceuticals for a wide range of diseases (diabetes mellitus, obesity, metabolic syndrome, autoimmune disorders, hypertension, myocardial hypertrophy and ischemia, neurodegenerative processes, liver diseases, etc.). The present review addresses GPR91 (SUCNR1) receptors, which have been identified in fatty tissue, liver, kidneys, heart, brain, retinal neurons, dendritic cells, and platelets, and which are regarded as physiological regulators and cell sensors for stress-induced damage and hypoxia.
Similar content being viewed by others
References
T. Bay, L. F. Eghorn, A. B. Klein, and P. Wellendorph, Biochem. Pharmacol., 87(2), 200 – 228 (2014).
P. M. Deen and J. H. Robben, J. Am. Soc. Nephrol., 22(8), 1416 – 1422 (2011).
J. H. Robben, R. A. Fenton, S. L. Vargas, et al., Kidney International., 76(12), 1258 – 1267 (2009).
A. C. Ariza, P. M. Deen, and J. H. Robben, Front. Endocrinol., 322, 1 – 8 (2012).
W. He, F. J. Miao, D. C. Lin, et al., Nature, 429(6988), 188 – 193 (2004).
S. V. Okovityi, V. V. Gaivoronskaya, A. N. Kulikov, and S. N. Shulenin, Clinical Pharmacology. Selected Lectures [in Russian], GÉOTAR- Media, Moscow (2009).
S. V. Okovityi, D. S. Sukhanov, V. A. Zaplutanov, and A. N. Smagina, Klin. Med., 90(9), 69 – 74 (2012).
L. D. Lukianova, Patol. Fiziol. Eksp. Ter., No. 1, 3 – 19 (2011).
M. N. Kondrashova, Vopr. Biol. Med. i Farm. Khimii, No. 1, 7 – 12 (2002).
C. C. Blad, C. Tang, and S. Offermanns, Nat. Rev. Drug Discov., 11(8), 603 – 619 (2012).
T. T. Chen, E. I. Maevsky, and M. L. Uchitel, Front. Endocrinol. (Lausanne), 6(7), 1 – 11 (2015).
J. Peti-Peterdi, H. Gevorgyan, L. Lam, and A. Riquier-Brison, Pflügers Arch. Eur. J. Physiol., 465(1), 53 – 58 (2013).
I. Toma, J. J. Kang, A. Sipos, et al., J. Clin. Investig., 18(7), 2526 – 2534 (2008).
J. Peti-Peterdi, Kidney Int., 78(12), 1214 – 1217 (2010).
S. Tonack, C. Tang, and S. Offermanns, Am. J. Physiol., 304(4), 501 – 513 (2012).
P. R. Correa, E. A. Kruglov, M. Thompson, et al., J. Hepatol., 47(2), 262 – 269 (2007).
C. J. Aguiar, J. A. Rocha-Franco, P. A. Sousa, et al., Cell Commun. Signal., 12(78), 1 – 17 (2014).
L. Yang, D. Yu, H. H. Fan, et al., Int. J. Clin. Experim. Pathol., 7(9), 5414 – 5428 (2014).
K. R. Feingold, A. Moser, J. K. Shigenaga, and C. Grunfeld, J. Lipid Res., 55(12), 2501 – 2508 (2014).
C. Högberg, O. Gidlof, C. Tan, et al., J. Thromb. Haemostasis, 9(2), 361 – 372 (2009).
R. Bomprezzi, Ther. Adv. Neurol. Disorders, 8(1), 20 – 30 (2015).
T. Rubic, G. Lametschwandtner, S. Jost, et al., Nature Immunol., 9(11), 1261 – 1269 (2008).
P. A. Burns and D. J. Wilson, Angiogenesis, 6(1), 73 – 77 (2003).
A. Naldini and F. Carraro, Curr. Drug Targets Inflamm. Allergy, 4(1), 3 – 8 (2005).
S. Favret, F. Binet, E. Lapalme, et al., Aging, 5(6), 427 – 444 (2013).
J. C. Rivera, P. Sapieha, J. S. Joyal, et al., Neonatology, 100(4), 343 – 353 (2011).
D. Hamel, M. Sanchez, F. Duhamel, et al., Arterioscler. Thromb. Vasc. Biol., 34(2), 285 – 293 (2014).
T. H. Adair, W. J. Gay, and J. P. Montani, Am. J. Physiol., 259(3), 393 – 404 (1990).
C. Roehrs, E. R. Garrido-Sanabria, A. C. Da Silva, et al., Neuroscience, 125(4), 964 – 971 (2004).
H. Taniguchi, C. Anacker, O. Wang, and K. Andresson, Exp. Neurol., 255, 30 – 37 (2014).
J. Peti-Peterdi, J. Clin. Investig., 123(7), 2788 – 2790 (2013).
E. B. Shustov and S. V. Okovityi, Biomeditsina, No. 2, 15 – 29 (2015).
T. Molnár, A. Dobolyi, G. Nyitrai, et al., BMC Neuroscience, 12(96), 1 – 17 (2011).
Z. M. Nawab, J. T. Daugirdas, T. S. Ing, et al., Trans Am. Soc. Artif. Intern. Organs, 30, 184 – 188 (1984).
T. Molnár, K. Antal, G. Nyitrai, and Z. Emri, Neuroscience, 162(2), 268 – 281 (2009).
T. Molnár, P. Barabas, L. Heja, et al., Neurosci. Res., 86(7), 1566 – 1576 (2008).
T. Molnár, J. Visy, A. Simon, et al., Bioorgan. Med. Chem. Lett., 18, No. 23, 6290 – 6292 (2008).
T. Molnár, E. K. Fekete, J. Kardos, et al., Neurosci. Res., 84(1), 27 – 36 (2006).
M. Gahr, B. J. Connemann, C. J. Schonfeldt-Lecuona, and R. W. Freudenmann, Fortschritte Neurologie-Psychiatrie, 81(3), 154 – 161 (2013).
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 49, No. 9, pp. 3 – 7, September, 2015.
Rights and permissions
About this article
Cite this article
Okovityi, S.V., Rad’ko, S.V. & Shustov, E.B. Succinate Receptors (SUCNR1) as a Potential Target for Pharmacotherapy. Pharm Chem J 49, 573–577 (2015). https://doi.org/10.1007/s11094-015-1331-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-015-1331-8