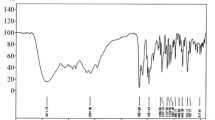
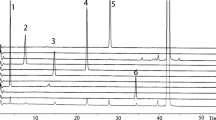
An analytical method for tablets of the new imidazobenzimidazole kappa-opioid agonist RU-1205 was developed. The principal pharmacopoeial parameters of tablet quality were studied. The contents of impurities and RU-1205 in the tablets were assayed quantitatively using HPLC. A method for determining the dose uniformity using UV spectrophotometry was developed. Procedures were proposed for determining the authenticity and dissolution profile of RU-1205 tablets.








Similar content being viewed by others
References
A. A. Spasov, V. A. Anisimova, P. M. Vasil’ev, et al., “Kappa-opioid agonist,” RU Pat. 2,413,512, Mar. 10, 2011; Byull. Izobret., No. 7 (2011).
A. A. Illarionov, L. N. Grushevskaya, and L. M. Gaevaya, Khim.-farm. Zh., 48(5), 50 – 56 (2014).
USSR State Pharmacopoeia. XIth Ed., Meditsina, Moscow, 1990, p. 154.
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 48, No. 6, pp. 48 – 53, June, 2014.
Rights and permissions
About this article
Cite this article
Illarionov, A.A., Grushevskaya, L.N., Gaevaya, L.M. et al. Analytical Method Development for Tablets of the New Kappa-Opioid Agonist RU-1205. Pharm Chem J 48, 408–413 (2014). https://doi.org/10.1007/s11094-014-1121-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-014-1121-8