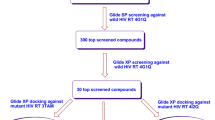
The search for new substances for the treatment and prophylaxis of HIV-AIDS remains relevant despite advances in highly active antiretroviral therapy (HAART), both because of the appearance of resistance to existing agents and because of their side effects and toxicity. Preventing the spread of HIV infection via sexual contacts by treating the uninfected partner has some advantage, as prophylactic effects can be obtained using simpler treatment (for example, using one agent rather than combinations of three drugs), as the need is to block the transfer of small numbers of infective virions. On the other hand, agents acting on the uninfected partner must be relatively nontoxic as they have to be used for long periods of time in healthy people. Effective chemoprophylaxis can be obtained using agents blocking the life cycle of the virus before integration into the human genome. Two widely used classes of agent therefore have potential – non-nucleoside reverse transcriptase (RT) inhibitors and integrase (IN) strand transfer inhibitors. In addition, proteins such as CD4, gp120, CCR5, CXCR4, and gp41 can be regarded as potential targets, as their functional roles at different stages in the interaction between virion and host cell have been established. As it is now known that all antiretroviral agents approved for medical use induce resistant viral strains, new anti-HIV agents must have chemical structures which are significantly different from known drugs and (if possible) must act on multiple pharmacological targets. Candidates can be sought by virtual screening of libraries of commercially available samples of chemical compounds; such libraries currently contain several million structural formulas for compounds which have been synthesized and are available for biological testing. Virtual screening can be performed using the computer programs PASS and PharmaExpert, allowing identification of substances with potential for experimental studies on the basis of a number of prognostic criteria (presence of the required types of activity, absence of side and toxic effects, appropriate physicochemical properties).We present here data on the current state of the search for new agents for the treatment and prophylaxis of HIV/AIDS, our approach to virtual screening for new potential substances for the prophylaxis of HIV/AIDS, and the results of using this approach in databases of chemical compounds available for screening. The potential for detection of anti-HIV compounds acting on multiple pharmacological targets among substances which have already been synthesized is discussed. We conclude that there is potential for the rational design of such drugs using computerized drug construction methods.



Similar content being viewed by others
Notes
Lopinavir is an HIV-1 protease inhibitor approved for medical use in the combined formulation Kaletra (lopinavir/ritonavir). Ritonavir inhibits P450 CYP3A4, which metabolizes lopinavir, leading to increases in plasma lopinavir and increases in its activity.
A HIV-1 protease approved for medical use in the combined formulation Stribild.
Cobicistat is a cytochrome P450 CYP3A4 inhibitor, which increases the actions of antiretroviral agents metabolized by this enzyme, allowing reductions in doses and, hence, side effects.
The names of types of activity are in English, as in PASS.
References
UNAIDS Report on the Global AIDS Epidemic, WHO Library (2012).
The Global HIV / AIDS Epidemics, Kaiser Family Foundation Fact Sheet (2012).
URL: http://www.unaids.org/ru.
C. D. Mathers and D. Loncar, PLoS Med., 3(11), e442 (2006).
Y. Mehellou and E. De Clercq, J. Med. Chem., 53(2), 521 – 538 (2010).
E. J. Arts and D. J. Hazuda, Cold Spring Harb. Perspect. Med., 2, a007161 (2012).
T. L. Hartman and R. W. Buckheit Jr., Molecular Biology International, 401965 (2012).
A. Carr, Nat. Rev. Drug Discovery, 2, 624 – 634 (2003).
J. Martinez-Picado, M. P. De Pasquale, N. Kartsonis, et al., Proc. Natl. Acad. Sci. USA, 97, 10948 – 10953 (2000).
S. J., Little, S. Holte, J. P. Routy, et al., N. Engl. J. Med., 347, 385 – 394 (2002).
Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents, US Department of Health and Human Services, 2011. AIDSinfo. URL: http://aidsinfo.nih.gov/contentfiles/AdultandAdolescentGL.pdf
R. Morphy, C. Kay, and Z. Rankovic, Drug Discovery Today, 9, 15, 641 – 651 (2004).
R. Morphy and Z. Rankovic, J. Med. Chem., 48(21), 6523 – 6543 (2005).
C. G. Wermuth, Drug Discovery Today, 9(19), 826 – 827 (2004).
I. Petersen, R. T. Jorgensen, C. Nielsen, et al., J. Med. Chem., 48(4), 1211 – 1220 (2005).
A. G. Skillman, K. W. Mauer, D. C. Roe, et al., Bioorg. Chem., 30(6), 443 – 458 (2002).
R. Di Santo, R. Costi, M. Artico, et al., J. Med. Chem., 49(6), 1939 – 1945 (2006).
Z.Wang and R. Vince, Bioorg. Med. Chem., 16(7), 3587 – 3595 (2008).
K. Princen, S. Halse, K. Vermeire, et al., J. Virol., 78(23), 12996 – 13006 (2004).
P. Zhan and X. Liu, Curr. Pharm. Des., 15(16), 1893 – 1917 (2009).
J. S. Olsen, D. Easterhoff, and S. Dewhurst, Future Med. Chem., 3(16), 2101 – 2116 (2011).
K. W. Buckheit and R. W. Buckheit Jr., Mol. Biol. Internat., 781305 (2012).
G. C. Morris and C. J. Lacey, Curr. Opin. Infect. Dis., 23(1), 57 – 63 (2010).
J. Nuttall, Drugs, 70(10), 1231 – 1243 (2010).
Q. A. Karim, S. S. Abdool Karim, J. A. Frolich, et al., Science, 329(5996), 1169 – 1174 (2010).
K. W. Buckheit and R. W. Buckheit Jr., Curr. HIV Res., 10(1), 97 – 104 (2012).
I. M. Kapetanovic, Chem. Biol. Interactions, 171(2), 165 – 176 (2008).
The Practice of Medicinal Chemistry, C. G. Wermuth (ed.), Academic Press, Salt Lake City (USA) (2008).
T. Scior, A. Bender, G. Tresadern, et al., J. Chem. Inf. Model., 52(4), 867 – 881 (2012).
Chemoinformatics Approaches to Virtual Screening, A. Varnek and A. Tropsha (eds.), RSC Publishing, Cambridge (UK) (2008).
V. V. Poroikov, D. A. Filimonov, and A. P. Budunova, Nauchno-Tekhn. Inform., Series 2(6), 11 – 13 (1993).
D. A. Filimonov, V. V. Poroikov, E. I. Karaicheva, et al., Éksperim. Klin. Farmakol., 58(2), 56 – 62 (1995).
T. A. Gloriozova, D. A. Filimonov, A. A. Lagunin, and V. V. Poroikov, Khim.-Farm. Zh., 32(12), 32 – 39 (1998).
D. A. Filimonov and V. V. Poroikov, Ros. Khim. Zh., 50(2), 66 – 75 (2006).
D. A. Filimonov and V. V. Poroikov, in: Chemoinformatics Approaches to Virtual Screening, Alexandre Varnek and Alexander Tropsha (eds.), RSC Cambridge (UK) Publishing, Cambridge (UK) (2008), pp. 182 – 216.
D. Filimonov, V. Poroikov, Yu. Borodina, and T. Gloriozova, J. Chem. Inf. Comput. Sci., 39(4), 666 – 670 (1999).
A. Dalby, J. G. Nourse, W. D. Hounshell, et al., J. Chem. Inf. Comput. Sci., 32(3), 244 – 255 (1992).
V. V. Poroikov, D. A. Filimonov, Yu. V. Borodina, et al., J. Chem. Inform. Comput. Sci., 40(6), 1349 – 1355 (2000).
J.-L. Reymond and M. Awale, ACS Chem. Neurosci., 3, 649 – 657 (2012).
V. V. Poroikov, D. A. Filimonov, W.-D. Ihlenfeldt, et al., J. Chem. Inform. Comput. Sci., 43(1), 228 – 236 (2003).
A. Geronikaki, P. Vicini, G. Theophilidis, et al., SAR and QSAR Environ. Res., 14(5 – 6), 487 – 497 (2003).
A. Geronikaki, E. Babaev, J. Dearden, et al., Bioorg. Med. Chem., 12(24), 6559 – 6568 (2004).
A. A. Lagunin, O. A. Gomazkov, D. A. Filimonov, et al., J. Med. Chem., 46(15), 3326 – 3332 (2003).
A. A. Geronikaki, A. A. Lagunin, D. I. Hadjipavlou-Litina, et al., J. Med. Chem., 51(6), 1601 – 1609 (2008).
A. Lagunin, A. Stepanchikova, D. Filimonov, and V. Poroikov, Bioinformatics, 16(8), 747 – 748 (2000).
A. Geronikaki, D. Druzhilovsky, A. Zakharov, and V. Poroikov, SAR and QSAR in Environ. Res., 19(1 & 2), 27 – 38 (2008).
V. Poroikov, A. Lagunin, and D. Filimonov, in: QSAR and Molecular Modelling in Rational Design of Bioactive Molecules, Esin Aki Sener and Ismail Yalcin (eds.), CADD & D Society, Ankara, Turkey (2005), pp. 514 – 515.
D. A. Filimonov, A. V. Zakharov, A. A. Lagunin, and V. V. Poroikov, SAR and QSAR Environ. Res., 20(7 – 8), 679 – 709 (2009).
G. V. Kokurkina, M. D. Dutov, S. A. Shevelev, et al., Eur. J. Med. Chem., 46(9), 4374 – 4382 (2011).
A. Lagunin, A. Zakharov, D. Filimonov, and V. Poroikov, Mol. Inform., 30(2 – 3), 241 – 250 (2011).
A. V. Zakharov, A. A. Lagunin, D. A. Filimonov, and V. V. Poroikov, Chem. Res. Toxicol., 25(11), 2378 – 2385 (2012).
V. H. Masand, D. T. Mahajan, K. N. Patil, et al., Chem. Biol. Drug Des. (2012), accepted for publication, URL: http://dx.doi.org/10.1111/cbdd.12099
O. A. Filz and V. V. Poroikov, Usp. Khimii, 81(2), 158 – 174 (2012).
O. A. Tarasova, Author’s Abstract of Master’s Thesis in Biological Sciences, Moscow (2012).
O. A. Filz, A. A. Lagunin, D. A. Filimonov, and V. V. Poroikov, SAR QSAR Environ. Res., 23(3 – 4), 279 – 296 (2012).
P. Eleftheriou, A. Geronikaki, D. Hadjipavlou-Litina, et al., Eur. J. Med. Chem., 47(1), 111 – 124 (2012).
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 47, No. 7, pp. 3 – 21, July, 2013.
Rights and permissions
About this article
Cite this article
Lagunin, A.A., Filimonov, D.A., Gloriozova, T.A. et al. Virtual Screening for Potential Substances for the Prophylaxis of HIV Infection in Libraries of Commercially Available Organic Compounds. Pharm Chem J 47, 343–360 (2013). https://doi.org/10.1007/s11094-013-0958-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-013-0958-6