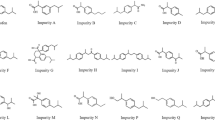
The objective of the study was to develop and validate a new GC-FID method for determination of impurities in kemantane (5-hydroxyadamantan-2-one) drug substance. The chromatographic mobilities of potential impurities in kemantane such as adamantan-2-one, adamantan-1-ol, adamantan-2-ol, adamantane-1,4-diol, adamantane-1,4-diol (syn-isomer), adamantane-2,6-diol, and adamantane-2,6-dione were studied. The chromatographic separation was performed on a quartz capillary column (VF-624ms, 60 m × 0.32 mm, 1.80 μm film thickness). The chromatographic system suitability parameters and method robustness factor were determined. The method had acceptable specificity, accuracy, linearity, and precision and high sensitivity. The developed method detected an impurity of adamantane-2,6-dione from 1.01 to 2.97% in four batches of kemantane drug substance.




Similar content being viewed by others
References
N. V. Klimova, L. N. Lavrova, Yu. I. Vikhlyaev, et al., USSR Pat. 535,085, Nov. 15, 1976; Byull. Izobret., No. 42 (1976).
N. G. Artsimovich, T. A. Fadeeva, N. V. Klimova, et al., Immunologiya, No. 6, 21–23 (1990).
A. V. Valdman, Yu. V. Burov, M. M. Borisov, et al., RU Pat. 2,038,081, Jun. 27, 1995; Byull. Izobret., No. 17 (1995).
R. S. Mirzoyan, T. S. Gan’shina, D. V. Maslennikov, et al., Eksp. Klin. Farmakol., 75(6), 27–30 (2012).
D. V. Maslennikov, N. I. Avdyunina, and B. M. Pyatin, in: Abstracts of Papers of the IVth Convention of Russian Pharmacologists [in Russian], Kazan (2012), p. 129.
M. I. Shmaryan, N. V. Klimova, L. N. Lavrova, et al., USSR Pat. 974,757, Sept. 27, 1995; Byull. Izobret., No. 31 (1995).
N. V. Klimova, N. I. Avdjunina, B. M. Pjatin, et al., RU Pat. 2,104,994, Feb. 20, 1998; Byull. Izobret., No. 3 (1998).
E. E. Finkel’shtein, S. V. Kurbatova, and E. A. Kolosova, Vestn. Samar. Gos. Univ., No. 4, 117–124 (2002).
M. N. Zemtsova, S. V. Kurbatova, I. K. Moiseev, et al., Zh. Fiz. Khim., 73, 1654–1657 (1999).
K. K. Konstantinova, V. G. Berezkin, S. V. Kurbatova, et al., Zh. Prikl. Khim., 78(9), 1522–1526 (2005).
S. S. Boiko, V. P. Zherdev, N. A. Kislyak, et al., Eksp. Klin. Farmakol., 57(6), 48–50 (1994).
USSR State Pharmacopoeia, XIth Ed., No. 1, Meditsina, Moscow (1987), pp. 199–251.
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 47, No. 6, pp. 44 – 50, June, 2013.
Rights and permissions
About this article
Cite this article
Tolkacheva, A.V., Grushevskaya, L.N., Avdyunina, N.I. et al. Development and validation of a GC-FID method for quantitation of impurities in kemantane drug substance. Pharm Chem J 47, 330–336 (2013). https://doi.org/10.1007/s11094-013-0955-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-013-0955-9