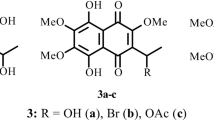
Preparative methods have been developed for the synthesis of azidonaphthazarins. It is established that 2-hydroxy- and 2-alkoxy-6,7-dichloronaphthazarins react regiospecifically with sodium azide in MeOH to yield the corresponding 6-azido derivatives. The cytotoxic activity and contraceptive properties of the synthesized azidonaphthazarins have been investigated using the sperm and eggs of the sea urchin Strongylocentrotus intermedius.


Similar content being viewed by others
Notes
Structures of naphthazarin derivatives are given herein only for one of the possible tautomers if not otherwise specified.
References
N. D. Pokhilo, A. Ya. Yakubovskaya, V. A. Denisenko, et al., Tetrahedron Lett. , 47(9), 1385–1387 (2006).
V. P. Anufriev and V. L. Novikov, Tetrahedron Lett., 36(14), 2515–2518 (1995).
V. P. Anufriev, V. L. Novikov, O. B. Maximov, et al., Bioorg. Med. Chem. Lett., 8(6), 587–592 (1998).
M. Matsuoka, K. Hamano, and T. Kitao, Synthesis, No. 11, 953–955 (1984).
H. Nakazumi, K. Kondo, and T. Kitao, Synthesis, No. 10, 878 (1982).
M. Service and A. C. Wardlaw, Comp. Biochem. Physiol., 79, 161–165 (1984).
GB Pat. No. 2,159,056; Chem. Abstr., 104, 83795 (1986).
L. V. Boguslavskaya, N. G. Khrapova, and O. B. Maksimov, Izv. Akad. Nauk, Ser. Khim., No. 7, 1471–1476 (1985).
RF Pat. No. 98,118,370 (1998); Chem. Abstr., 132, 284239b (2000).
RF Pat. No. 98,118,369 (1998); Chem. Abstr., 132, 284240v (2000).
V. P. Glazunov, A. Ya. Chizhova, M. I. Shuvalova, et al., Izv. Akad. Nauk, Ser. Khim., No. 1, 85–91 (2001).
P. A. Dinnel, J. M. Link, and O. J. Stober, Arch. Environ. Contam. Toxicol., 16, 23–32 (1987).
J. Bergquist, C. Stranberg, M. Andersson, et al., Toxicol. in Vitro, 73(3), 205–212 (1993).
N. D. Pokhilo, M. I. Kiseleva, V. V. Makhan?kov, et al., Khim. Prir. Soedin., No. 3, 228–231 (2008).
G. V. Malinovskaya, A. Ya. Chizhova, and V. P. Anufriev, Izv. Akad. Nauk, Ser. Khim., No. 5, 1019–1020 (1999).
V. P. Anufriev, N. N. Balaneva, A. Ya. Chizhova, et al., dep. VINITI, No. 6275-B-88; Ref. Zh. Khim., 23Zh154 (1988).
RF Pat. No. 2,193,550 (2002); Chem. Abstr., 139, 350545w (2003).
V. P. Anufriev, V. L. Novikov, G. V. Malinovskaya, et al., Synth. Commun., 27(1), 119–126 (1997).
RF Pat. No. 2,277,083 (2006); Chem. Abstr., 145, 7881j (2006).
G. A. Buznikov and V. K. Podmarev, in: Objects for Biological Development [in Russian], T. A. Ditmar (ed.), Nauka, Moscow (1975), pp. 188–216.
N. G. Prokof?eva, E. L. Chaikina, A. A. Kicha, et al., Comp. Biochem. Physiol., Part B, 134, 695–701 (2003).
M. I. Kiseleva, N. M. Shevchenko, T. N. Krupnova, et al., Zh. Evol. Biokhim. Fiziol., 41(1), 51–57 (2005).
M. I. Kiseleva, L. A. Balabanova, B. A. Rasskazov, et al., Mar. Biotechnol., 10(4), 466–470 (2008).
N. Kobayashi, in: Ecotoxicological Testing for the Marine Environment, G. Persoone, E. Jaspers, and C. Claus (eds.), State Univ. Ghent and Inst. Mar. Sci. Res., Bredene, Belgium (1984), Vol. 1, pp. 341–405.
M. M. Anisimov, N. G. Prokofyeva, L. Y. Korotkikh, et al., Toxicon, 18, 221–223 (1980).
M. M. Anisimov, G. N. Likhatskaya, E. B. Shentsova, et al., Biol. Morya (Vladivostok), 21(6), 417–420 (1995).
P. A. Dinnel, Biol. Morya (Vladivostok), 21(6), 390–397 (1995).
Acknowledgments
The work was financed partially by the Interdisciplinary Integrated Project of the Far-East, Siberian, and Urals Branches of the RAS (No. 09-II-SU-05-001) and the RAS Presidium Program “Molecular and Cellular Biology”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 45, No. 9, pp. 8–11, September, 2011.
Rights and permissions
About this article
Cite this article
Pokhilo, N.D., Kiseleva, M.I. & Anufriev, V.F. Synthesis and cytotoxic activity of azidonaphthazarins. Pharm Chem J 45, 522–525 (2011). https://doi.org/10.1007/s11094-011-0671-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-011-0671-2