Abstract
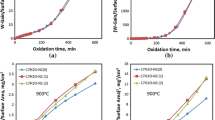
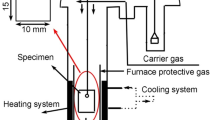
Under certain assumptions, Levich derived an equation for calculating gas-phase mass transport rate. This equation is commonly used to calculate the linear oxidation rate constant when the oxidation kinetics is controlled by gas phase transport and has been used in this study to calculate oxidation kinetics of steel at 850–1,180 °C in flowing air. In theory, at the early stage, two kinetics components, one linear and another parabolic, simultaneously operate at different locations of the sample, contributing to the overall kinetics and therefore, the oxidation kinetics should show an initial mixed linear-and-parabolic pattern, followed by a purely parabolic stage. However, the actual gas flow pattern in a laboratory tube furnace does not meet the requirements for deriving the Levich’s equation and as a result, significant discrepancies are found between the calculated and experimental results. To resolve the discrepancy, experimental conditions should be modified to meet the gas flow conditions required for deriving the Levich’s equation. Alternatively, a more appropriate equation under the current gas flow conditions in a laboratory tube furnace should be derived.





Similar content being viewed by others
References
R. Y. Chen and W. Y. D. Yuen, Oxidation of Metals 70, 39 (2008).
R. Y. Chen and W. Y. D. Yuen, Oxidation of Metals 73, 353 (2010).
D. R. Gaskell, An Introduction to Transport Phenomena in Materials Engineering (Macmillan Publishing Company, New York, 1992).
D. J. Young, High Temperature Oxidation and Corrosion of Metals, 1st edn. (Elsevier, Amsterdam, 2008), p. 69.
H. Abuluwefa, R. I. L. Guthrie, and F. Ajersch, Oxidation of Metals 46, 423 (1996).
H.-J. Selenz and F. Oeters, Archiv für das Eisenhüttenwesen 55, 201 (1984).
V. H. J. Lee, B. Gleeson, and D. J. Young, Oxidation of Metals 63, 15 (2005).
V. G. Levich, Physicochemical Hydrodynamics (Prentice-Hall, Englewood Cliffs, NJ, 1962).
C. Wagner, Atom Movements (ASM, Cleveland, OH, 1951), p. 153.
L. Himmel, R. F. Mehl, and C. E. Birchenall, Transactions of the AIME 5, 827 (1953).
R. Y. Chen and W. Y. D. Yuen, Metallurgical and Materials Transactions A 40A, 3091 (2009).
W. K. Chen and N. L. Peterson, The Journal of Physics and Chemistry of Solids 36, 1097 (1975).
G. Garnaud and R. A. Rapp, Oxidation of Metals 11, 193 (1977).
Acknowledgment
Permission from the management of BlueScope Steel Research for publication of the information contained in this paper is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yuen, W.Y.D., Chen, R.Y. Short-time Oxidation Behavior of Low-carbon, Low-silicon Steel in Air at 850–1,180 °C—III: Mixed Linear-and-Parabolic to Parabolic Transition Determined Using Local Mass-Transport Theories. Oxid Met 74, 255–274 (2010). https://doi.org/10.1007/s11085-010-9212-8
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-010-9212-8