Abstract
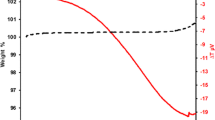
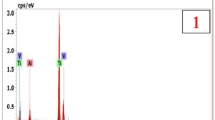
Thermogravimetric analysis (TGA) testing was used to measure the change in weight of polished samples of Al–XSi (X = 0 and 1.2 mass%) alloys. The samples were heated at 843 K for 6 h in dry air or nitrogen gas. X-ray diffraction was used to monitor the formation of the oxide films on the surface of the samples. The surface oxide films were more compact after the Al alloy samples were heated in air, and the oxide films showed some cracks after being heated in nitrogen gas. The thermally formed surface oxide films on the Al–1.2 mass% Si alloy samples heated in air and in nitrogen gas possessed loose structures, which comprised mainly γ-alumina, diaspore, and gibbsite, along with metallic silicon and/or aluminum. The weight variation curve of the films appeared serrated; this can be attributed to chain reactions (3Si + 3O2 → 3SiO2 + 4Al → 3Si + 2Al2O3) that occurred within the film.











Similar content being viewed by others
References
P. F. Doherty and R. S. Davis, Journal of Applied Physics 34, 619 (1963).
A. F. Beck, M. A. Heine, E. J. Caule, and M. Yor, Corrosion Science 7, 1 (1967).
M. J. Dignam, W. R. Faucett, and H. Bohnei, Journal of the Electrochemical Society 113, 656 (1966).
J. I. Eldridge, R. J. Hussey, D. F. Mitchell, and M. J. Graham, Oxidation of Metals 30, 301 (1988).
T. S. Shih and Z. B. Liu, Materials Transactions 475, 1347 (2006).
T. S. Shih and I. C. Chen, Materials Transactions 468, 1868 (2005).
L. W. Huang, P. W. Wang, T. S. Shih, and J. H. Liou, Materials Transactions 43, 2913 (2002).
Q. Hou, R. Mutharasan, and M. Koczak, Materials Science & Engineering: A 195, 121 (1995).
Q. Zheng and R. G. Reddy, Journal of Materials Science 39, 141 (2004).
P. S. Santos, H. S. Santos, and S. P. Toledo, Materials Research 3, 104 (2000).
C. Novak, G. Pokol, V. Izvekov, and T. Gal, Journal of Thermal Analysis 36, 1895 (1990).
Q. Chen, W. Zeng, S. Gu, G. Yang, H. Zhou, and Z. Yin, Journal of Chemical Thermodynamics 27, 443 (1995).
R. A. Robie, B. S. Hemingway, and J. R. Fisher, Thermodynamic Properties of Mineral and Related Substance at 298.15 K and 1 bar (10 5 Pascals) Pressure and at Higher Temperatures (Washington DC, US Govt. Print. Off., 1978), p. 18, 19, 184–185.
T. Tsuchida, Solid State Ionics 63–65, 464 (1993).
L. Yang, D. Zhu, C. Xu, and J. Zhang, Metallurgical and Materials Transactions. A, Physical Metallurgy and Materials Science A 27, 2094 (1996).
B. C. Lippens and J. J. Steggerda, Physics and Chemical Aspects of Absorbents and Catalysts (Academic Press, London/New York, 1970), p. 191.
S. J. Wilson, Journal of Solid State Chemistry 30, 247 (1979).
Acknowledgement
The authors would like to thank the National Science Council for providing financial support for this study (NSC95-2221-E-008-040).
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Constructed solid model of γ-alumina showing the void size (approximately 0.11–.0.12 nm).

Rights and permissions
About this article
Cite this article
Shih, TS., Chen, PC. & Huang, CC. Thermally Formed Oxide Films on Al–XSi Alloys Heated in Different Gases. Oxid Met 70, 69–83 (2008). https://doi.org/10.1007/s11085-008-9112-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-008-9112-3