Abstract
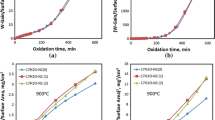
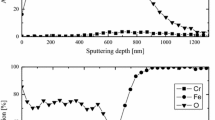
The oxidation and copper enrichment behaviors of several copper-containing mild steels under isothermal and step-isothermal conditions at 980–1220°C in ambient air, and the effects of nickel additions, are examined. The oxidation kinetics for all steels does not obey the parabolic law because of the formation of blisters in the scale. At 980°C, decreased parabolic oxidation kinetics is observed, whereas at 1120 and 1220°C, the oxidation kinetics exhibits an undulating pattern. High nickel content and high oxidation temperature are two prerequisites for the occlusion mechanism to operate during steel oxidation. For the low nickel steel, a planar scale-steel interface develops at all temperatures, and a copper phase is always seen to form and spread along the scale-steel interface. For the high nickel steels, a planar scale-steel interface develops at 980 and 1120°C, but at 1220°C, the scale-steel interface becomes rugged and the copper-rich phase is occluded into the scale. Introduction of a 980°C and/or 1220°C oxidation step significantly affects the copper enrichment behaviors of all steels normally exhibited at 1120°C.
Similar content being viewed by others
References
C. M. Smithyman (1996) Iron & Steelmaker 23 IssueID8 47
K. Noro M. Takeuchi Y. Mizukami (1997) ISIJ International 37 198
E. T. Stephenson (1983) Metallurgical Transaction A 14A 343
K. Sachs (1979) Metal Technology 6 33
L. Habraken, and J. Lecomte-Beckers, in Copper in Iron and Steel, I. Le May, and L. M. Schetky eds. (John Wiley & Sons, 1982), pp. 45–81.
D. A. Melford (1980) Philosophical Transactions of the Royal Society London A 295 89
D. A. Melford (1962) Journal of the Iron Steel Institute 200 290
W. J. M. Salter (1966) Journal of the Iron Steel Institute 204 478
A. R. Cox J. M. Winn (1965) Journal of the Iron Steel Institute 203 175
A. Nicholson J. D. Murray (1965) Journal of the Iron Steel Institute 203 1007
M. I. Copeland, and J. E. Kelley, U.S. Bureau Mines Report of Investigations 7682 (United States Department of the Interior, 1972).
P. J. Lewis (1993) Ironmaking and Steelmaking 20 126
R. Rolls A. Preece (1960) Metal Treatment and Drop Forging 27 139
D. A. Melford (1966) Journal of the Iron Steel Institute 204 495
M. I. Copeland, and J. S. Howe, U.S. Bureau Mines Report of Investigations 8080 (United States Department of the Interior, 1975)
M. I. Copeland, U.S. Bureau Mines Report of Investigations 7936 (United States Department of the Interior, 1974).
G. L. Fisher (1969) Journal of the Iron Steel Institute 207 1010
O. T. Llewellyn (1995) Ironmaking and Steelmaking 22 25
Metal-Induced Embrittlement, Metal Handbook, ASM International, 10th ed. (ASM International, Materials Park, Ohio, 1990), Vol. 1, p. 717.
T. Kajitani M. Wakoh N. Tokumitsu S. Ogibayashi S. Mizoguchi (1995) Tetsu to Hagane 81 185
R. Y. Chen, and W. Y. D. Yuen, unpublished results, 1996.
D. Caplan (1960) Journal of Electrochemistry Society 107 359
R. Y. Chen W. Y. D. Yuen (2003) Oxidation of Metals 59 433
T. Asai T. Soshiroda M. Miyahara (1997) ISIJ International 37 272
L. J. Swartzendruber, in Binary Alloy Phase Diagrams, T. B. Massalski, H. O. Okamoto, P. R. Subramanian, and L. Kacprzak, eds. (ASM International, 1990), pp. 1408–1410.
B. D. Bastow D. H. Kirkwood (1971) Journal of the Institute of Metals 99 277
H. J. Grabke V. Leroy H. Viefhaus (1995) ISIJ International 35 95
Y. Zou E. W. Langer (1989) Materials Science and Engineering A 110 203
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, R.Y., Yuen, W.Y.D. Isothermal and Step Isothermal Oxidation of Copper-Containing Steels in Air at 980–1220°C. Oxid Met 63, 145–168 (2005). https://doi.org/10.1007/s11085-004-3197-0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11085-004-3197-0