Abstract
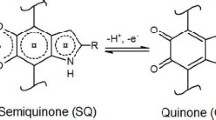
The source of the optical properties of natural melanin persists as an impressive conundrum, hindering a thorough comprehension of the complicated photo-protective contribution of these extensive natural pigments and the pragmatic design of revolutionary bioinspired materials for optoelectronic functionalities. Melanin is a natural pigment distributed in vivo, and provides protection from the sun rays, exhibiting a strong wide-band UV–visible absorption. In the present study, we investigated the optical properties of natural melanin at different pH concentrations within the framework of experimental techniques, namely, UV–visible absorption, Fourier transform infrared spectroscopy (FTIR), and fluorescence spectroscopies. The results indicate that the optical absorption and fluorescence of natural melanin are enhanced within the near-UV and visible regimes with an increase in the solution concentration, are dependent on the pH level, and can impact the emergence of a broad band of visible absorption. The variations in the optical absorption spectra of natural melanin solutions at various pH levels will demonstrate novel rational strategies for controlling their optical properties for potential optoelectronic applications.






Similar content being viewed by others
References
Alotto, P., Guarnieri, M., Moro, F.: Redox flow batteries for the storage of renewable energy: a review. Renew. Sustain. Energy Rev. 29, 325–335 (2014). https://doi.org/10.1016/j.rser.2013.08.001
Ambrico, M., Ambrico, P.F., Cardone, A., Ligonzo, T., Cicco, S.R., Mundo, R.Di, Farinola, G.M.: Melanin layer on silicon: an attractive structure for a possible exploitation in bio-polymer based metal-insulator-silicon devices. Adv. Mater. 23(29), 3332–3336 (2011). https://doi.org/10.1002/adma.201101358
Ambrico, M., Ambrico, P.F., Ligonzo, T., Cardone, A., Cicco, S.R., D’Ischia, M., Farinola, G.M.: From commercial tyrosine polymers to a tailored polydopamine platform: concepts, issues and challenges en route to melanin-based bioelectronics. J. Mater. Chem. C 3, 6413–6423 (2015). https://doi.org/10.1039/C5TC00570A
Ball, V., Del Frari, D., Michel, M., Buehler, M.J., Toniazzo, V., Singh, M.K., Ruch, D.: Deposition mechanism and properties of thin polydopamine films for high added value applications in surface science at the nanoscale. BioNanoScience 2, 16–34 (2012). https://doi.org/10.1007/s12668-011-0032-3
Bernsmann, F., Ponche, A., Ringwald, C., Hemmerle, J., Raya, J., Bechinger, B., Ball, V.: Characterization of dopamine-melanin growth on silicon oxide. J. Phys. Chem. C 113(19), 8234–8242 (2009). https://doi.org/10.1021/jp901188h
Bochenek, K., Gudowska-Nowak, E.: Electronic properties of random polymers: modelling optical spectra of melanins. Acta Phys. Pol. B 34(5), 2775–2790 (2003)
Bonavolontà, C., de Lisio, C., d’Ischia, M., Maddalena, P., Manini, P., Pezzella, A., Valentino, M.: Anomalous evolution of broadband optical absorption reveals dynamic solid state reorganization during eumelanin build-up in thin films. Sci. Rep. (2017). https://doi.org/10.1038/s41598-017-00597-8
Bothma, J.P., De Boor, J., Divakar, U., Schwenn, P.E., Meredith, P.: Device-quality electrically conducting melanin thin films. Adv. Mater. 20(18), 3539–3542 (2008). https://doi.org/10.1002/adma.200703141
Bridelli, M.G., Crippa, P.R.: Theoretical analysis of the adsorption of metal ions to the surface of melanin particles. Adsorption 14(1), 101–109 (2008). https://doi.org/10.1007/s10450-007-9059-8
Capozzi, V., Perna, G., Carmone, P., Gallone, A., Lastella, M., Mezzenga, E., Cicero, R.: Optical and photoelectronic properties of melanin. Thin Solid Films 511–512, 362–366 (2006). https://doi.org/10.1016/j.tsf.2005.12.065
Chen, C.T., Ball, V., De Almeida Gracio, J.J., Singh, M.K., Toniazzo, V., Ruch, D., Buehler, M.J.: Self-assembly of tetramers of 5,6-dihydroxyindole explains the primary physical properties of eumelanin: experiment, simulation, and design. ACS Nano 7(2), 1524–1532 (2013). https://doi.org/10.1021/nn305305d
Chen, C.-T., Chuang, C., Cao, J., Ball, V., Ruch, D., Buehler, M.J.: Excitonic effects from geometric order and disorder explain broadband optical absorption in eumelanin. Nat. Commun. 5, 3859 (2014). https://doi.org/10.1038/ncomms4859
Costa, T.G., Younger, R., Poe, C., Farmer, P.J., Szpoganicz, B.: Studies on synthetic and natural melanin and its affinity for Fe(III) ion. Chem. Appl, Bioinorg (2012). https://doi.org/10.1155/2012/712840
Dadachova, E., Bryan, R.A., Huang, X., Moadel, T., Schweitzer, A.D., Aisen, P., Casadevall, A.: Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi. PLoS ONE 2(5) (2007). https://doi.org/10.1371/journal.pone.0000457
De Albuquerque, J.E., Giacomantonio, C., White, A.G., Meredith, P.: Determination of thermal and optical parameters of melanins by photopyroelectric spectroscopy. Appl. Phys. Lett. 87(6), 061920 (2005). https://doi.org/10.1063/1.2009833
Denis, J.C., Schumacher, S., Galbraith, I.: Quantitative description of interactions between linear organic chromophores. Journal of Chemical Physics 137(22), 224102 (2012). https://doi.org/10.1063/1.4768244
El-Obeid, A., Hassib, A., Pontén, F., Westermark, B.: Effect of herbal melanin on IL-8: a possible role of Toll-like receptor 4 (TLR4). Biochem. Biophys. Res. Commun. 344(4), 1200–1206 (2006). https://doi.org/10.1016/j.bbrc.2006.04.035
Giacomantonio, C.: Charge transport in melanin, a disordered bio-organic conductor. University of Queensland. Retrieved from https://people.smp.uq.edu.au/BenPowell/Clare_Giacomantonio_Hons_thesis.pdf (2005)
Guimard, N.K., Guimard, N.K., Gomez, N., Gomez, N., Schmidt, C.E., Schmidt, C.E.: Conducting polymers in biomedical engineering. Prog. Polym. Sci. 32, 876–921 (2007). https://doi.org/10.1016/j.progpolymsci.2007.05.012
Jastrzebska, M., Kocot, A., Tajber, L.: Photoconductivity of synthetic dopa–melanin polymer. J. Photochem. Photobiol. B Biol. 66, 201–206 (2002)
Kim, I.G., Nam, H.J., Ahn, H.J., Jung, D.-Y.: Electrochemical growth of synthetic melanin thin films by constant potential methods. Electrochim. Acta 56(7), 2954–2959 (2011). https://doi.org/10.1016/j.electacta.2010.12.095
Klosterman, L., Bettinger, C.J.: Calcium-mediated control of polydopamine film oxidation and iron chelation. Int. J. Mol. Sci. 18(1), 1–14 (2017). https://doi.org/10.3390/ijms18010014
Lee, C.W., Kim, O.Y., Lee, J.Y.: Organic materials for organic electronic devices. J. Ind. Eng. Chem. 20, 1198–1208 (2014). https://doi.org/10.1016/j.jiec.2013.09.036
Ligonzo, T., Ambrico, M., Augelli, V., Perna, G., Schiavulli, L., Tamma, M.A., Capozzi, V.: Electrical and optical properties of natural and synthetic melanin biopolymer. J. Non-Cryst. Solids 355(22–23), 1221–1226 (2009). https://doi.org/10.1016/j.jnoncrysol.2009.05.014
Liu, Y., Simon, J.D.: Metal-ion interactions and the structural organization of Sepia eumelanin. Pigment Cell Res. 18(1), 42–48 (2005). https://doi.org/10.1111/j.1600-0749.2004.00197.x
Mbonyiryivuze, A., Omollo, I., Ngom, B.D., Mwakikunga, B.: Natural dye sensitizer for Gr ӓ tzel cells: sepia melanin. Phys. Mater. Chem. 3(1), 1–6 (2015). https://doi.org/10.12691/pmc-3-1-1
Meller, G., Grasser, T.: Organic electronics. Polym. Sci, Adv (2010). https://doi.org/10.1007/978-3-642-04538-7
Meng, S., Kaxiras, E.: Theoretical models of eumelanin protomolecules and their optical properties. Biophys. J. 94(6), 2095–2105 (2008). https://doi.org/10.1529/biophysj.107.121087
Morresi, L., Ficcadenti, M., Pinto, N., Murri, R., Cuccioloni, M., Angeletti, M., Tombesi, P.: Optical and electrical behavior of synthetic melanin thin films spray-coated. Energy Procedia 2, 177–182 (2010). https://doi.org/10.1016/j.egypro.2010.07.025
Napolitano, A., Pezzella, A., Prota, G., Seraglia, R., Traldi, P.: Structural analysis of synthetic melanins from 5,6-dihydroxyindole by matrix-assisted laser desorption ionization mass spectrometry. Rapid Commun. Mass Spectrom. 10(4), 468–472 (1996)
Obeid, M.T., Hussain, W.A., Radhi, W.A.: Linear optical properties of pheomelanine pigment extraction from red wool. J. Zankoy Sulaimani Part A 17(1), 177–184 (2015). https://doi.org/10.17656/jzs.10371
Riley, P.A.: Melanin. Int. J. Biochem. Cell Biol. 29(11), 1235–1239 (1997). https://doi.org/10.1016/S1357-2725(97)00013-7
Sardar, D.K., Mayo, M.L., Glickman, R.D.: Optical characterization of melanin. J. Biomed. Opt. 6(4), 404–411 (2001)
Selvaraju, S., Adhikari, S., Hopson, R.A., Dai, S., Rheingold, A.L., Borunda, M.F., Nelson, T.L.: Effects of structural variations on the optical and electronic properties of eumelanin-inspired small molecules. J. Mater. Chem. C 4, 3875–3875 (2016)
Stark, K.B., Gallas, J.M., Zajac, G.W., Eisner, M., Golab, J.T.: Spectroscopic study and simulation from recent structural models for eumelanin: I. Monomer, dimers. J. Phys. Chem. B 107(41), 3061–3067 (2003). https://doi.org/10.1021/jp034965r
Svennersten, K., Larsson, K.C., Berggren, M., Richter-Dahlfors, A.: Organic bioelectronics in nanomedicine. Biochim. Biophys. Acta Gen. Subj. 1810, 276–285 (2011). https://doi.org/10.1016/j.bbagen.2010.10.001
Vairale, P., Waman, V., Mayabadi, A., Pathan, H.: Electrochemical synthesis of melanin thin films: evolution of structural and optical properties. Int. J. Adv. Res. Phys. Sci. 1(7), 35–45 (2014)
Zonios, G., Bykowski J., Kollias N.: Skin melanin, hemoglobin, and light scattering properties can be quantitatively assessed in vivo. J. Investig. Dermatol. 117(6), 1452–1457 (2001)
Zvyagin, I.: Charge transport via delocalized states in disordered materials. In: Charge Transport in Disordered Solids with Applications in Electronics, pp. 1–47 (2006). https://doi.org/10.1002/0470095067.ch1
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Madkhali, N., Alqahtani, H., Al-Terary, S. et al. Control of optical absorption and fluorescence spectroscopies of natural melanin at different solution concentrations. Opt Quant Electron 51, 227 (2019). https://doi.org/10.1007/s11082-019-1936-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11082-019-1936-3