Abstract
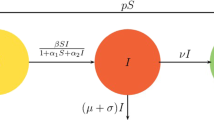
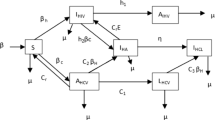
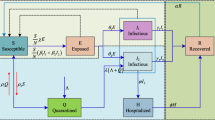
Few existing studies establish the fact that although human pathogen exists in the environmental reservoir, infectious disease outbreak is rare. In this article, we investigate the dynamics of cholera disease where the transmission occurs via contact with a contaminated reservoir. A deterministic iSIR compartmental epidemic model is formulated and explored the realistic situation, in which the quantity of ingested pathogens must exceed a critical threshold, the minimal infectious dose (MID), in order to infect healthy population. Bacteria growth rate is considered to follow the Allee effect. This model system shows multi-stability and undergoes transcritical and saddle-node bifurcations with respect to various parameters. Thus, external environmental noise can give rise to observable dynamics for the stochastic counterpart. One can see a noise-induced frequent transitions between two separate interior attractors, in particular for the cases where the deterministic model displays bistability. The global sensitivity using PRCC method is performed. Also, the optimal control for the developed deterministic cholera epidemic model is carried out with numerical illustrations. Our model primarily designed to explain and predict the indirectly transmitted cholera epidemic model incorporating the idea of MID. This study gives a different outlook for the transmission of disease from the human-centered perspective to socioeconomic context.


















Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the article.
References
https://www.who.int/emergencies/disease-outbreak-news/item/2022-don426 (2022)
Baishya, M., Chakraborti, C.: Non-equilibrium fluctuation in Volterra–Lotka systems. Bull. Math. Biol. 49, 125–131 (1987)
Bandyopadhyay, M., Chakrabarti, C.: Deterministic and stochastic analysis of a nonlinear prey-predator system. J. Biol. Syst. 11(02), 161–172 (2003)
Bandyopadhyay, M., Chattopadhyay, J.: Ratio-dependent predator-prey model: effect of environmental fluctuation and stability. Nonlinearity 18(2), 913 (2005)
Berhe, H.W.: Optimal control strategies and cost-effectiveness analysis applied to real data of cholera outbreak in Ethiopia’s Oromia region. Chaos, Solitons Fractals 138, 109933 (2020)
Capasso, V., Paveri-Fontana, S.: A mathematical model for the 1973 cholera epidemic in the European Mediterranean region. Rev. d’Epidemiol. Sante Publique 27(2), 121–132 (1979)
Capone, F., Carfora, M.F., De Luca, R., Torcicollo, I.: Analysis of a model for waterborne diseases with Allee effect on bacteria. Nonlinear Anal. Model. Control 25(6), 1035–1058 (2020)
Carletti, M.: On the stability properties of a stochastic model for phage-bacteria interaction in open marine environment. Math. Biosci. 175(2), 117–131 (2002)
Chitsulo, L., Engels, D., Montresor, A., Savioli, L.: The global status of schistosomiasis and its control. Acta Trop. 77(1), 41–51 (2000)
Codeço, C.T.: Endemic and epidemic dynamics of cholera: the role of the aquatic reservoir. BMC Infect. Dis. 1(1), 1–14 (2001)
Colwell, R., Brayton, P., Herrington, D., Tall, B., Huq, A., Levine, M.: Viable but non-culturable vibrio cholerae o1 revert to a cultivable state in the human intestine. World J. Microbiol. Biotechnol. 12, 28–31 (1996)
Das, P., Das, P., Mukherjee, S.: Stochastic dynamics of Michaelis–Menten kinetics based tumor-immune interactions. Phys. A Stat. Mech. Appl. 541, 123603 (2020)
Das, P., Mondal, P., Das, P., Roy, T.K.: Stochastic persistence and extinction in tumor-immune system perturbed by white noise. Int. J. Dyn. Control 10(2), 620–629 (2022)
Das, P., Mukherjee, S., Das, P., Banerjee, S.: Characterizing chaos and multifractality in noise-assisted tumor-immune interplay. Nonlinear Dyn. 101(1), 675–685 (2020)
Das, P., Upadhyay, R.K., Misra, A.K., Rihan, F.A., Das, P., Ghosh, D.: Mathematical model of Covid-19 with comorbidity and controlling using non-pharmaceutical interventions and vaccination. Nonlinear Dyn. 106(2), 1213–1227 (2021)
Fields, B.S., Benson, R.F., Besser, R.E.: Legionella and legionnaires’ disease: 25 years of investigation. Clin. Microbiol. Rev. 15(3), 506–526 (2002)
Ghosh, M., Das, P., Das, P.: A comparative study of deterministic and stochastic dynamics of rumor propagation model with counter-rumor spreader. Nonlinear Dyn. 111(18), 16875 (2023)
Habees, A.A., Aldabbas, E., Bragazzi, N.L., Kong, J.D.: Bacteria-bacteriophage cycles facilitate cholera outbreak cycles: an indirect susceptible-infected-recovered-bacteria-phage (isirbp) model-based mathematical study. J. Biol. Dyn. 16(1), 29–43 (2022)
Hartley, D.M., Morris, J.G., Jr., Smith, D.L.: Hyperinfectivity: a critical element in the ability of v. cholerae to cause epidemics? PLoS Med. 3(1), e7 (2006)
Janeway, C., Murphy, K.P., Travers, P., Walport, M.: Janeway’s Immuno Biology. Garland Science, New York, NY (2008)
Jensen, M.A., Faruque, S.M., Mekalanos, J.J., Levin, B.R.: Modeling the role of bacteriophage in the control of cholera outbreaks. Proc. Natl. Acad. Sci. 103(12), 4652–4657 (2006)
Joh, R.I., Wang, H., Weiss, H., Weitz, J.S.: Dynamics of indirectly transmitted infectious diseases with immunological threshold. Bull. Math. Biol. 71, 845–862 (2009)
Karatzas, I., Karatzas, I., Shreve, S., Shreve, S.E.: Brownian Motion and Stochastic Calculus, vol. 113. Springer, Cham (1991)
Kaul, R.B., Kramer, A.M., Dobbs, F.C., Drake, J.M.: Experimental demonstration of an Allee effect in microbial populations. Biol. Lett. 12(4), 20160070 (2016)
Kong, J.D., Davis, W., Li, X., Wang, H.: Stability and sensitivity analysis of the iSIR model for indirectly transmitted infectious diseases with immunological threshold. SIAM J. Appl. Math. 74(5), 1418–1441 (2014)
Lemos-Paião, A.P., Silva, C.J., Torres, D.F.: An epidemic model for cholera with optimal control treatment. J. Comput. Appl. Math. 318, 168–180 (2017)
Levine, M.M., Black, R.E., Clemens, M.L., Nalin, D.R., Cisneros, L., Finkelstein, R.A.: Volunteer Studies in Development of Vaccines Against Cholera and Enterotoxigenic Eschericha coli: A Review [Book Chapter]. Elsevier/North-Holland Biomedical Press, Amsterdam, The Netherlands (1981)
Marino, S., Hogue, I.B., Ray, C.J., Kirschner, D.E.: A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 254(1), 178–196 (2008)
May, R.M.: Stability and Complexity in Model Ecosystems. Princeton University Press, Princeton, NJ (2019)
Mukandavire, Z., Liao, S., Wang, J., Gaff, H., Smith, D.L., Morris, J.G., Jr.: Estimating the reproductive numbers for the 2008–2009 cholera outbreaks in zimbabwe. Proc. Natl. Acad. Sci. 108(21), 8767–8772 (2011)
Panja, P.: Optimal control analysis of a cholera epidemic model. Biophys. Rev. Lett. 14(01), 27–48 (2019)
Phan, T.A., Tian, J.P., Wang, B.: Dynamics of cholera epidemic models in fluctuating environments. Stoch. Dyn. 21(02), 2150011 (2021)
Rose, J.B.: Environmental ecology of cryptosporidium and public health implications. Annu. Rev. Public Health 18(1), 135–161 (1997)
Settati, A., Lahrouz, A., Assadouq, A., El Fatini, M., El Jarroudi, M., Wang, K.: The impact of nonlinear relapse and reinfection to derive a stochastic threshold for SIRI epidemic model. Chaos Solitons Fractals 137, 109897 (2020)
Song, Y., Yuan, S.: Bifurcation analysis in a predator-prey system with time delay. Nonlinear Anal. Real World Appl. 7(2), 265–284 (2006)
Sotomayor, J.: Generic bifurcations of dynamical systems. In: Dynamical Systems, pp. 561–582. Elsevier, Amsterdam (1973)
Tapaswi, P., Mukhopadhyay, A.: Effects of environmental fluctuation on plankton allelopathy. J. Math. Biol. 39, 39–58 (1999)
Tian, J.P., Wang, J.: Global stability for cholera epidemic models. Math. Biosci. 232(1), 31–41 (2011)
Upadhyay, R.K., Banerjee, M., Parshad, R., Raw, S.N.: Deterministic chaos versus stochastic oscillation in a prey-predator-top predator model. Math. Model. Anal. 16(3), 343–364 (2011)
Wolfe, M.S.: Giardiasis. Clin. Microbiol. Rev. 5(1), 93–100 (1992)
Wu, J., Dhingra, R., Gambhir, M., Remais, J.V.: Sensitivity analysis of infectious disease models: methods, advances and their application. J. R. Soc. Interface 10(86), 20121018 (2013)
Yang, X., Chen, L., Chen, J.: Permanence and positive periodic solution for the single-species nonautonomous delay diffusive models. Comput. Math. Appl. 32(4), 109–116 (1996)
Zhang, X., Peng, H.: Stationary distribution of a stochastic cholera epidemic model with vaccination under regime switching. Appl. Math. Lett. 102, 106095 (2020)
Zhou, X., Shi, X., Wei, M.: Dynamical behavior and optimal control of a stochastic mathematical model for cholera. Chaos Solitons Fractals 156, 111854 (2022)
Funding
No funding is available for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this article.
Ethical approval
The authors state that this research complies with ethical standards. This research does not involve either human participants or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Acharya, S., Mondal, B., Upadhyay, R.K. et al. Exploring noise-induced dynamics and optimal control strategy of iSIR cholera transmission model. Nonlinear Dyn 112, 3951–3975 (2024). https://doi.org/10.1007/s11071-023-09221-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-023-09221-z