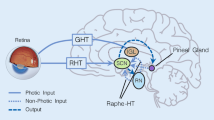
Abstract
Phase-splitting and various arrhythmic behaviors have been observed in mammals. Although it is known that desynchronization of SCN neurons plays an important role, the exact mechanisms underlying this behavior are still not completely understood. A previous study suggested that phase-splitting behavior was explained by the SCN output signal feeding back to the SCN with a time delay of about 12 h. However, experimental evidence showed that the feedback is instantaneous and does not have a time delay. In the present study, a Kuramoto model with instant feedback is proposed to explain the mechanisms underlying phase splitting and arrhythmic behavior. Our results show that phase-splitting and arrhythmic behavior results from the feedback which is described by two key parameters, the strength of the feedback as well as the phase difference of the feedback to the SCN activity. Additionally, in accordance with experimental findings, we observe that the SCN period in the split state is equal to 24 h, while it is larger than 24 h for a state that has a robust circadian rhythm. Our study provides an alternative explanation for the phase-splitting and arrhythmic behavior and opens a window to understand how feedback influences the SCN, and consequently its behavior.





Similar content being viewed by others
References
Czeisler, C.A., Duffy, J.F., Shanahan, T.L., Brown, E.N., Mitchell, J.F., Rimmer, D.W., Ronda, J.M., Silva, E.J., Allan, J.S., Emens, J.S.: Stability, precision, and near-24-hour period of the human circadian pacemaker. Science 284(5423), 2177 (1999)
Daido, H.: Why circadian rhythms are circadian: competitive population dynamics of biological oscillators. Phys. Rev. Lett. 87(4), 048101 (2001)
Gu, C., Wang, J., Liu, Z.: Free-running period of neurons in the suprachiasmatic nucleus: its dependence on the distribution of neuronal coupling strengths. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 80(3 Pt 1), 030904 (2009)
Welsh, D.K., Joseph, S., Takahashi, Kay, Steve A.: Suprachiasmatic nucleus: cell autonomy and network properties. Annu. Rev. Physiol. 72(1), 551–577 (2010)
Refinetti, R.: Circadian Physiology, 2nd edn. CRC Press, Boca Raton (2005)
Welsh, D.K., Logothetis, D.E., Meister, M., Reppert, S.M.: Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14(4), 697–706 (1995)
Honma, S., Nakamura, W., Shirakawa, T., Honma, K.: Diversity in the circadian periods of single neurons of the rat suprachiasmatic nucleus depends on nuclear structure and intrinsic period. Neurosci. Lett. 358(3), 173–176 (2004)
Yamaguchi, S., Isejima, H., Matsuo, T., Okura, R., Yagita, K., Kobayashi, M., Okamura, H.: Synchronization of cellular clocks in the suprachiasmatic nucleus. Science 302(5649), 1408–1412 (2003)
Albus, H., Mariska, J., Vansteensel, Stephan, M., Gene, D.B., Johanna, H.M.: A gabaergic mechanism is necessary for coupling dissociable ventral and dorsal regional oscillators within the circadian clock. Curr. Biol. 15(10), 886–893 (2005)
Sara, J.A., Christopher, S.C., Anthony, J.H., Waschek, J., Erik, D.H.: Vasoactive intestinal polypeptide mediates circadian rhythmicity and synchrony in mammalian clock neurons. Nat. Neurosci. 8(4), 1129–1135 (2005)
La, Horacio O.D., Meyer, J., Carpino, A., Schwartz, W.J.: Antiphase oscillation of the left and right suprachiasmatic nuclei. Science 290(5492), 799–801 (2000)
Ohta, H., Yamazaki, S., Mcmahon, D.G.: Constant light desynchronizes mammalian clock neurons. Nat. Neurosci. 8(3), 267–269 (2005)
Indic, P., Schwartz, W.J., Paydarfar, D.: Design principles for phase-splitting behaviour of coupled cellular oscillators: clues from hamsters with ‘split’ circadian rhythms. J. R. Soc. Interface 5(25), 873–883 (2008)
Gu, C., Wang, J., Wang, J., Liu, Z.: Mechanism of phase splitting in two coupled groups of suprachiasmatic-nucleus neurons. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 83(4 Pt 2), 046224 (2011)
Van, O.F., Lucassen, E.A., Houben, T., Vanderleest, H.T., Antle, M.C., Meijer, J.H.: Amplitude of the SCN clock enhanced by the behavioral activity rhythm. PLoS ONE 7(6), e39693 (2012)
Schroder, S., Herzog, E.D., Kiss, I.Z.: Transcription-based oscillator model for light-induced splitting as antiphase circadian gene expression in the suprachiasmatic nuclei. J. Biol. Rhythms 27(1), 79–90 (2012)
Pittendrigh, C.S., Daan, S.: A functional analysis of circadian pacemakers in nocturnal rodents. J. Comp. Physiol. 106(3), 291–331 (1976)
Acknowledgements
The work is supported by the National Natural Science Foundation of China under Grant Nos. 11875042 and 11505114, the Program for Professor of Special Appointment (Orientational Scholar) at Shanghai Institutions of Higher Learning under Grant Nos. D-USST02 and QD2015016, and the Shanghai project for construction of discipline peaks. Jos Rohling was supported by the Netherlands Organization for Scientific Research under Complexity Grant No. 645.000.010.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gu, C., Gu, X., Ren, H. et al. Splitting between two subgroups of the SCN neurons with instantaneous feedback. Nonlinear Dyn 97, 1245–1251 (2019). https://doi.org/10.1007/s11071-019-05044-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-019-05044-z