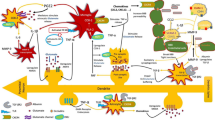
Abstract
Epilepsy is a chronic neurological disease that is characterized by spontaneous and recurrent seizures. Regulated cell death is a controlled process and has been shown to be involved in neurodegenerative diseases. Necroptosis is a type of regulated cell death, and its association with epilepsy has been documented. Necroptosis signaling can be divided into two pathways: canonical and non-canonical pathways. Inhibition of caspase-8, dimerization of receptor-interacting protein kinase 1 (RIP1) and RIP3, activation of mixed-lineage kinase domain-like protein (MLKL), movement of MLKL to the plasma membrane, and cell rupture occurred in these pathways. Through literature review, it has been revealed that there is a relationship between seizure, neuroinflammation, and oxidative stress. The seizure activity triggers the activation of various pathways within the central nervous system, including TNF-α/matrix metalloproteases, Neogenin and Calpain/ Jun N-terminal Kinase 1, which result in distinct responses in the brain. These responses involve the activation of neurons and astrocytes, consequently leading to an increase in the expression levels of proteins and genes such as RIP1, RIP3, and MLKL in a time-dependent manner in regions such as the hippocampus (CA1, CA3, dentate gyrus, and hilus), piriform cortex, and amygdala. Furthermore, the imbalance in calcium ions, depletion of adenosine triphosphate, and elevation of extracellular glutamate and potassium within these pathways lead to the progression of necroptosis, a reduction in seizure threshold, and increased susceptibility to epilepsy. Therefore, it is plausible that therapeutic targeting of these pathways could potentially provide a promising approach for managing seizures and epilepsy.


Similar content being viewed by others
Data Availability
No data were utilized in the research described in the manuscript.
References
Scharfman HE (2007) The neurobiology of epilepsy. Curr Neurol Neurosci Rep 7:348–354. https://doi.org/10.1007/s11910-007-0053-z
Beghi E (2020) The epidemiology of epilepsy. Neuroepidemiology 54:185–191. https://doi.org/10.1159/000503831
Lee SK (2019) Epilepsy in the elderly: treatment and consideration of comorbid diseases. J Epilepsy Res 9:27–35. https://doi.org/10.14581/jer.19003
Stewart E, Lah S, Smith ML (2019) Patterns of impaired social cognition in children and adolescents with epilepsy: the borders between different epilepsy phenotypes. Epilepsy Behav 100:106146. https://doi.org/10.1016/j.yebeh.2019.01.031
Blume WT (2003) Diagnosis and management of epilepsy. CMAJ 168:441–448
Shorvon SD (2011) The etiologic classification of epilepsy. Epilepsia 52:1052–1057. https://doi.org/10.1111/j.1528-1167.2011.03041.x
Vezzani A, French J, Bartfai T, Baram TZ (2011) The role of inflammation in epilepsy. Nat Rev Neurol 7:31–40. https://doi.org/10.1038/nrneurol.2010.178
Rana A, Musto AE (2018) The role of inflammation in the development of epilepsy. J Neuroinflamm 15:144. https://doi.org/10.1186/s12974-018-1192-7
Ho YH, Lin YT, Wu CW, Chao YM, Chang AY, Chan JY (2015) Peripheral inflammation increases seizure susceptibility via the induction of neuroinflammation and oxidative stress in the hippocampus. J Biomed Sci 22:46. https://doi.org/10.1186/s12929-015-0157-8
Fabisiak T, Patel M (2022) Crosstalk between neuroinflammation and oxidative stress in epilepsy. Front Cell Dev Biol 10:976953. https://doi.org/10.3389/fcell.2022.976953
Fricker M, Tolkovsky AM, Borutaite V, Coleman M, Brown GC (2018) Neuronal cell death. Physiol Rev 98:813–880. https://doi.org/10.1152/physrev.00011.2017
Du K, He M, Zhao D, Wang Y, Ma C, Liang H, Wang W, Min D, Xue L, Guo F (2022) Mechanism of cell death pathways in status epilepticus and related therapeutic agents. Biomed Pharmacother 149:112875. https://doi.org/10.1016/j.biopha.2022.112875
Atabaki R, Khaleghzadeh-Ahangar H, Esmaeili N, Mohseni-Moghaddam P (2023) Role of pyroptosis, a pro-inflammatory programmed cell death, in epilepsy. Cell Mol Neurobiol 43:1049–1059. https://doi.org/10.1007/s10571-022-01250-3
Newton K, Manning G (2016) Necroptosis and inflammation. Annu Rev Biochem 85:743–763. https://doi.org/10.1146/annurev-biochem-060815-014830
Dhuriya YK, Sharma D (2018) Necroptosis: a regulated inflammatory mode of cell death. J Neuroinflamm 15:1–9. https://doi.org/10.1186/s12974-018-1192-7
Pasparakis M, Vandenabeele P (2015) Necroptosis and its role in inflammation. Nature 517:311–320. https://doi.org/10.1038/nature14191
Lu Z, Van Eeckhoutte HP, Liu G, Nair PM, Jones B, Gillis CM, Nalkurthi BC, Verhamme F, Buyle-Huybrecht T, Vandenabeele P (2021) Necroptosis signaling promotes inflammation, airway remodeling, and emphysema in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 204:667–681. https://doi.org/10.1164/rccm.202009-3442OC
Wang R, Li H, Wu J, Cai Z-Y, Li B, Ni H, Qiu X, Chen H, Liu W, Yang Z-H (2020) Gut stem cell necroptosis by genome instability triggers bowel inflammation. Nature 580:386–390. https://doi.org/10.1038/s41586-020-2127-x
Cai Q, Gan J, Luo R, Qu Y, Li S, Wan C, Mu D (2017) The role of necroptosis in status epilepticus-induced brain injury in juvenile rats. Epilepsy Behav 75:134–142. https://doi.org/10.1016/j.yebeh.2017.05.025
Yan WT, Lu S, Yang YD, Ning WY, Cai Y, Hu XM, Zhang Q, Xiong K (2021) Research trends, hot spots and prospects for necroptosis in the field of neuroscience. Neural Regen Res 16:1628–1637. https://doi.org/10.4103/1673-5374.303032
Hsu H, Shu HB, Pan MG, Goeddel DV (1996) TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways. Cell 84:299–308. https://doi.org/10.1016/s0092-8674(00)80984-8
Füllsack S, Rosenthal A, Wajant H, Siegmund D (2019) Redundant and receptor-specific activities of TRADD, RIPK1 and FADD in death receptor signaling. Cell Death Dis 10:122. https://doi.org/10.1038/s41419-019-1396-5
Micheau O, Tschopp J (2003) Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114:181–190. https://doi.org/10.1016/s0092-8674(03)00521-x
Zhang DW, Shao J, Lin J, Zhang N, Lu BJ, Lin SC, Dong MQ, Han J (2009) RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325:332–336. https://doi.org/10.1126/science.1172308
Zhang Y, Chen X, Gueydan C, Han J (2018) Plasma membrane changes during programmed cell deaths. Cell Res 28:9–21. https://doi.org/10.1038/cr.2017.133
Wu XN, Yang ZH, Wang XK, Zhang Y, Wan H, Song Y, Chen X, Shao J, Han J (2014) Distinct roles of RIP1–RIP3 hetero- and RIP3–RIP3 homo-interaction in mediating necroptosis. Cell Death Differ 21:1709–1720. https://doi.org/10.1038/cdd.2014.77
Wang H, Sun L, Su L, Rizo J, Liu L, Wang LF, Wang FS, Wang X (2014) Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol Cell 54:133–146. https://doi.org/10.1016/j.molcel.2014.03.003
Kaiser WJ, Sridharan H, Huang C, Mandal P, Upton JW, Gough PJ, Sehon CA, Marquis RW, Bertin J, Mocarski ES (2013) Toll-like receptor 3-mediated necrosis via TRIF, RIP3, and MLKL. J Biol Chem 288:31268–31279. https://doi.org/10.1074/jbc.M113.462341
Thapa RJ, Nogusa S, Chen P, Maki JL, Lerro A, Andrake M, Rall GF, Degterev A, Balachandran S (2013) Interferon-induced RIP1/RIP3-mediated necrosis requires PKR and is licensed by FADD and caspases. Proc Natl Acad Sci U S A 110:E3109–3118. https://doi.org/10.1073/pnas.1301218110
McComb S, Cessford E, Alturki NA, Joseph J, Shutinoski B, Startek JB, Gamero AM, Mossman KL, Sad S (2014) Type-I interferon signaling through ISGF3 complex is required for sustained Rip3 activation and necroptosis in macrophages. Proc Natl Acad Sci U S A 111:E3206–E3213. https://doi.org/10.1073/pnas.1407068111
Wang J, Liu Y, Li XH, Zeng XC, Li J, Zhou J, Xiao B, Hu K (2017) Curcumin protects neuronal cells against status-epilepticus-induced hippocampal damage through induction of autophagy and inhibition of necroptosis. Can J Physiol Pharmacol 95:501–509. https://doi.org/10.1139/cjpp-2016-0154
Wang J, Li Y, Huang WH, Zeng XC, Li XH, Li J, Zhou J, Xiao J, Xiao B, Ouyang DS, Hu K (2017) The protective effect of aucubin from Eucommia ulmoides against status epilepticus by inducing autophagy and inhibiting necroptosis. Am J Chin Med 45:557–573. https://doi.org/10.1142/S0192415X17500331
Jia R, Jia N, Yang F, Liu Z, Li R, Jiang Y, Zhao J, Wang L, Zhang S, Zhang Z, Zhang H, Wu S, Gao F, Jiang W (2019) Hydrogen alleviates necroptosis and cognitive deficits in lithium–pilocarpine model of status epilepticus. Cell Mol Neurobiol 39:857–869. https://doi.org/10.1007/s10571-019-00685-5
Lin DQ, Cai XY, Wang CH, Yang B, Liang RS (2020) Optimal concentration of necrostatin-1 for protecting against hippocampal neuronal damage in mice with status epilepticus. Neural Regen Res 15:936–943. https://doi.org/10.4103/1673-5374.268903
Degterev A, Maki JL, Yuan J (2013) Activity and specificity of necrostatin-1, small-molecule inhibitor of RIP1 kinase. Cell Death Differ 20:366. https://doi.org/10.1038/cdd.2012.133
Murru S, Hess S, Barth E, Almajan ER, Schatton D, Hermans S, Brodesser S, Langer T, Kloppenburg P, Rugarli EI (2019) Astrocyte-specific deletion of the mitochondrial m-AAA protease reveals glial contribution to neurodegeneration. Glia 67:1526–1541. https://doi.org/10.1002/glia.23626
Younis NS, Mohamed ME, Alolayan AA, Alhussain GY, Al-Mousa HA, Alshamrani JA, AlMutayib MM, AlQahtani MM, Alhaddad ZA, Alfarhan ZS, AlOmran ZA, Almostafa MM (2022) Identification of epilepsy concomitant candidate genes recognized in Saudi epileptic patients. Eur Rev Med Pharmacol Sci 26:2143–2157. https://doi.org/10.26355/eurrev_202203_28362
Muona M, Berkovic SF, Dibbens LM et al (2015) A recurrent de novo mutation in KCNC1 causes progressive myoclonus epilepsy. Nat Genet 47:39–46. https://doi.org/10.1038/ng.3144
Wu Z, Deshpande T, Henning L, Bedner P, Seifert G, Steinhäuser C (2021) Cell death of hippocampal CA1 astrocytes during early epileptogenesis. Epilepsia 62:1569–1583. https://doi.org/10.1111/epi.16910
Zhao XM, Chen Z, Zhao JB, Zhang PP, Pu YF, Jiang SH, Hou JJ, Cui YM, Jia XL, Zhang SQ (2016) Hsp90 modulates the stability of MLKL and is required for TNF-induced necroptosis. Cell Death Dis 7:e2089. https://doi.org/10.1038/cddis.2015.390
Bedner P, Dupper A, Hüttmann K, Müller J, Herde MK, Dublin P, Deshpande T, Schramm J, Häussler U, Haas CA, Henneberger C, Theis M, Steinhäuser C (2015) Astrocyte uncoupling as a cause of human temporal lobe epilepsy. Brain 138:1208–1222. https://doi.org/10.1093/brain/awv067
Chen PY, Tsai YW, Chang AY, Chang HH, Hsiao YH, Huang CW, Sung PS, Chen BH, Fu TF (2020) Increased leptin-b expression and metalloprotease expression contributed to the pyridoxine-associated toxicity in zebrafish larvae displaying seizure-like behavior. Biochem Pharmacol 182:114294. https://doi.org/10.1016/j.bcp.2020.114294
Li C, Gu H, Yu M, Yang P, Zhang M, Ba H, Yin Y, Wang J, Yin B, Zhou X, Li Z (2019) Inhibition of transmembrane TNF-α shedding by a specific antibody protects against septic shock. Cell Death Dis 10:586. https://doi.org/10.1038/s41419-019-1808-6
Becker-Pauly C, Rose-John S (2013) TNFα cleavage beyond TACE/ADAM17: matrix metalloproteinase 13 is a potential therapeutic target in sepsis and colitis. EMBO Mol Med 5:970–972. https://doi.org/10.1002/emmm.201302899
Sun W, Wu X, Gao H, Yu J, Zhao W, Lu JJ, Wang J, Du G, Chen X (2017) Cytosolic calcium mediates RIP1/RIP3 complex-dependent necroptosis through JNK activation and mitochondrial ROS production in human colon cancer cells. Free Radical Biol Med 108:433–444. https://doi.org/10.1016/j.freeradbiomed.2017.04.010
Huang WY, Lai YL, Liu KH, Lin S, Chen HY, Liang CH, Wu HM, Hsu KS (2022) TNFα-mediated necroptosis in brain endothelial cells as a potential mechanism of increased seizure susceptibility in mice following systemic inflammation. J Neuroinflamm 19:29. https://doi.org/10.1186/s12974-022-02406-0
Zhang S, Xie H, Wang Y, Li D, Du L, Wu Y, Yang G-Y (2017) Enriched environment improves behavioral performance and attenuates inflammatory response induced by TNF-α in healthy adult mice. Eur J Inflam 15:200–209. https://doi.org/10.1177/1721727x17730471
Hu X, Xu Y, Zhang H, Li Y, Wang X, Xu C, Ni W, Zhou K (2022) Role of necroptosis in traumatic brain and spinal cord injuries. J Adv Res 40:125–134. https://doi.org/10.1016/j.jare.2021.12.002
Chen AQ, Fang Z, Chen XL, Yang S, Zhou YF, Mao L, Xia YP, Jin HJ, Li YN, You MF, Wang XX, Lei H, He QW, Hu B (2019) Microglia-derived TNF-α mediates endothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke. Cell Death Dis 10:487. https://doi.org/10.1038/s41419-019-1716-9
Moerke C, Jaco I, Dewitz C, Müller T, Jacobsen AV, Gautheron J, Fritsch J, Schmitz J, Bräsen JH, Günther C, Murphy JM, Kunzendorf U, Meier P, Krautwald S (2019) The anticonvulsive Phenhydan® suppresses extrinsic cell death. Cell Death Differ 26:1631–1645. https://doi.org/10.1038/s41418-018-0232-2
Abd El-Aal SA, El-Abhar HS, Abulfadl YS (2022) Morin offsets PTZ-induced neuronal degeneration and cognitive decrements in rats: the. Eur J Pharmacol 931:175213. https://doi.org/10.1016/j.ejphar.2022.175213. modulation of TNF-α/TNFR-1/RIPK1,3/MLKL/PGAM5/Drp-1, IL-6/JAK2/STAT3/GFAP and Keap-1/Nrf-2/HO-1 trajectories
Choi IY, Shim JH, Kim MH, Yu WD, Kim YJ, Choi G, Lee JH, Kim HJ, Cho KO (2021) Truncated neogenin promotes hippocampal neuronal death after acute seizure. Neuroscience 470:78–87. https://doi.org/10.1016/j.neuroscience.2021.06.039
Wilson NH, Key B (2007) Neogenin: one receptor, many functions. Int J Biochem Cell Biol 39:874–878. https://doi.org/10.1016/j.biocel.2006.10.023
Sun D, Tan ZB, Sun XD, Liu ZP, Chen WB, Milibari L, Ren X, Yao LL, Lee D, Shen C, Pan JX, Huang ZH, Mei L, Xiong WC (2021) Hippocampal astrocytic neogenin regulating glutamate uptake, a critical pathway for preventing epileptic response. Proc Natl Acad Sci U S A 118:e2022921118. https://doi.org/10.1073/pnas.2022921118
Bollino D, Balan I, Aurelian L (2015) Valproic acid induces neuronal cell death through a novel calpain-dependent necroptosis pathway. J Neurochem 133:174–186. https://doi.org/10.1111/jnc.13029
Lam PM, Carlsen J, González MI (2017) A calpain inhibitor ameliorates seizure burden in an experimental model of temporal lobe epilepsy. Neurobiol Dis 102:1–10. https://doi.org/10.1016/j.nbd.2017.02.003
Jayaraman A, Htike TT, James R, Picon C, Reynolds R (2021) TNF-mediated neuroinflammation is linked to neuronal necroptosis in Alzheimer’s disease hippocampus. Acta Neuropathol Commun 9:159. https://doi.org/10.1186/s40478-021-01264-w
Bronisz E, Kurkowska-Jastrzębska I (2016) Matrix metalloproteinase 9 in epilepsy: the role of neuroinflammation in seizure development. Mediators Inflamm 2016:7369020. https://doi.org/10.1155/2016/7369020
Dubey D, McRae PA, Rankin-Gee EK, Baranov E, Wandrey L, Rogers S, Porter BE (2017) Increased metalloproteinase activity in the hippocampus following status epilepticus. Epilepsy Res 132:50–58. https://doi.org/10.1016/j.eplepsyres.2017.02.021
von Rüden EL, Zellinger C, Gedon J, Walker A, Bierling V, Deeg CA, Hauck SM, Potschka H (2020) Regulation of Alzheimer’s disease-associated proteins during epileptogenesis. Neuroscience 424:102–120. https://doi.org/10.1016/j.neuroscience.2019.08.037
Hsia HE, Tüshaus J, Brummer T, Zheng Y, Scilabra SD, Lichtenthaler SF (2019) Functions of 'A disintegrin and metalloproteases (ADAMs)' in the mammalian nervous system. Cell Mol Life Sci 76:3055–3081. https://doi.org/10.1007/s00018-019-03173-7
Rempe RG, Hartz AMS, Soldner ELB, Sokola BS, Alluri SR, Abner EL, Kryscio RJ, Pekcec A, Schlichtiger J, Bauer B (2018) Matrix metalloproteinase-mediated blood-brain barrier dysfunction in epilepsy. J Neurosci 38:4301–4315. https://doi.org/10.1523/jneurosci.2751-17.2018
Sha L, Wang X, Li J, Shi X, Wu L, Shen Y, Xu Q (2017) Pharmacologic inhibition of Hsp90 to prevent GLT-1 degradation as an effective therapy for epilepsy. J Exp Med 214:547–563. https://doi.org/10.1084/jem.20160667
Jacobsen AV, Lowes KN, Tanzer MC, Lucet IS, Hildebrand JM, Petrie EJ, van Delft MF, Liu Z, Conos SA, Zhang JG, Huang DC, Silke J, Lessene G, Murphy JM (2016) HSP90 activity is required for MLKL oligomerisation and membrane translocation and the induction of necroptotic cell death. Cell Death Dis 7:e2051. https://doi.org/10.1038/cddis.2015.386
Kovac S, Domijan AM, Walker MC, Abramov AY (2012) Prolonged seizure activity impairs mitochondrial bioenergetics and induces cell death. J Cell Sci 125:1796–1806. https://doi.org/10.1242/jcs.099176
Nazıroğlu M, Övey İS (2015) Involvement of apoptosis and calcium accumulation through TRPV1 channels in neurobiology of epilepsy. Neuroscience 293:55–66. https://doi.org/10.1016/j.neuroscience.2015.02.041
Bernardi P (1999) Mitochondrial transport of cations: channels, exchangers, and permeability transition. Physiol Rev 79:1127–1155. https://doi.org/10.1152/physrev.1999.79.4.1127
Sisodia SS, St George-Hyslop PH (2002) gamma-Secretase, Notch, Abeta and Alzheimer’s disease: where do the presenilins fit in? Nat Rev Neurosci 3:281–290. https://doi.org/10.1038/nrn785
Kimberly WT, LaVoie MJ, Ostaszewski BL, Ye W, Wolfe MS, Selkoe DJ (2003) Gamma-secretase is a membrane protein complex comprised of presenilin, nicastrin, Aph-1, and Pen-2. Proc Natl Acad Sci U S A 100:6382–6387. https://doi.org/10.1073/pnas.1037392100
Cai Z, Zhang A, Choksi S, Li W, Li T, Zhang XM, Liu ZG (2016) Activation of cell-surface proteases promotes necroptosis, inflammation and cell migration. Cell Res 26:886–900. https://doi.org/10.1038/cr.2016.87
Hernández DE, Salvadores NA, Moya-Alvarado G, Catalán RJ, Bronfman FC, Court FA (2018) Axonal degeneration induced by glutamate excitotoxicity is mediated by necroptosis. J Cell Sci 131:jcs214684. https://doi.org/10.1242/jcs.214684
Halestrap AP (2009) What is the mitochondrial permeability transition pore? J Mol Cell Cardiol 46:821–831. https://doi.org/10.1016/j.yjmcc.2009.02.021
Nikseresht S, Khodagholi F, Dargahi L, Ahmadiani A (2017) Necroptosis resumes apoptosis in hippocampus but not in frontal cortex. J Cell Biochem 118:4628–4638. https://doi.org/10.1002/jcb.26127
Lerman-Sagie T, Watemberg N, Kramer U, Shahar E, Lerman P (2001) Absence seizures aggravated by valproic acid. Epilepsia 42:941–943. https://doi.org/10.1046/j.1528-1157.2001.042007941.x
Belcastro V, Caraballo RH, Romeo A, Striano P (2013) Early-onset absence epilepsy aggravated by valproic acid: a video-EEG report. Epileptic Disord 15:440–443. https://doi.org/10.1684/epd.2013.0616
Romoli M, Mazzocchetti P, D'Alonzo R, Siliquini S, Rinaldi VE, Verrotti A, Calabresi P, Costa C (2019) Valproic acid and epilepsy: from molecular mechanisms to clinical evidences. Curr Neuropharmacol 17:926–946. https://doi.org/10.2174/1570159x17666181227165722
Hussein AM, Awadalla A, Abbas KM, Sakr HF, Elghaba R, Othman G, Mokhtar N, Helal GM (2021) Chronic valproic acid administration enhances oxidative stress, upregulates IL6 and downregulates Nrf2, Glut1 and Glut4 in rat’s liver and brain. NeuroReport 32:840–850. https://doi.org/10.1097/wnr.0000000000001663
Salimi A, Alyan N, Akbari N, Jamali Z, Pourahmad J (2022) Selenium and L-carnitine protects from valproic acid-Induced oxidative stress and mitochondrial damages in rat cortical neurons. Drug Chem Toxicol 45:1150–1157. https://doi.org/10.1080/01480545.2020.1810259
Vosler PS, Brennan CS, Chen J (2008) Calpain-mediated signaling mechanisms in neuronal injury and neurodegeneration. Mol Neurobiol 38:78–100. https://doi.org/10.1007/s12035-008-8036-x
Zhang L, Ma N, Liu Q, Ma Y (2013) Genome-wide screening for genes associated with valproic acid sensitivity in fission yeast. PLoS ONE 8:e68738. https://doi.org/10.1371/journal.pone.0068738
Desfossés-Baron K, Hammond-Martel I, Simoneau A, Sellam A, Roberts S, Wurtele H (2016) Valproate inhibits MAP kinase signalling and cell cycle progression in S. cerevisiae. Sci Rep 6:36013. https://doi.org/10.1038/srep36013
Wahab A (2010) Difficulties in treatment and management of epilepsy and challenges in new drug development. Pharmaceuticals (Basel) 3:2090–2110. https://doi.org/10.3390/ph3072090
Molnár T, Pallagi P, Tél B, Király R, Csoma E, Jenei V, Varga Z, Gogolák P, Odile Hueber A, Máté Z, Erdélyi F, Szabó G, Pettkó-Szandtner A, Bácsi A, Virág L, Maléth J, Koncz G (2021) Caspase-9 acts as a regulator of necroptotic cell death. FEBS J 288:6476–6491. https://doi.org/10.1111/febs.15898
Acknowledgments
This study did not receive any financial support.
Funding
This study did not receive any funding.
Author information
Authors and Affiliations
Contributions
RA contributed to the study’s conception and design. The introduction section of the manuscript was written by HKA, and all subsequent sections were written by RA in the first draft. HKA also contributed to the design of the figures. PMM commented on previous versions of the manuscript and provided valuable grammatical and scientific editing, as well as the initial idea for the study. The final version of the manuscript has been reviewed and approved by all authors.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
The authors mentioned have participated in this manuscript.
Consent to Publish
The authors read and approved the publication of this manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohseni-Moghaddam, P., Khaleghzadeh-Ahangar, H. & Atabaki, R. Role of Necroptosis, a Regulated Cell Death, in Seizure and Epilepsy. Neurochem Res 49, 1–13 (2024). https://doi.org/10.1007/s11064-023-04010-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-023-04010-x