Abstract
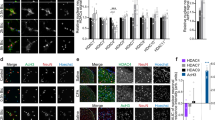
Epigenetic regulation of gene expression has been implicated in the development of chronic pain. However, little is known about whether this regulation is involved in the development and treatment of chronic pain comorbidities such as fibromyalgia syndrome (FMS) and temporomandibular disorder (TMD), a comorbidity predominantly occurring among women. Here we explored the impact of the histone deacetylase (HDAC) inhibitor suberoylanilide hydroxamic acid (SAHA) on somatic hyperalgesia induced by stress or stress combined with orofacial inflammation, which mimicked the comorbidity of FMS and TMD in rats. Our data showed that somatic thermal hyperalgesia and mechanical allodynia induced by both conditions were completely prevented by intrathecal injection of SAHA, which upregulated 5-HT2C receptors but downregulated 5-HT3 receptors in the spinal dorsal horn. Subsequent spinal administration of RS102221 to inhibit 5-HT2C receptors or SR57227 to activate 5-HT3 receptors reversed the analgesic effect of SAHA under both conditions. These results indicate that SAHA attenuates the pro-nociceptive effects of stress combined with orofacial inflammation and the effects of stress alone. This likely occurs through epigenetic regulation of spinal 5-HT2C and 5-HT3 receptor expression, suggesting that SAHA has potential therapeutic value in FMS or comorbid FMS-TMD patients with somatic hyperalgesia.






Similar content being viewed by others
References
Disease GBD, Injury I, Prevalence C (2017) Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the global burden of disease study 2016. Lancet 390(10100):1211–1259. https://doi.org/10.1016/S0140-6736(17)32154-2
Treede RD, Rief W, Barke A, Aziz Q, Bennett MI, Benoliel R, Cohen M, Evers S, Finnerup NB, First MB, Giamberardino MA, Kaasa S, Kosek E, Lavand’homme P, Nicholas M, Perrot S, Scholz J, Schug S, Smith BH, Svensson P, Vlaeyen JW, Wang SJ (2015) A classification of chronic pain for ICD-11. Pain 156(6):1003–1007. https://doi.org/10.1097/j.pain.0000000000000160
Clauw DJ (2015) Fibromyalgia and related conditions. Mayo Clin Proc 90(5):680–692. https://doi.org/10.1016/j.mayocp.2015.03.014
Greene CS (2010) Managing the care of patients with temporomandibular disorders: a new guideline for care. J Am Dent Assoc 141(9):1086–1088. https://doi.org/10.14219/jada.archive.2010.0337
Goldberg DS, McGee SJ (2011) Pain as a global public health priority. BMC Public Health 11:770. https://doi.org/10.1186/1471-2458-11-770
Cabo-Meseguer A, Cerda-Olmedo G, Trillo-Mata JL (2017) Fibromyalgia: prevalence, epidemiologic profiles and economic costs. Med Clin (Barc) 149(10):441–448. https://doi.org/10.1016/j.medcli.2017.06.008
Slade GD, Bair E, Greenspan JD, Dubner R, Fillingim RB, Diatchenko L, Maixner W, Knott C, Ohrbach R (2013) Signs and symptoms of first-onset tmd and sociodemographic predictors of its development: the oppera prospective cohort study. J Pain. https://doi.org/10.1016/j.jpain.2013.07.014
Queiroz LP (2013) Worldwide epidemiology of fibromyalgia. Curr Pain Headache Rep 17(8):356. https://doi.org/10.1007/s11916-013-0356-5
Isong U, Gansky SA, Plesh O (2008) Temporomandibular joint and muscle disorder-type pain in U.S. adults: the national health interview survey. J Orofac Pain 22(4):317–322
Fischer S, Doerr JM, Strahler J, Mewes R, Thieme K, Nater UM (2016) Stress exacerbates pain in the everyday lives of women with fibromyalgia syndrome—the role of cortisol and alpha-amylase. Psychoneuroendocrinology 63:68–77. https://doi.org/10.1016/j.psyneuen.2015.09.018
Staniszewski K, Lygre H, Bifulco E, Kvinnsland S, Willassen L, Helgeland E, Berge T, Rosen A (2018) Temporomandibular disorders related to stress and HPA-axis regulation. Pain Res Manag 2018:7020751. https://doi.org/10.1155/2018/7020751
Jones GT (2016) Psychosocial vulnerability and early life adversity as risk factors for central sensitivity syndromes. Curr Rheumatol Rev 12(2):140–153. https://doi.org/10.2174/1573397112666151231113438
Grossi PK, Bueno CH, de Abreu Silva MA, Pellizzer EP, Grossi ML (2018) Evaluation of sexual, physical, and emotional abuse in women diagnosed with temporomandibular disorders: a case-control study. Int J Prosthodont 31(6):543–551. https://doi.org/10.11607/ijp.5828
Jeffery DD, Bulathsinhala L, Kroc M, Dorris J (2014) Prevalence, health care utilization, and costs of fibromyalgia, irritable bowel, and chronic fatigue syndromes in the military health system, 2006-2010. Mil Med 179(9):1021–1029. https://doi.org/10.7205/MILMED-D-13-00419
White BA, Williams LA, Leben JR (2001) Health care utilization and cost among health maintenance organization members with temporomandibular disorders. J Orofac Pain 15(2):158–169
Li ZL, Xue Y, Tao ZY, Du WZ, Jiang YG, Cao DY (2019) Spinal 5-HT3 receptor contributes to somatic hyperalgesia induced by sub-chronic stress. Mol Pain 15:1744806919859723. https://doi.org/10.1177/1744806919859723
Li JH, Yang JL, Wei SQ, Li ZL, Collins AA, Zou M, Wei F, Cao DY (2020) Contribution of central sensitization to stress-induced spreading hyperalgesia in rats with orofacial inflammation. Mol Brain 13(1):106. https://doi.org/10.1186/s13041-020-00645-x
McClure JJ, Li X, Chou CJ (2018) Advances and challenges of HDAC inhibitors in cancer therapeutics. Adv Cancer Res 138:183–211. https://doi.org/10.1016/bs.acr.2018.02.006
Karsli-Ceppioglu S (2016) Epigenetic mechanisms in psychiatric diseases and epigenetic therapy. Drug Dev Res 77(7):407–413. https://doi.org/10.1002/ddr.21340
Descalzi G, Ikegami D, Ushijima T, Nestler EJ, Zachariou V, Narita M (2015) Epigenetic mechanisms of chronic pain. Trends Neurosci 38(4):237–246. https://doi.org/10.1016/j.tins.2015.02.001
Cao DY, Bai G, Ji Y, Traub RJ (2015) Epigenetic upregulation of metabotropic glutamate receptor 2 in the spinal cord attenuates oestrogen-induced visceral hypersensitivity. Gut 64(12):1913–1920. https://doi.org/10.1136/gutjnl-2014-307748
Xue Y, Wei SQ, Wang PX, Wang WY, Liu EQ, Traub RJ, Cao DY (2020) Down-regulation of spinal 5-HT2A and 5-HT2C receptors contributes to somatic hyperalgesia induced by orofacial inflammation combined with stress. Neuroscience 440:196–209. https://doi.org/10.1016/j.neuroscience.2020.05.044
Xu GZ, Xue Y, Wei SQ, Li JH, Traub RJ, Wang MD, Cao DY (2019) Valproate reverses stress-induced somatic hyperalgesia and visceral hypersensitivity by up-regulating spinal 5-HT2C receptor expression in female rats. Neuropharmacology 165:107926. https://doi.org/10.1016/j.neuropharm.2019.107926
Zhou L, He B, Deng J, Pang S, Tang H (2019) Histone acetylation promotes long-lasting defense responses and longevity following early life heat stress. PLoS Genet 15(4):e1008122. https://doi.org/10.1371/journal.pgen.1008122
Cao DY, Bai G, Ji Y, Karpowicz JM, Traub RJ (2016) Express: histone hyperacetylation modulates spinal type II metabotropic glutamate receptor alleviating stress-induced visceral hypersensitivity in female rats. Mol Pain. https://doi.org/10.1177/1744806916660722
Nakai K, Nakae A, Oba S, Mashimo T, Ueda K (2010) 5-HT2C receptor agonists attenuate pain-related behaviour in a rat model of trigeminal neuropathic pain. Eur J Pain 14(10):999–1006. https://doi.org/10.1016/j.ejpain.2010.04.008
Obata H, Saito S, Sakurazawa S, Sasaki M, Usui T, Goto F (2004) Antiallodynic effects of intrathecally administered 5-HT2C receptor agonists in rats with nerve injury. Pain 108(1–2):163–169. https://doi.org/10.1016/j.pain.2003.12.019
Morales M, Wang SD (2002) Differential composition of 5-hydroxytryptamine3 receptors synthesized in the rat cns and peripheral nervous system. J Neurosci 22(15):6732–6741
Guo W, Miyoshi K, Dubner R, Gu M, Li M, Liu J, Yang J, Zou S, Ren K, Noguchi K, Wei F (2014) Spinal 5-HT3 receptors mediate descending facilitation and contribute to behavioral hypersensitivity via a reciprocal neuron-glial signaling cascade. Mol Pain 10:35. https://doi.org/10.1186/1744-8069-10-35
Koca T, Kocyigit B, Seyithanoglu M, Berk E (2019) The importance of G-protein coupled estrogen receptor in patients with fibromyalgia. Arch Rheumatol 34(4):419–425. https://doi.org/10.5606/ArchRheumatol.2019.7236
Wise EA, Riley JL III, Robinson ME (2000) Clinical pain perception and hormone replacement therapy in postmenopausal women experiencing orofacial pain. Clin J Pain 16(2):121–126. https://doi.org/10.1097/00002508-200006000-00005
Taylor AG, Fischer-White TG, Anderson JG, Adelstein KE, Murugesan M, Lewis JE, Scott MM, Gaykema RP, Goehler LE (2016) Stress, inflammation and pain: A potential role for monocytes in fibromyalgia-related symptom severity. Stress Health 32(5):503–513. https://doi.org/10.1002/smi.2648
Coppens E, Kempke S, Van Wambeke P, Claes S, Morlion B, Luyten P, Van Oudenhove L (2018) Cortisol and subjective stress responses to acute psychosocial stress in fibromyalgia patients and control participants. Psychosom Med 80(3):317–326. https://doi.org/10.1097/PSY.0000000000000551
Lei J, Liu MQ, Fu KY (2016) Disturbed sleep, anxiety and stress are possible risk indicators for temporomandibular disorders with myofascial pain. Beijing Da Xue Xue Bao Yi Xue Ban 48(4):692–696
Marpaung C, Lobbezoo F, van Selms MKA (2018) Temporomandibular disorders among dutch adolescents: Prevalence and biological, psychological, and social risk indicators. Pain Res Manag 2018:5053709. https://doi.org/10.1155/2018/5053709
Casale R, Sarzi-Puttini P, Botto R, Alciati A, Batticciotto A, Marotto D, Torta R (2019) Fibromyalgia and the concept of resilience. Clin Exp Rheumatol 37(Suppl 116):105–113
Ayouni I, Chebbi R, Hela Z, Dhidah M (2019) Comorbidity between fibromyalgia and temporomandibular disorders: a systematic review. Oral Surg Oral Med Oral Pathol Oral Radiol 128(1):33–42. https://doi.org/10.1016/j.oooo.2019.02.023
Lin CR, Cheng JK, Wu CH, Chen KH, Liu CK (2017) Epigenetic suppression of potassium-chloride co-transporter 2 expression in inflammatory pain induced by complete freund’s adjuvant (CFA). Eur J Pain 21(2):309–321. https://doi.org/10.1002/ejp.925
Zhang Z, Cai YQ, Zou F, Bie B, Pan ZZ (2011) Epigenetic suppression of GAD65 expression mediates persistent pain. Nat Med 17(11):1448–1455. https://doi.org/10.1038/nm.2442
Danaher RJ, Zhang L, Donley CJ, Laungani NA, Hui SE, Miller CS, Westlund KN (2018) Histone deacetylase inhibitors prevent persistent hypersensitivity in an orofacial neuropathic pain model. Mol Pain 14:1744806918796763. https://doi.org/10.1177/1744806918796763
Schaefer EW, Loaiza-Bonilla A, Juckett M, DiPersio JF, Roy V, Slack J, Wu W, Laumann K, Espinoza-Delgado I, Gore SD, Mayo PCPIIC (2009) A phase 2 study of vorinostat in acute myeloid leukemia. Haematologica 94(10):1375–1382. https://doi.org/10.3324/haematol.2009.009217
Imbe H, Kimura A (2018) Increase of histone acetylation in the gabaergic neurons in the rostral ventromedial medulla associated with mechanical hypersensitivity after repeated restraint stress. Brain Res Bull 142:394–402. https://doi.org/10.1016/j.brainresbull.2018.09.004
Imbe H, Kimura A (2016) Repeated forced swim stress affects the expression of pCREB and deltaFosB and the acetylation of histone H3 in the rostral ventromedial medulla and locus coeruleus. Brain Res Bull 127:11–22. https://doi.org/10.1016/j.brainresbull.2016.08.007
Imbe H, Kimura A (2015) Repeated forced swim stress prior to complete freund’s adjuvant injection enhances mechanical hyperalgesia and attenuates the expression of pCREB and deltaFosB and the acetylation of histone H3 in the insular cortex of rat. Neuroscience 301:12–25. https://doi.org/10.1016/j.neuroscience.2015.05.065
Suzuki R, Rygh LJ, Dickenson AH (2004) Bad news from the brain: descending 5-HT pathways that control spinal pain processing. Trends Pharmacol Sci 25(12):613–617. https://doi.org/10.1016/j.tips.2004.10.002
Cortes-Altamirano JL, Olmos-Hernandez A, Jaime HB, Carrillo-Mora P, Bandala C, Reyes-Long S, Alfaro-Rodriguez A (2018) Review: 5-HT1, 5-HT2, 5-HT3 and 5-HT7 receptors and their role in the modulation of pain response in the central nervous system. Curr Neuropharmacol 16(2):210–221. https://doi.org/10.2174/1570159X15666170911121027
Tao ZY, Wang PX, Wei SQ, Traub RJ, Li JF, Cao DY (2019) The role of descending pain modulation in chronic primary pain: potential application of drugs targeting serotonergic system. Neural Plast. https://doi.org/10.1155/2019/1389296
Obata H, Ito N, Sasaki M, Saito S, Goto F (2007) Possible involvement of spinal noradrenergic mechanisms in the antiallodynic effect of intrathecally administered 5-HT2C receptor agonists in the rats with peripheral nerve injury. Eur J Pharmacol 567(1–2):89–94. https://doi.org/10.1016/j.ejphar.2007.03.029
Xu WJ, Wang YY, Zhao Y, Jia H, Tang JS, Huo FQ, Liu H (2020) Involvement of 5-HT2A, 5-HT2B and 5-HT2C receptors in mediating the ventrolateral orbital cortex-induced antiallodynia in a rat model of neuropathic pain. NeuroReport 31(2):167–173. https://doi.org/10.1097/WNR.0000000000001377
Baptista-de-Souza D, Pelarin V, Canto-de-Souza L, Nunes-de-Souza RL, Canto-de-Souza A (2018) Interplay between 5-HT2C and 5-HT1A receptors in the dorsal periaqueductal gray in the modulation of fear-induced antinociception in mice. Neuropharmacology 140:100–106. https://doi.org/10.1016/j.neuropharm.2018.07.027
de Oliveira R, de Oliveira RC, Falconi-Sobrinho LL, da Silva Soares R Jr, Coimbra NC (2017) 5-hydroxytryptamine2A/2C receptors of nucleus raphe magnus and gigantocellularis/paragigantocellularis pars alpha reticular nuclei modulate the unconditioned fear-induced antinociception evoked by electrical stimulation of deep layers of the superior colliculus and dorsal periaqueductal grey matter. Behav Brain Res 316:294–304. https://doi.org/10.1016/j.bbr.2016.09.016
Gregoire S, Neugebauer V (2013) 5-HT2CR blockade in the amygdala conveys analgesic efficacy to ssris in a rat model of arthritis pain. Mol Pain 9:41. https://doi.org/10.1186/1744-8069-9-41
Ji G, Zhang W, Mahimainathan L, Narasimhan M, Kiritoshi T, Fan X, Wang J, Green TA, Neugebauer V (2017) 5-HT2C receptor knockdown in the amygdala inhibits neuropathic-pain-related plasticity and behaviors. J Neurosci 37(6):1378–1393. https://doi.org/10.1523/JNEUROSCI.2468-16.2016
Kayser V, Elfassi IE, Aubel B, Melfort M, Julius D, Gingrich JA, Hamon M, Bourgoin S (2007) Mechanical, thermal and formalin-induced nociception is differentially altered in 5-HT1A-/-, 5-HT1B-/-, 5-HT2A-/-, 5-HT3A-/- and 5-HTT-/- knock-out male mice. Pain 130(3):235–248. https://doi.org/10.1016/j.pain.2006.11.015
Zeitz KP, Guy N, Malmberg AB, Dirajlal S, Martin WJ, Sun L, Bonhaus DW, Stucky CL, Julius D, Basbaum AI (2002) The 5-HT3 subtype of serotonin receptor contributes to nociceptive processing via a novel subset of myelinated and unmyelinated nociceptors. J Neurosci 22(3):1010–1019
Rahman W, Suzuki R, Rygh LJ, Dickenson AH (2004) Descending serotonergic facilitation mediated through rat spinal 5-HT3 receptors is unaltered following carrageenan inflammation. Neurosci Lett 361(1–3):229–231. https://doi.org/10.1016/j.neulet.2003.12.069
Wang W, Zhong X, Li Y, Guo R, Du S, Wen L, Ying Y, Yang T, Wei XH (2019) Rostral ventromedial medulla-mediated descending facilitation following P2X7 receptor activation is involved in the development of chronic post-operative pain. J Neurochem 149(6):760–780. https://doi.org/10.1111/jnc.14650
Greenwood-Van Meerveld B, Mohammadi E, Tyler K, Pietra C, Bee LA, Dickenson A (2014) Synergistic effect of 5-hydroxytryptamine 3 and neurokinin 1 receptor antagonism in rodent models of somatic and visceral pain. J Pharmacol Exp Ther 351(1):146–152. https://doi.org/10.1124/jpet.114.216028
Chen Y, Oatway MA, Weaver LC (2009) Blockade of the 5-HT3 receptor for days causes sustained relief from mechanical allodynia following spinal cord injury. J Neurosci Res 87(2):418–424. https://doi.org/10.1002/jnr.21860
Donovan-Rodriguez T, Urch CE, Dickenson AH (2006) Evidence of a role for descending serotonergic facilitation in a rat model of cancer-induced bone pain. Neurosci Lett 393(2–3):237–242. https://doi.org/10.1016/j.neulet.2005.09.073
Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Wurbel H (2020) The arrive guidelines 2.0: updated guidelines for reporting animal research. J Physiol 598(18):3793–3801. https://doi.org/10.1113/JP280389
Schiffman E, Ohrbach R, Truelove E, Look J, Anderson G, Goulet JP, List T, Svensson P, Gonzalez Y, Lobbezoo F, Michelotti A, Brooks SL, Ceusters W, Drangsholt M, Ettlin D, Gaul C, Goldberg LJ, Haythornthwaite JA, Hollender L, Jensen R, John MT, De Laat A, de Leeuw R, Maixner W, van der Meulen M, Murray GM, Nixdorf DR, Palla S, Petersson A, Pionchon P, Smith B, Visscher CM, Zakrzewska J, Dworkin SF International Rdc/Tmd Consortium Network IafDR, and Orofacial Pain Special Interest Group IAftSoP (2014) Diagnostic criteria for temporomandibular disorders (DC/TMD) for clinical and research applications: Recommendations of the international RDC/TMD consortium network* and orofacial pain special interest groupdagger. J Oral Facial Pain Headache 28(1):6–27. https://doi.org/10.11607/jop.1151
Lobbezoo F, Drangsholt M, Peck C, Sato H, Kopp S, Svensson P (2004) Topical review: new insights into the pathology and diagnosis of disorders of the temporomandibular joint. J Orofac Pain 18(3):181–191
Ambalavanar R, Moutanni A, Dessem D (2006) Inflammation of craniofacial muscle induces widespread mechanical allodynia. Neurosci Lett 399(3):249–254. https://doi.org/10.1016/j.neulet.2006.02.003
Hargreaves K, Dubner R, Brown F, Flores C, Joris J (1988) A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32(1):77–88. https://doi.org/10.1016/0304-3959(88)90026-7
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63. https://doi.org/10.1016/0165-0270(94)90144-9
Acknowledgements
The authors wish to thank Ms. Holly Ross (Department of Neural and Pain Sciences, University of Maryland School of Dentistry) for English reading and editing.
Funding
This work was supported by the National Natural Science Foundation of China (81971049, 81671097) and the Social Development Program of Shaanxi Province, China (2020SF-018).
Author information
Authors and Affiliations
Contributions
DYC designed the study, ZYT, XYQ, and SQW performed the experiments. GB analyzed the data and edited the paper. ZYT, JFL, and DYC analyzed the data and wrote the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
Experimental protocols were approved by the Institutional Animal Care and Use Committees of Xi’an Jiaotong University, China (No. 2016-006).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11064_2022_3540_MOESM1_ESM.eps
Supplementary material 1 SAHA did not affect the thermal withdrawal latency and mechanical withdrawal threshold in rats in the E2 + non-FS group and the E2 + saline + non-FS group (EPS 47.1 kb)
Rights and permissions
About this article
Cite this article
Tao, ZY., Qiu, XY., Wei, SQ. et al. SAHA Inhibits Somatic Hyperalgesia Induced by Stress Combined with Orofacial Inflammation Through Targeting Different Spinal 5-HT Receptor Subtypes. Neurochem Res 47, 1405–1418 (2022). https://doi.org/10.1007/s11064-022-03540-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-022-03540-0