Abstract
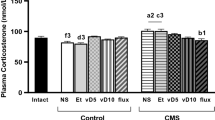
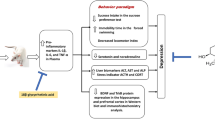
Pathophysiology of depression in elderlies is linked to aging-associated increase in indoleamine 2,3-dioxygenase (IDO) levels and activity and kynurenine (Kyn) metabolites. Moreover, these aging-induced changes may alter the brain’s responses to stress. Growing evidence suggested that young plasma can positively affect brain dysfunctions in old age. The present study aimed to investigate whether the antidepressant effects of young plasma administration in aged rats subjected to chronic unpredictable mild stress (CUMS) and underlying mechanisms, focusing on the prefrontal cortex (PFC). Young (3 months old) and aged (22 months old) male rats were divided into five groups; young control, aged control, aged rats subjected to CUMS (A + CUMS), aged rats subjected to CUMS and treated with young plasma (A + CUMS + YP), and aged rats subjected to CUMS and treated with old plasma (A + CUMS + OP). Plasma was injected (1 ml, intravenously) three times per week for four weeks. Young plasma significantly improved CUMS-induced depressive-like behaviors, evidenced by the increased sucrose consumption ratio in the sucrose preference test and the reduced immobility time in the forced swimming test. Furthermore, young plasma markedly reduced the levels of interferon-gamma (IFN-γ), IDO, Kyn, and Kyn to tryptophan (Kyn/Trp) ratio in PFC tissue. Expression levels of the serotonin transporter and growth-associated protein (GAP)-43 were also significantly increased after chronic administration of young plasma. These findings provide evidence for the antidepressant effect of young plasma in old age; however, whether it improves depressive behaviors or faster recovery from stress-induced deficits is required to be elucidated.







Similar content being viewed by others
References
Grady C (2012) The cognitive neuroscience of ageing. Nat Rev Neurosci 13:491–505. https://doi.org/10.1038/nrn3256
Mattson MP, Arumugam TV (2018) Hallmarks of brain aging: adaptive and pathological modification by metabolic states. Cell Metab 27:1176–1199. https://doi.org/10.1016/j.cmet.2018.05.011
Costa IC, Carvalho HN, Fernandes L (2013) Aging, circadian rhythms and depressive disorders: a review. Am J Neurodegener Dis 2:228–246
Birrer RB, Vemuri SP (2004) Depression in later life: a diagnostic and therapeutic challenge. Am Fam Physician 15:2375–2382
Gareri P, De Fazio P, De Sarro G (2002) Neuropharmacology of depression in aging and age-related diseases. Ageing Res Rev 1:113–134. https://doi.org/10.1016/S0047-6374(01)00370-0
Lavretsky H (2016) Intervention research in late-life depression: challenges and opportunities. Am J Geriatr Psychiatry 24:6–10. https://doi.org/10.1016/j.jagp.2015.10.011
Khundakar A, Morris C, Oakley A et al (2009) Morphometry analysis of neuronal and glial cell pathology in the dorsolateral prefrontal cortex in late-life depression. Br J Psychiatry 195:163–169. https://doi.org/10.1192/bjp.bp.108.052688
Esiri MM (2007) Ageing and the brain. J Pathol 211:181–187. https://doi.org/10.1002/path.2089
Oxenkrug GF (2010) Tryptophan-kynurenine metabolism as a common mediator of genetic and environmental impacts in major depressive disorder: the serotonin hypothesis revisited 40 years later. Isr J Psychiatry Relat Sci 47:56–63. https://doi.org/10.3410/f.13356987.14726252
Amori L, Guidetti P, Pellicciari R et al (2009) On the relationship between the two branches of the kynurenine pathway in the rat brain in vivo. J Neurochem 109:316–325. https://doi.org/10.1111/j.1471-4159.2009.05893.x
Schwarcz R, Pellicciari R (2002) Manipulation of brain kynurenines: glial targets, neuronal effects, and clinical opportunities. J Pharmacol Exp Ther 303:1–10. https://doi.org/10.1124/jpet.102.034439
Ladomersky E, Scholtens DM, Kocherginsky M et al (2019) The coincidence between increasing age, immunosuppression, and the incidence of patients with glioblastoma. Front Pharmacol 10:200. https://doi.org/10.3389/fphar.2019.00200
Schröcksnadel K, Wirleitner B, Winkler C, Fuchs D (2006) Monitoring tryptophan metabolism in chronic immune activation. Clin Chim Acta 364:82–90. https://doi.org/10.1016/j.cca.2005.06.013
Chung HY, Cesari M, Anton S et al (2009) Molecular inflammation: underpinnings of aging and age-related diseases. Ageing Res Rev 8:18–30. https://doi.org/10.1016/j.arr.2008.07.002
Oxenkrug G (2011) Interferon-gamma—inducible inflammation: contribution to aging and aging-associated psychiatric disorders. Aging Dis 2:474–486
Wu Y, Zhong X, Mai N et al (2018) Kynurenine pathway changes in late-life depression. J Affect Disord 235:76–81. https://doi.org/10.1016/j.jad.2018.04.007
Oxenkrug G (2013) Serotonin–kynurenine hypothesis of depression: historical overview and recent developments. Curr Drug Targets 14:514–521. https://doi.org/10.2174/1389450111314050002
Meier TB, Drevets WC, Wurfel BE et al (2016) Relationship between neurotoxic kynurenine metabolites and reductions in right medial prefrontal cortical thickness in major depressive disorder. Brain Behav Immun 53:39–48. https://doi.org/10.1016/j.bbi.2015.11.003
Meltzer CC, Smith G, Dekosky ST et al (1998) Serotonin in aging, late-life depression, and Alzheimer’s disease: the emerging role of functional imaging. Neuropsychopharmacology 18:407–430. https://doi.org/10.1016/S0893-133X(97)00194-2
Míguez JM, Aldegunde M, Paz-Valiñas L et al (1999) Selective changes in the contents of noradrenaline, dopamine and serotonin in rat brain areas during aging. J Neural Transm 106:1089–1098. https://doi.org/10.1007/s007020050225
Moll GH, Mehnert C, Wicker M et al (2000) Age-associated changes in the densities of presynaptic monoamine transporters in different regions of the rat brain from early juvenile life to late adulthood. Dev Brain Res 119:251–257. https://doi.org/10.1016/S0165-3806(99)00182-0
Tsukada H, Kakiuchi T, Nishiyama S et al (2001) Effects of aging on 5-HT1A receptors and their functional response to 5-HT1A agonist in the living brain: PET study with [carbonyl-11C]WAY-100635 in conscious monkeys. Synapse 42:242–251. https://doi.org/10.1002/syn.10011
Bouchard J, Villeda SA (2015) Aging and brain rejuvenation as systemic events. J Neurochem 132:5–19. https://doi.org/10.1111/jnc.12969
Shytikov D, Balva O, Debonneuil E et al (2014) Aged mice repeatedly injected with plasma from young mice: a survival study. Biores Open Access 3:226–232. https://doi.org/10.1089/biores.2014.0043
Villeda SA, Plambeck KE, Middeldorp J et al (2014) Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice. Nat Med 20:659–663. https://doi.org/10.1038/nm.3569
Katsimpardi L, Litterman NK, Schein PA et al (2014) Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science 344:630–634. https://doi.org/10.1126/science.1251141
Biagini G, Pich EM, Carani C et al (1993) Indole-pyruvic acid, a tryptophan ketoanalogue, antagonizes the endocrine but not the behavioral effects of repeated stress in a model of depression. Biol Psychiatry 33:712–719. https://doi.org/10.1016/0006-3223(93)90121-S
Forbes NF, Stewart CA, Matthews K, Reid IC (1996) Chronic mild stress and sucrose consumption: validity as a model of depression. Physiol Behav 60:1481–1484. https://doi.org/10.1016/S0031-9384(96)00305-8
Liu A, Guo E, Yang J et al (2018) Young plasma reverses age-dependent alterations in hepatic function through the restoration of autophagy. Aging Cell. https://doi.org/10.1111/acel.12708
Akimoto H, Oshima S, Sugiyama T et al (2019) Changes in brain metabolites related to stress resilience: metabolomic analysis of the hippocampus in a rat model of depression. Behav Brain Res 359:342–352. https://doi.org/10.1016/j.bbr.2018.11.017
Porsolt RD, Anton G, Blavet N, Jalfre M (1978) Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur J Pharmacol 47:379–391. https://doi.org/10.1016/0014-2999(78)90118-8
Spijker S (2011) Dissection of rodent brain regions. Neuromethods. Springer, Totowa, pp 13–26
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates: hard cover edition. Elsevier, Amsterdam
Vetreno RP, Yaxley R, Paniagua B et al (2017) Adult rat cortical thickness changes across age and following adolescent intermittent ethanol treatment. Addict Biol 22:712–723. https://doi.org/10.1111/adb.12364
Yamaguchi N, Nakajima N, Okada S, Yuri K (2016) Effects of aging on stress-related responses of serotonergic neurons in the dorsal raphe nucleus of male rats. Neurobiol Stress 3:43–51. https://doi.org/10.1016/j.ynstr.2016.01.002
Bachis A, Cruz MI, Nosheny RL, Mocchetti I (2008) Chronic unpredictable stress promotes neuronal apoptosis in the cerebral cortex. Neurosci Lett 442:104–108. https://doi.org/10.1016/j.neulet.2008.06.081
Herrera-Pérez JJ, Martínez-Mota L, Fernández-Guasti A (2008) Aging increases the susceptibility to develop anhedonia in male rats. Prog Neuro-Psychopharmacol Biol Psychiatry 32:1798–1803. https://doi.org/10.1016/j.pnpbp.2008.07.020
Leonard BE (2007) Inflammation, depression and dementia: are they connected? Neurochem Res 32:1749–1756. https://doi.org/10.1007/s11064-007-9385-y
Campos AC, Vaz GN, Saito VM, Teixeira AL (2014) Further evidence for the role of interferon-gamma on anxiety- and depressive-like behaviors: involvement of hippocampal neurogenesis and NGF production. Neurosci Lett 578:100–105. https://doi.org/10.1016/j.neulet.2014.06.039
Inserra A, Mastronardi CA, Rogers G et al (2019) Neuroimmunomodulation in major depressive disorder: focus on caspase 1, inducible nitric oxide synthase, and interferon-gamma. Mol Neurobiol 56:4288–4305. https://doi.org/10.1007/s12035-018-1359-3
Mehdipour M, Mehdipour T, Skinner CM et al (2020) Plasma dilution improves cognition and attenuates neuroinflammation in old mice. GeroScience. https://doi.org/10.1007/s11357-020-00297-8
Ghosh AK, O’Brien M, Mau T et al (2019) Adipose tissue senescence and inflammation in aging is reversed by the young Milieu. J Gerontol Ser A Biol Sci Med Sci 74:1709–1715. https://doi.org/10.1093/gerona/gly290
Liu D, Lun L, Huang Q et al (2018) Youthful systemic milieu alleviates renal ischemia-reperfusion injury in elderly mice. Kidney Int 94:268–279. https://doi.org/10.1016/j.kint.2018.03.019
Mohr DC, Goodkin DE, Islar J et al (2001) Treatment of depression is associated with suppression of nonspecific and antigen-specific TH1 responses in multiple sclerosis. Arch Neurol 58:1081–1086. https://doi.org/10.1001/archneur.58.7.1081
Godbout JP, Moreau M, Lestage J et al (2008) Aging exacerbates depressive-like behavior in mice in response to activation of the peripheral innate immune system. Neuropsychopharmacology 33:2341–2351. https://doi.org/10.1038/sj.npp.1301649
Oxenkrug GF (2011) Interferon-gamma-inducible kynurenines/pteridines inflammation cascade: implications for aging and aging-associated psychiatric and medical disorders. J Neural Transm 118:75–85. https://doi.org/10.1007/s00702-010-0475-7
Stone TW, Darlington LG (2013) The kynurenine pathway as a therapeutic target in cognitive and neurodegenerative disorders. Br J Pharmacol 169:1211–1227. https://doi.org/10.1111/bph.12230
Hughes MM, Carballedo A, McLoughlin DM et al (2012) Tryptophan depletion in depressed patients occurs independent of kynurenine pathway activation. Brain Behav Immun 26:979–987. https://doi.org/10.1016/j.bbi.2012.05.010
Yoshimoto K, Kato B, Ueda S et al (2001) Dopamine and serotonin uptake inhibitors on the release of dopamine and serotonin in the nucleus accumbens of young and aged rats. Mech Ageing Dev 122:1707–1721. https://doi.org/10.1016/S0047-6374(01)00292-5
Herrera-Pérez JJ, Martínez-Mota L, Fernández-Guasti A (2010) Aging impairs the antidepressant-like response to citalopram in male rats. Eur J Pharmacol 633:39–43. https://doi.org/10.1016/j.ejphar.2010.01.022
Couch Y, Anthony DC, Dolgov O et al (2013) Microglial activation, increased TNF and SERT expression in the prefrontal cortex define stress-altered behaviour in mice susceptible to anhedonia. Brain Behav Immun 29:136–146. https://doi.org/10.1016/j.bbi.2012.12.017
Slotkin TA, Miller DB, Fumagalli F et al (1999) Modeling geriatric depression in animals: Biochemical and behavioral effects of olfactory bulbectomy in young versus aged rats. J Pharmacol Exp Ther 289:334–345
Slotkin TA, Cousins MM, Tate CA, Seidler FJ (2005) Serotonergic cell signaling in an animal model of aging and depression: olfactory bulbectomy elicits different adaptations in brain regions of young adult vs aging. Neuropsychopharmacology 30:52–57. https://doi.org/10.1038/sj.npp.1300569
Denny J (2006) Molecular mechanisms, biological actions, and neuropharmacology of the growth-associated protein GAP-43. Curr Neuropharmacol 4:293–304. https://doi.org/10.2174/157015906778520782
Benowitz LI, Routtenberg A (1997) GAP-43: an intrinsic determinant of neuronal development and plasticity. Trends Neurosci 20:84–91. https://doi.org/10.1016/S0166-2236(96)10072-2
Hrdina P, Faludi G, Li Q et al (1998) Growth-associated protein (GAP-43), its mRNA, and protein kinase C (PKC) isoenzymes in brain regions of depressed suicides. Mol Psychiatry 3:411–418. https://doi.org/10.1038/sj.mp.4000435
Donovan SL, Mamounas LA, Andrews AM et al (2002) GAP-43 is critical for normal development of the serotonergic innervation in forebrain. J Neurosci 22:3543–3552. https://doi.org/10.1523/jneurosci.22-09-03543.2002
Coleman PD, Rogers KE, Flood DG (1990) Neuronal plasticity in normal aging and deficient plasticity in Alzheimer’s disease: a proposed intercellular signal cascade. Progress in brain research. Elsevier, Amsterdam, pp 75–87
Mohan A, Thalamuthu A, Mather KA et al (2018) Differential expression of synaptic and interneuron genes in the aging human prefrontal cortex. Neurobiol Aging 70:194–202. https://doi.org/10.1016/j.neurobiolaging.2018.06.011
Storsve AB, Fjell AM, Tamnes CK et al (2014) Differential longitudinal changes in cortical thickness, surface area and volume across the adult life span: regions of accelerating and decelerating change. J Neurosci 34:8488–8498. https://doi.org/10.1523/JNEUROSCI.0391-14.2014
Kassem MS, Lagopoulos J, Stait-Gardner T et al (2013) Stress-induced grey matter loss determined by MRI is primarily due to loss of dendrites and their synapses. Mol Neurobiol 47:645–661. https://doi.org/10.1007/s12035-012-8365-7
Rebo J, Mehdipour M, Gathwala R et al (2016) A single heterochronic blood exchange reveals rapid inhibition of multiple tissues by old blood. Nat Commun 7:13363. https://doi.org/10.1038/ncomms13363
Castellano JM, Kirby ED, Wyss-Coray T (2015) Blood-borne revitalization of the aged brain. JAMA Neurol 72:1191–1194. https://doi.org/10.1001/jamaneurol.2015.1616
Wang W, Wang L, Ruan L et al (2018) Extracellular vesicles extracted from young donor serum attenuate inflammaging via partially rejuvenating aged T-cell immunotolerance. FASEB J 32:5899–5912. https://doi.org/10.1096/fj.201800059R
Schafer MJ, LeBrasseur NK (2019) The influence of GDF11 on brain fate and function. Geroscience 41:1–11. https://doi.org/10.1007/s11357-019-00054-6
Castellano JM, Mosher KI, Abbey RJ et al (2017) Human umbilical cord plasma proteins revitalize hippocampal function in aged mice. Nature 544:488–492. https://doi.org/10.1038/nature22067
Baker LD, Barsness SM, Borson S et al (2012) Effects of growth hormone-releasing hormone on cognitive function in adults with mild cognitive impairment and healthy older adults: results of a controlled trial. Arch Neurol 69:1420–1429. https://doi.org/10.1001/archneurol.2012.1970
Gan K, Südhof T (2019) Specific factors in blood from young but not old mice directly promote synapse formation and NMDA-receptor recruitment. Proc Natl Acad Sci 116:201902672. https://doi.org/10.1073/pnas.1902672116
Acknowledgements
This study was supported by a Grant from the Aging Research Institute of Tabriz University of Medical Sciences. Our data were derived from the thesis of Arshad Ghaffari-Nasab for a Ph.D. degree in physiology (Thesis No. 60281).
Funding
This study was supported by a grant from the Aging Research Institute of Tabriz University of Medical Sciences [Grant Number 60281].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical Approval
All procedures and protocols were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the National Institute of Health (NIH; Publication No. 85-23, revised 1985) and approved by the regional ethics committee of Tabriz University of Medical Sciences (No: IR.TBZMED.VCR.REC.1397.197).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghaffari-Nasab, A., Badalzadeh, R., Mohaddes, G. et al. Young Plasma Induces Antidepressant-Like Effects in Aged Rats Subjected to Chronic Mild Stress by Suppressing Indoleamine 2,3-Dioxygenase Enzyme and Kynurenine Pathway in the Prefrontal Cortex. Neurochem Res 47, 358–371 (2022). https://doi.org/10.1007/s11064-021-03440-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03440-9