Abstract
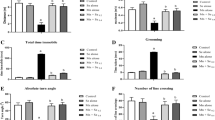
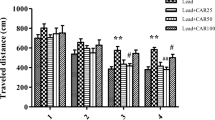
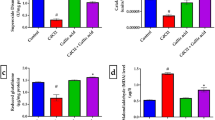
Clinical and experimental evidences reveal that excess exposure to manganese is neurotoxic and leads to cellular damage. However, the mechanism underlying manganese neurotoxicity remains poorly understood but oxidative stress has been implicated to be one of the key pathophysiological features related to it. The present study investigates the effects associated with manganese induced toxicity in rats and further to combat these alterations with a well-known antioxidant N-acetylcysteine which is being used in mitigating the damage by its radical scavenging activity. The study was designed to note the sequential changes along with the motor and memory dysfunction associated with biochemical and histo-pathological alterations following exposure and treatment for 2 weeks. The results so obtained showed decrease in the body weights, behavioral deficits with increased stress markers and also neuronal degeneration in histo-pathological examination after manganese intoxication in rats. To overcome the neurotoxic effects of manganese, N-acetylcysteine was used in the current study due to its pleiotropic potential in several pathological ailments. Taken together, N-acetylcysteine helped in ameliorating manganese induced neurotoxic effects by diminishing the behavioral deficits, normalizing acetylcholinesterase activity, and augmentation of redox status.











Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- 5-HT:
-
Serotonin
- AChE:
-
Acetylcholinesterase
- ANOVA:
-
Analysis of variance
- BBB:
-
Blood brain barrier
- BSA:
-
Bovine serum albumin
- DTNB:
-
5,5’-Dithiobis (2-nitrobenzoic acid)
- DTT:
-
Dithiothreitol
- DA:
-
Dopamine
- SOD:
-
Superoxide dismutase
- GPx:
-
Glutathione peroxidase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- TG:
-
Total glutathione
- GST:
-
Glutathione transferase
- H&E:
-
Hematoxylin and eosin
- H2O2 :
-
Hydrogen peroxide
- HPLC:
-
High performance liquid chromatography
- i.p.:
-
Intraperitoneally
- LPO:
-
Lipid peroxidation
- LSD:
-
Least significance difference
- MnCl2·4H2O:
-
Manganese chloride tetrahydrate
- MnSOD:
-
Manganese superoxide dismutase
- NBT:
-
Nitroblue tetrazolium
- NAC:
-
N-acetylcysteine
- PMF:
-
Post mitochondrial fraction
- ROS:
-
Reactive oxygen species
- SD:
-
Standard deviation
- SPSS:
-
Statistical software package
- TBA:
-
Thiobarbituric acid
- TCA:
-
Trichloroacetic acid
- SNpc:
-
Substantia nigra pars compacta
- PD:
-
Parkinson’s disease
- BBB:
-
Blood brain barrier
- OH° :
-
Hydroxyl radical
- O2°− :
-
Superoxide radical
- H2O:
-
Water
- MDA:
-
Malondialdehyde
- O.D.:
-
Optical density
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- CDNB:
-
2,4-Dinitrochlorobenzene
References
Szpetnar M, Luchowska-Kocot D, Boguszewska-Czubara A, Kurzepa J (2016) The influence of manganese and glutamine intake on antioxidants and neurotransmitter amino acids levels in rats’ brain. Neurochem Res 41:2129–2139
Horning KJ, Caito SW, Tipps KG, Bowman AB, Aschner M (2015) Manganese is essential for neuronal health. Annu Rev Nutr 35:71–108
Kwakye GF, Paoliello MM, Mukhopadhyay S, Bowman AB, Aschner M (2015) Manganese-induced parkinsonism and Parkinson’s disease: shared and distinguishable features. Int J Environ Res Public Health 12(7):7519–7540
Sarkar S, Malovic E, Harischandra DS, Ngwa HA, Ghosh A, Hogan C, Rokad D, Zenitsky G, Jin H, Anantharam V, Kanthasamy AG, Kanthasamy A (2018) Manganese exposure induces neuroinflammation by impairing mitochondrial dynamics in astrocytes. Neurotoxicology 64:204–218
Harischandra DS, Ghaisas S, Zenitsky G, Jin H, Kanthasamy A, Anantharam V, Kanthasamy AG (2019) Manganese-induced neurotoxicity: new insights into the triad of protein misfolding, mitochondrial impairment, and neuroinflammation. Front Neurosci 13:654
Flynn MR, Susi P (2009) Neurological risks associated with manganese exposure from welding operations—a literature review. Int J Hyg Environ Health 212:459–469
Long Z, Jiang YM, Li XR, Fadel W, Xu J, Yeh CL, Long LL, Luo HL, Harezlak J (2014) Vulnerability of welders to manganese exposure—a neuroimaging study. Neurotoxicology 45:285–292
Guilarte TR, Gonzales KK (2015) Manganese-induced parkinsonism is not idiopathic Parkinson’s disease: environmental and genetic evidence. Toxicol Sci 146:204–212
de Water E, Proal E, Wang V, Martínez Medina S, Schnaas L, Téllez-Rojo MM, Wright RO, Tang CY, Horton MK (2018) Prenatal manganese exposure and intrinsic functional connectivity of emotional brain areas in children. Neurotoxicology 64:85–93
Kornblith ES, Casey SL, Lobdell DT, Colledge MA, Bowler RM (2018) Environmental exposure to manganese in air: tremor, motor and cognitive symptom profiles. Neurotoxicology 64:152–158
Bouabid S, Delaville C, Deurwaerde PD, Lakhdar-Ghazal N, Benazzouz A (2014) Manganese-induced atypical parkinsonism is associated with altered basal ganglia activity and changes in tissue levels of monoamines in the rat. PLoS ONE 9:1–11
Gu F, Chauhan V, Chauhan A (2015) Glutathione redox imbalance in brain disorders. Curr Opin Clin Nutr Metab Care 18:89–95
Mahmoud SM, Moneim AE, Qayed MM, El-Yamany NA (2019) Potential role of N-acetylcysteine on chlorpyrifos-induced neurotoxicity in rats. Environ Sci Pollut Res 26:20731–20741
Chouchani ET, Kazak L, Jedrychowski MP, Lu GZ, Erickson BK, Szpyt J, Pierce KA, Laznik-Bogoslavski D, Vetrivelan R, Clish CB, Robinson AJ, Gygi SP, Bruce M (2016) Spiegelman mitochondrial ROS regulate thermogenic energy expenditure and sulfenylation of UCP1. Nature 532:112–116
Ezerina D, Takano Y, Hanaoka H, Urano Y, Dick TP (2018) N-acetyl cysteine functions as a fast-acting antioxidant by triggering intracellular H2S and sulfane sulfur production. Cell Chem Biol 25:447–459
Abdel-Daim MM, Dessouki AA, Abdel-Rahman HG, Eltaysh R, Alkahtani S (2019) Hepatorenal protective effects of taurine and N-acetylcysteine against fipronil-induced injuries: the antioxidant status and apoptotic markers expression in rats. Sci Total Environ 650:2063–2073
Samuni Y, Goldstein S, Dean OM, Berk M (2013) The chemistry and biological activities of N-acetylcysteine. Biochem Biophys Acta 1830:4117–4129
Šalamon Š, Kramar B, Marolt TP, Poljšak B, Milisav I (2019) Review medical and dietary uses of N-acetylcysteine. Antioxidants 8:111
Zhang Q, Ju Y, Ma Y, Wang T (2018) N-acetylcysteine improves oxidative stress and inflammatory response in patients with community acquired pneumonia: a randomized controlled trial. Medicine 97(45):e13087
Dean O, Giorlando F, Berk M (2011) N-acetylcysteine in psychiatry: current therapeutic evidence and potential mechanisms of action. J Psychiatry Neurosci 36:78–86
Dringen R, Hirrlinger J (2003) Glutathione pathways in the brain. Biol Chem 384:505–516
Chen G, Shi J, Hu Z, Hang C (2008) Inhibitory effect on cerebral inflammatory response following traumatic brain injury in rats: a potential neuroprotective mechanism of N-acetylcysteine. Mediat Inflamm. https://doi.org/10.1155/2008/716458
Nascimento MM, Suliman ME, Silva M, Chinaglia T, Marchioro J, Hayashi SY, Riella MC, Lindholm B, Anderstam B (2010) Effect of oral N-acetylcysteine treatment on plasma inflammatory and oxidative stress markers in peritoneal dialysis patients: a Placebo-Controlled Study. Perit Dial Int 30:336–342
Garcia-Bueno B, Bioque M, Mac-Dowell KS, Barcones MF, Martinez- Cengotitabengoa M, Pina-Camacho L, Rodriguez-Jimenez R, Saiz PA, Castro C, Lafuente A, Santabarbara J, Gonzalez-Pinto A, Parellada M, Rubio G, Garcia-Portilla MP, Mico JA, Bernardo M, Leza JC (2014) Pro-/anti-inflammatory dysregulation in patients with first episode of psychosis: toward an integrative inflammatory hypothesis of schizophrenia. Schizophr Bull 40:376–387
Breier A, Liffick E, Hummer TA, Vohs JL, Yanga Z, Mehdiyoun NF, Visco AC, Metzler E, Zhang Y, Francis MM (2018) Effects of 12-month, double-blind N-acetyl cysteine on symptoms, cognition and brain morphology in early phase schizophrenia spectrum disorders. Schizophr Res 199:395–402
Santos D, Milatovic D, Andradea V, Batoreua MC, Aschner M, dos Santos APM (2012) The inhibitory effect of manganese on acetylcholinesterase activity enhances oxidative stress and neuroinflammation in the rat brain. Toxicology 292:90–98
Bowler RM, Gocheva V, Harris M, Ngo L, Abdelouahab N, Wilkinson J, Doty RL, Park R, Roels HA (2011) Prospective study on neurotoxic effects in manganese-exposed bridge construction welders. Neurotoxicology 32:596–605
Roels H, Meiers G, Delos M, Ortega I, Lauwerys R, Buchet JP, Lison D (1997) Influence of the route of administration and the chemical form (MnCl2, MnO2) on the absorption and cerebral distribution of manganese in rats. Arch Toxicol 71:223–230
Bowman AB, Aschner M (2014) Considerations on manganese (Mn) treatments for in vitro studies. Neurotoxicology 41:141–142
Lowry OH, Rosenbrough NJ, Farr AL, Ranell RJ (1951) Protein measurements with the Follin’s phenol reagent. J Biol Chem 193:265–275
Bishnoi M, Chopra K, Kulkarni SK (2006) Involvement of adenosinergic receptor system in an animal model of tardive dyskinesia and associated behavioral, biochemical and neurochemical changes. Eur J Pharmacol 552:55–56
Kulkarni SK (1999) Handbook of experimental pharmacology, 3rd edn. Vallabh Prakashan, New Delhi
Costall B, Naylor RJ (1974) On catalepsy and catatonia and the predictability of the catalepsy test for neuroleptic activity. Psychopharmacologia 34:233–241
Sharma AC, Kulkarni SK (1992) Evaluation of learning and memory mechanisms employing elevated plus-maze in rats and mice. Prog Neuropsychopharmacol Biol Psychiatry 16:117–125
Miyamoto M, Kameyama M, Nagaoka A, Nagawa Y (1985) Lesioning of the rat basal forebrain leads to memory impairments in passive and active avoidance tasks. Brain Res 328:97–104
Church WH (2005) Column chromatography analysis of brain tissue: an advanced laboratory exercise for neuroscience majors. J Undergrad Neurosci Educ 3:36–41
Ellman GL, Lourtney KD, Andres VJ (1961) A new and rapid calorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Best TM, Feiebig R, Corr DT, Brickson S, Ji L (1999) Free radical activity, antioxidant enzyme, and glutathione changes with muscle stretch injury in rabbits. J Appl Physiol 87:74–82
Reddy DS, Kulkarni SK (1998) Possible role of nitric oxide in the nootropic and antiamnesic effects of neurosteroids on aging- and dizocilpineinduced learning impairment. Brain Res 799:215–229
Wills ED (1966) Mechanism of lipid peroxide formation in animal tissues. Biochem J 99:667–676
Ellman GL (1959) Tissue sulfhydral groups. Arch Biochem Biophys 82:70–77
Zahler WL, Cleland WW (1968) A specific and sensitive assay for disulfides. J Biol Chem 243:12901–12912
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–121
Carlberg I, Mannervik B (1985) Glutathione reducatse. Methods Enzymol 113:489–490
Habig WH, Pabst MJ, Jacoby WB (1974) Glutathion-s-transferase. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Kono Y (1978) Generation of superoxide radicals during auto oxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Luck H (1963) Catalase. In: Bermeyer HW (ed) Methods of enzymatic analysis. Academic press, Cambridge, pp 885–894
Pearse AGE (1968) Histochemistry theoretical and applied. Churchill livingstone, London 1:660. Placebo-controlled study. Perit Dial Int 30:336–342
Humason GL (1962) Basic procedures-animal tissue technique. Animal tissue techniques. W. H. Freeman and Company, San Francisco, pp 130–132
Kamboj SS, Vaishta RK, Sandhir R (2010) N-acetylcysteine inhibits hyperglycemia-induced oxidative stress and apoptosis markers in diabetic neuropathy. J Neurochem 112:77–91
Dhanda S, Kaur S, Sandhir R (2013) Preventive effect of N-acetyl-l-cysteine on oxidative stress and cognitive impairment in hepatic encephalopathy following bile ductligation. Free Radic Biol Med 56:204–215
Muller N, Weidinger E, Leitner B, Schwarz MJ (2015) The role of inflammation in schizophrenia. Front Neurosci 9:372
Li SJ, Qin WX, Peng DJ, Yuan ZX, He SN, Luo YN, Aschner M, Jiang YM, Liang DY, Xie BY, Xu F (2018) Sodium P-aminosalicylic acid inhibits sub-chronic manganese-induced neuroinflammation in rats by modulating MAPK and COX-2. Neurotoxicology 64:219–229
Zhao F, Cai T, Liu M, Zheng G, Luo W, Chen J (2009) Manganese induces dopaminergic neurodegeneration via microglial activation in a rat model of manganism. Toxicol Sci 107:156–164
Milatovic D, Zaja-Milatovic S, Gupta RC, Yu Y, Aschner M (2009) Oxidative damage and neurodegeneration in manganese induced neurotoxicity. Toxicol Appl Pharmacol 240:219–225
Zhang P, Lokuta KM, Turner DE, Liu B (2010) Synergistic dopaminergic neurotoxicity of manganese and lipopolysaccharide: differential involvement of microglia and astroglia. J Neurochem 112:434–443
Filipov NM, Seegal RF, Lawrence DA (2005) Manganese potentiates in vitro production of proinflammatory cytokines and nitric oxide by microglia through a nuclear factor kappa B-dependent mechanism. Toxicol Sci 84:139–148
Adedara I, Abolaji AO, Awogbindin IO, Farombi EO (2017) Suppression of the brain-pituitary-testicular axis function following acute arsenic and manganese co-exposure and withdrawal in rats. J Trace Elem Med Biol 39:21–29
Martinez-Finley EJ, Gavin CE, Aschner M, Gunter TE (2013) Manganese neurotoxicity and the role of reactive oxygen species. Free Radic Biol Med 62:65–75
Aschner M, Guilarte TR, Schneider JS, Zheng W (2007) Manganese: recent advances in understanding its transport and neurotoxicity. Toxicol Appl Pharmacol 221:131–147
O’Neal SL, Lee JW, Zheng W, Cannon JR (2014) Subacute manganese exposure in rats is a neurochemical model of early manganese toxicity. Neurotoxicology 44:303–313
Yang X, Yang H, Wu F, Qi Z, Li J, Xu B, Liu W, Xu Z, Deng Y (2018) Mn inhibits GSH synthesis via downregulation of neuronal EAAC1 and astrocytic xCT to cause oxidative damage in the striatum of mice. Oxid Med Cell Longev. https://doi.org/10.1155/2018/4235695
Milatovic D, Zaja-Milatovic S, Gupta RC, Yu Y, Aschner M (2011) Protective effects of antioxidants and anti-inflammatory agents against manganese-induced oxidative damage and neuronal injury. Toxicol Appl Pharmacol 256:219–226
dos Santos APM, Santos D, Au C, Milatovic D, Aschner M, Batoréu MCC (2008) Antioxidants prevent the cytotoxicity of manganese in RBE4 cells. Brain Res 1236:200–205
Zhang S, Fu J, Zhou Z (2004) In vitro effect of manganese chloride exposure on reactive oxygen speciesm generation and respiratory chain complexes activities of mitochondria isolated from rat brain. Toxicol In Vitro 18:71–77
Ulusu NN, Sahilli M, Avci A, Canbolat O, Ozansoy G, Ari N, Bali M, Stefek M, Stolc S, Gajdosik A, Karasu C (2003) Pentose phosphate pathway, glutathione-dependent enzymes and antioxidant defense during oxidative stress in diabetic rodent brain and peripheral organs: effects of stobadine and vitamin E. Neurochem Res 28:815–823
Chtourou Y, Fetoui H, Sefi M, Trabelsi K, Barkallah M, Boudawara T, Kallel H, Zeghal N (2010) Silymarin, a natural antioxidant, protects cerebral cortex against manganese-induced neurotoxicity in adult rats. Biometals 23:985–996
Adedara IA, Ego VC, Subair TI, Oyediran O, Farombi EO (2017) Quercetin improves neurobehavioral performance through restoration of brain antioxidant status and acetylcholinesterase activity in manganese-treated rats. Neurochem Res 42:1219–1229
Dickinson DA, Forman HJ (2002) Glutathione in defense and signaling:lessons from a small thiol. Ann N Y Acad Sci 973:488–504
Rushworth GF, Megson IL (2014) Existing and potential therapeutic uses for N-acetylcysteine: the need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol Ther 141:150–159
Kamboj SS, Chopra K, Sandhir R (2008) Neuroprotective effect of N-acetylcysteine in the development of diabetic encephalopathy in streptozotocin-induced diabetes. Metab Brain Dis 23:427–443
Chtourou Y, Fetoui H, Garoui EM, Boudawara T, Zeghal N (2012) Improvement of cerebellum redox states and cholinergic functions contribute to the beneficial effects of silymarin against manganese-induced neurotoxicity. Neurochem Res 37:468–479
Latronico T, Brana MT, Merra E, Fasano A, Di Bari G, Casalino E, Liuzzi GM (2013) Impact of manganese neurotoxicity on MMP-9 production and superoxide dismutase activity in rat primary astrocytes. Effect of resveratrol and therapeutical implications for the treatment of CNS diseases. Toxicol Sci 135:218–228
Hashemi G, Mirjalili M, Basiri Z, Tahamoli-Roudsari A, Kheiripour N, Shahdoust M (2018) Evaluating the effects of oral N-acetyl cysteine on inflammatory and oxidative stress biomarkers in rheumatoid arthritis. Curr Rheumatol Rev 15(3):246–253
Aparicio-Trejo OE, Reyes-Fermín LM, Briones-Herrera A, Tapia E, León-Contreras JC, Hernández-Pando R, Sánchez-Lozada LG, Pedraza-Chaverri J (2019) Protective effects of N-acetyl-cysteine in mitochondria bioenergetics, oxidative stress, dynamics and S-glutathionylation alterations in acute kidney damage induced by folic acid. Free Radic Biol Med 130:379–396
Bouchard M, Mergler D, Baldwin ME, Panisset M (2008) Manganese cumulative exposure and symptoms: a follow up study of alloy workers. Neurotoxicology 29:577–583
Delaville C, Chetrit J, Abdallah K, Morin S, Cardoit L, De Deurwaerdère P, Benazzouz A (2012) Emerging dysfunctions consequent to combined monoaminergic depletions in parkinsonism. Neurobiol Dis 45:763–773
Costa M, Bernardi J, Fiuza T, Costa L, Brand R, Pereira ME (2016) N-acetylcysteine protects memory decline induced by streptozotocin in mice. Chem Biol Interact 253:10–17
Rommelfanger KS, Edwards GL, Freeman KG, Liles LC, Miller GW, Weinshenker D (2007) Norepinephrine loss produces more profound motor deficits than MPTP treatment in mice. Proc Natl Acad Sci 104:13804–13809
Santos D, Milatovic D, Andrade V, Batoreu MC, Aschner M, dos Santos APM (2012) The inhibitory effect of manganese on acetylcholinesterase activity enhances oxidative stress and neuroinflammation in the rat brain. Toxicology 292(2–3):90–98
Sanchez-Betancourt J, Anaya-Martinez V, Gutierrez-Valdez AL, Ordonez-Librado JL, Montiel-Flores E, Espinosa-Villanueva J, Reynoso-Erazo L, Avila-Costa MR (2012) Manganese mixture inhalation is a reliable Parkinson disease model in rats. Neurotoxicology 33:1346–1355
Cordova FM, Aguiar AS Jr, Peres TV, Lopes MW, Gonçalves FM, Pedro DZ, Lopes SC, Pilati C, Prediger RD, Farina M, Erikson KM, Aschner M, Leal RB (2013) Manganese-exposed developing rats display motor deficits and striatal oxidative stress that are reversed by Trolox. Arch Toxicol 87:1231–1244
Olanow CW (2004) Manganese-induced parkinsonism and Parkinson’s disease. Ann N Y Acad Sci 1012:209–223
Abe H, Ohishi T, Nakane F, Shiraki A, Tanaka T, Yoshida T, Shibutani M (2015) Exposure to MnCl2·4H2O during development induces activation of microglial and perivascular macrophage populations in the hippocampal dentate gyrus of rats. J Appl Toxicol 35:529–535
Wang T, Li X, Yang D, Zhang H, Zhao P, Fu J, Yao B, Zhou Z (2015) ER stress and ER stress-mediated apoptosis are involved in manganese-induced neurotoxicity in the rat striatum in vivo. Neurotoxicology 48:109–119
Funding
This work was supported by UGC SAP, DST-FIST, PURSE New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical Approval
All the protocols performed were approved by Animal Ethical Committee (IAEC) of Panjab University, Chandigarh, India with approval no. PU|45|99|CPCSEA|IAEC|2018|91.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chopra, D., Sharma, S., Sharma, N. et al. N-Acetylcysteine Ameliorates Neurotoxic Effects of Manganese Intoxication in Rats: A Biochemical and Behavioral Study. Neurochem Res 46, 1953–1969 (2021). https://doi.org/10.1007/s11064-021-03312-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03312-2