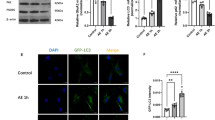
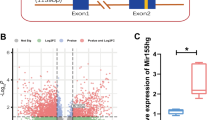
Abstract
Propofol is one of the most common intravenous anesthetics which may cause neuronal cell death in young mice. HOX transcript antisense RNA (HOTAIR) was abnormally expressed in neurodegenerative diseases. However, the effect of HOTAIR on propofol-induced pyroptosis of neurons and related mechanisms are still unknown. In this study, propofol treatment significantly reduced neuronal the viability of neurons, and promoted the expression of inflammation-related factors. Propofol treatment also promoted neuron death and neuronal pyroptosis. All the above effects might be related to the propofol-induced overexpression of HOTAIR. Interestingly, knockdown of HOTAIR by shRNA (sh-HOTAIR) significantly inhibited neuronal pyroptosis, but increased neuronal viability. Further analysis showed that HOTAIR and Nod-like receptor protein1 (NLRP1) were the targets of miR-455-3p, respectively. Notably, propofol treatment decreased the level of miR-455-3p, while increased the level of NLRP1. In addition, sh-HOTAIR increased the level of miR-455-3p, which further inhibited the expression of NLRP1 and the activation of NLRP1 inflammasome, thereby inhibiting neuronal pyroptosis. More importantly, NLRP1 overexpression decreased neuronal viability, and reactivated NLRP1 inflammasome, thus reversing the inhibitory effect of sh-HOTAIR on pyroptosis. Our findings indicated that HOTAIR inhibited propofol-induced pyroptosis of neurons by regulating miR-455-3p/NLRP1 axis, indicating that HOTAIR may be a potential therapeutic target for propofol-induced neurotoxicity.






Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Yan Y, Qiao S, Kikuchi C, Zaja I (2017) Propofol induces apoptosis of neurons but not astrocytes, oligodendrocytes, or neural stem cells in the neonatal mouse hippocampus. Brain Sci 7(10):130. https://doi.org/10.3390/brainsci7100130
Kamenik M, Kos D, Möller Petrun A, Green DW, Zorko N, Mekiš D (2018) Haemodynamic stability during anaesthesia induction with propofol–impact of phenylephrine. A double-blind, randomised clinical trial. Signa Vitae 14(1):20–26
Zheng SQ, An LX, Cheng X, Wang YJ (2013) Sevoflurane causes neuronal apoptosis and adaptability changes of neonatal rats. Acta Anaesthesiol Scand 57(9):1167–1174. https://doi.org/10.1111/aas.12163
Creeley C, Dikranian K, Dissen G, Martin L, Olney J, Brambrink A (2013) Propofol-induced apoptosis of neurones and oligodendrocytes in fetal and neonatal rhesus macaque brain. Br J Anaesth 110(Suppl 1):i29–i38. https://doi.org/10.1093/bja/aet173
Bosnjak ZJ, Logan S, Liu Y, Bai X (2016) Recent insights into molecular mechanisms of propofol-induced developmental neurotoxicity: implications for the protective strategies. Anesth Analg 123(5):1286–1296. https://doi.org/10.1213/ane.0000000000001544
Miao EA, Leaf IA, Treuting PM, Mao DP, Dors M, Sarkar A, Warren SE, Wewers MD, Aderem A (2010) Caspase-1-induced pyroptosis is an innate immune effector mechanism against intracellular bacteria. Nat Immunol 11(12):1136–1142. https://doi.org/10.1038/ni.1960
Kummer JA, Broekhuizen R, Everett H, Agostini L, Kuijk L, Martinon F, van Bruggen R, Tschopp J (2007) Inflammasome components NALP 1 and 3 show distinct but separate expression profiles in human tissues suggesting a site-specific role in the inflammatory response. J Histochem Cytochem 55(5):443–452. https://doi.org/10.1369/jhc.6A7101.2006
Masters SL, Gerlic M, Metcalf D, Preston S, Pellegrini M, O’Donnell JA, McArthur K, Baldwin TM, Chevrier S, Nowell CJ, Cengia LH, Henley KJ, Collinge JE, Kastner DL, Feigenbaum L, Hilton DJ, Alexander WS, Kile BT, Croker BA (2012) NLRP1 inflammasome activation induces pyroptosis of hematopoietic progenitor cells. Immunity 37(6):1009–1023. https://doi.org/10.1016/j.immuni.2012.08.027
Wang S, Zhang X, Guo Y, Rong H, Liu T (2017) The long noncoding RNA HOTAIR promotes Parkinson’s disease by upregulating LRRK2 expression. Oncotarget 8(15):24449–24456. https://doi.org/10.18632/oncotarget.15511
Lin Q, Hou S, Dai Y, Jiang N, Lin Y (2019) LncRNA HOTAIR targets miR-126-5p to promote the progression of Parkinson’s disease through RAB3IP. Biol Chem 400(9):1217–1228. https://doi.org/10.1515/hsz-2018-0431
Obaid M, Udden SMN, Deb P, Shihabeddin N, Zaki MH, Mandal SS (2018) LncRNA HOTAIR regulates lipopolysaccharide-induced cytokine expression and inflammatory response in macrophages. Sci Rep 8(1):15670. https://doi.org/10.1038/s41598-018-33722-2
Wang JY, Feng Y, Fu YH, Liu GL (2018) Effect of sevoflurane anesthesia on brain is mediated by lncRNA HOTAIR. J Mol Neurosci 64(3):346–351. https://doi.org/10.1007/s12031-018-1029-y
Kumar S, Reddy PH (2019) A new discovery of microRNA-455-3p in Alzheimer’s disease. J Alzheimers Dis 72(s1):S117–s130. https://doi.org/10.3233/jad-190583
Guo S, Zhen Y, Zhu Z, Zhou G, Zheng X (2019) Cinnamic acid rescues behavioral deficits in a mouse model of traumatic brain injury by targeting miR-455-3p/HDAC2. Life Sci 235:116819. https://doi.org/10.1016/j.lfs.2019.116819
Kumar S, Reddy AP, Yin X, Reddy PH (2019) Novel MicroRNA-455-3p and its protective effects against abnormal APP processing and amyloid beta toxicity in Alzheimer’s disease. Biochim Biophys Acta Mol Basis Dis 1865(9):2428–2440. https://doi.org/10.1016/j.bbadis.2019.06.006
Huang JY, Wang K, Vermehren-Schmaedick A, Adelman JP, Cohen MS (2016) PARP6 is a regulator of hippocampal dendritic morphogenesis. Sci Rep 6:18512. https://doi.org/10.1038/srep18512
Tang JS, Xie BX, Bian XL, Xue Y, Wei NN, Zhou JH, Hao YC, Li G, Zhang LR, Wang KW (2015) Identification and in vitro pharmacological characterization of a novel and selective α7 nicotinic acetylcholine receptor agonist, Br-IQ17B. Acta Pharmacol Sin 36(7):800–812. https://doi.org/10.1038/aps.2015.9
Dai CL, Li H, Hu X, Zhang J, Liu F, Iqbal K, Gong CX (2020) Neonatal exposure to anesthesia leads to cognitive deficits in old age: prevention with intranasal administration of insulin in mice. Neurotox Res 38:299–311. https://doi.org/10.1007/s12640-020-00223-y
Eibl JK, Strasser BC, Ross GM (2012) Structural, biological, and pharmacological strategies for the inhibition of nerve growth factor. Neurochem Int 61(8):1266–1275. https://doi.org/10.1016/j.neuint.2012.10.008
Liang C, Du F, Cang J, Xue Z (2018) Pink1 attenuates propofol-induced apoptosis and oxidative stress in developing neurons. J Anesth 32(1):62–69. https://doi.org/10.1007/s00540-017-2431-2
Knauss JL, Sun T (2013) Regulatory mechanisms of long noncoding RNAs in vertebrate central nervous system development and function. Neuroscience 235:200–214. https://doi.org/10.1016/j.neuroscience.2013.01.022
Zhao J, Yue D, Zhou Y, Jia L, Wang H, Guo M, Xu H, Chen C, Zhang J, Xu L (2017) The role of MicroRNAs in Aβ deposition and tau phosphorylation in Alzheimer’s disease. Front Neurol 8:342. https://doi.org/10.3389/fneur.2017.00342
Wu X, Wang Y, Sun Z, Ren S, Yang W, Deng Y, Tian C, Yu Y, Gao B (2019) Molecular expression and functional analysis of genes in children with temporal lobe epilepsy. J Integr Neurosci 18(1):71–77. https://doi.org/10.31083/j.jin.2019.01.13
Kumar S, Vijayan M, Reddy PH (2017) MicroRNA-455-3p as a potential peripheral biomarker for Alzheimer’s disease. Hum Mol Genet 26(19):3808–3822. https://doi.org/10.1093/hmg/ddx267
Kumar S, Reddy PH (2018) MicroRNA-455-3p as a potential biomarker for Alzheimer’s disease: an update. Front Aging Neurosci 10:41. https://doi.org/10.3389/fnagi.2018.00041
Guo H, Callaway JB, Ting JP (2015) Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat Med 21(7):677–687. https://doi.org/10.1038/nm.3893
Chi W, Li F, Chen H, Wang Y, Zhu Y, Yang X, Zhu J, Wu F, Ouyang H, Ge J, Weinreb RN, Zhang K, Zhuo Y (2014) Caspase-8 promotes NLRP1/NLRP3 inflammasome activation and IL-1β production in acute glaucoma. Proc Natl Acad Sci U S A 111(30):11181–11186. https://doi.org/10.1073/pnas.1402819111
Tan MS, Tan L, Jiang T, Zhu XC, Wang HF, Jia CD, Yu JT (2014) Amyloid-β induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer’s disease. Cell Death Dis 5(8):e1382. https://doi.org/10.1038/cddis.2014.348
Mawhinney LJ, de Rivero Vaccari JP, Dale GA, Keane RW, Bramlett HM (2011) Heightened inflammasome activation is linked to age-related cognitive impairment in Fischer 344 rats. BMC Neurosci 12:123. https://doi.org/10.1186/1471-2202-12-123
Tan CC, Zhang JG, Tan MS, Chen H, Meng DW, Jiang T, Meng XF, Li Y, Sun Z, Li MM, Yu JT, Tan L (2015) NLRP1 inflammasome is activated in patients with medial temporal lobe epilepsy and contributes to neuronal pyroptosis in amygdala kindling-induced rat model. J Neuroinflammation 12:18. https://doi.org/10.1186/s12974-014-0233-0
Ding J, Wang K, Liu W, She Y, Sun Q, Shi J, Sun H, Wang DC, Shao F (2016) Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535(7610):111–116. https://doi.org/10.1038/nature18590
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
H.G. and X.W. designed the study, supervised the data collection, analyzed the data, Y.Z. interpreted the data and prepare the manuscript for publication, S.L. supervised the data collection, analyzed the data and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors state that there are no conflicts of interest to disclose.
Ethical approval
Ethical approval was obtained from the Ethics Committee of The First Affiliated Hospital of Nanchang University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gong, H., Wan, X., Zhang, Y. et al. Downregulation of HOTAIR reduces neuronal pyroptosis by targeting miR-455-3p/NLRP1 axis in propofol-treated neurons in vitro. Neurochem Res 46, 1141–1150 (2021). https://doi.org/10.1007/s11064-021-03249-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03249-6