Abstract
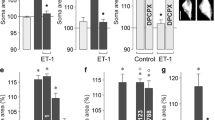
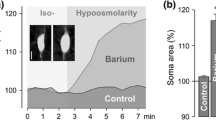
Retinal glial (Müller) cells possess an endogenous purinergic signal transduction cascade which normally prevents cellular swelling in osmotic stress. The cascade can be activated by osmotic or glutamate receptor-dependent ATP release. We determined whether activation of this cascade is altered in Müller cells of transgenic rats that suffer from a slow photoreceptor degeneration due to the expression of a truncated human cilia gene polycystin-2 (CMV-PKD21/703 HA). Age-matched Sprague–Dawley rats served as control. Retinal slices were superfused with a hypoosmotic solution (60 % osmolarity). Müller cells in retinas of PKD21/703 rats swelled immediately in hypoosmotic stress; this was not observed in control retinas. Pharmacological blockade of P2Y1 or adenosine A1 receptors induced osmotic swelling of Müller cells from control rats. The swelling induced by the P2Y1 receptor antagonist was mediated by induction of oxidative–nitrosative stress, mitochondrial dysfunction, production of inflammatory lipid mediators, and a sodium influx from the extracellular space. Exogenous VEGF or glutamate prevented the hypoosmotic swelling of Müller cells from PKD21/703 rats; this effect was mediated by activation of the purinergic signaling cascade. In neuroretinas of PKD21/703 rats, the gene expression levels of P2Y1 and A1 receptors, pannexin-1, connexin 45, NTPDases 1 and 2, and various subtypes of nucleoside transporters are elevated compared to control. The data may suggest that the osmotic swelling of Müller cells from PKD21/703 rats is caused by an abrogation of the osmotic ATP release while the glutamate-induced ATP release is functional. In the normal retina, ATP release and autocrine P2Y1 receptor activation serve to inhibit the induction of oxidative–nitrosative stress, mitochondrial dysfunction, and production of inflammatory lipid mediators, which otherwise will induce a sodium influx and cytotoxic Müller cell swelling under anisoosmotic conditions. Purinergic receptors may represent a target for the protection of retinal glial cells from mitochondrial oxidative stress.







Similar content being viewed by others
Abbreviations
- AOPCP:
-
Adenosine-5′-O-(α,β-methylene)-diphosphate
- ARL-67156:
-
6-N,N-diethyl-D-β,γ-dibromomethylene ATP
- ATP:
-
Adenosine 5′-triphosphate
- BAPTA/AM:
-
Bis-(o-aminophenoxy)ethane-N,N,N′,N′-tetra-acetic acid acetoxymethyl ester
- CNT:
-
Concentrative nucleoside transporter
- DPCPX:
-
8-Cyclopentyl-1,3-dipropylxanthine
- ENT:
-
Equilibrative nucleoside transporters
- Kir:
-
Inwardly rectifying potassium
- L-NAME:
-
Nω-nitro-l-arginine methyl ester hydrochloride
- LY341495:
-
(2S)-2-amino-2-[(1S,2S)-2-carboxycycloprop-1-yl]-3-(xanth-9-yl) propanoic acid
- MRS2179:
-
N 6-Methyl-2′-deoxyadenosine-3′,5′-bisphosphate
- NBTI:
-
N-Nitrobenzylthioinosine
- NPPB:
-
5-Nitro-2-(3-phenylpropylamino)benzoic acid
- NTPDase:
-
Nucleoside triphosphate diphosphohydrolase
- PKD:
-
Polycystic kidney disease
- SU1498:
-
(E)-3-(3,5-diisopropyl-4-hydroxyphenyl)-2-[(3-phenyl-n-propyl)amino-carbonyl]acrylnitrile
- VEGF:
-
Vascular endothelial growth factor
References
Balboa MA, Balsinde J (2006) Oxidative stress and arachidonic acid mobilization. Biochim Biophys Acta 1761:385–391
Barot M, Gokulgandhi MR, Mitra AK (2011) Mitochondrial dysfunction in retinal diseases. Curr Eye Res 36:1069–1077
Birkle DL, Bazan NG (1989) Light exposure stimulates arachidonic acid metabolism in intact rat retina and isolated rod outer segments. Neurochem Res 14:185–190
Bresnick GH (1983) Diabetic maculopathy. A critical review highlighting diffuse macular edema. Ophthalmology 90:1301–1317
Bringmann A, Uckermann O, Pannicke T, Iandiev I, Reichenbach A, Wiedemann P (2005) Neuronal versus glial cell swelling in the ischaemic retina. Acta Ophthalmol Scand 83:528–538
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Müller cells in the healthy and diseased retina. Prog Retin Eye Res 25:397–424
Chebabo SR, Hester MA, Aitken PG, Somjen GG (1995) Hypotonic exposure enhances synaptic transmission and triggers spreading depression in rat hippocampal tissue slices. Brain Res 695:203–216
Chua J, Nivison-Smith L, Fletcher EL, Trenholm S, Awatramani GB, Kalloniatis M (2013) Early remodeling of Müller cells in the rd/rd mouse model of retinal dystrophy. J Comp Neurol 521:2439–2453
Dmitriev AV, Govardovskii VI, Schwahn HN, Steinberg RH (1999) Light-induced changes of extracellular ions and volume in the isolated chick retina-pigment epithelium preparation. Vis Neurosci 16:1157–1167
Felmy F, Pannicke T, Richt JA, Reichenbach A, Guenther E (2001) Electrophysiological properties of rat retinal Müller (glial) cells in postnatally developing and in pathologically altered retinae. Glia 34:190–199
Feng Y, Wang Y, Stock O, Pfister F, Tanimoto N, Seeliger MW, Hillebrands JL, Hoffmann S, Wolburg H, Gretz N, Hammes HP (2009) Vasoregression linked to neuronal damage in the rat with defect of polycystin-2. PLoS One 4:e7328
Gallagher AR, Hoffmann S, Brown N, Cedzich A, Meruvu S, Podlich D, Feng Y, Könecke V, de Vries U, Hammes HP, Gretz N, Witzgall R (2006) A truncated polycystin-2 protein causes polycystic kidney disease and retinal degeneration in transgenic rats. J Am Soc Nephrol 17:2719–2730
Garcia TB, Pannicke T, Vogler S, Berk BA, Grosche A, Wiedemann P, Seeger J, Reichenbach A, Herculano AM, Bringmann A (2014) Nerve growth factor inhibits osmotic swelling of rat retinal glial (Müller) and bipolar cells by inducing glial cytokine release. J Neurochem 131:303–313
Hirrlinger PG, Wurm A, Hirrlinger J, Bringmann A, Reichenbach A (2008) Osmotic swelling characteristics of glial cells in the murine hippocampus, cerebellum, and retina in situ. J Neurochem 105:1405–1417
Iandiev I, Biedermann B, Bringmann A, Reichel MB, Reichenbach A, Pannicke T (2006) Atypical gliosis in Müller cells of the slowly degenerating rds mutant mouse retina. Exp Eye Res 82:449–457
Iandiev I, Pannicke T, Hollborn M, Wiedemann P, Reichenbach A, Grimm C, Remé CE, Bringmann A (2008) Localization of glial aquaporin-4 and Kir4.1 in the light-injured murine retina. Neurosci Lett 434:317–321
Jayakumar AR, Rao KV, Panickar KS, Moriyama M, Reddy PV, Norenberg MD (2008) Trauma-induced cell swelling in cultured astrocytes. J Neuropathol Exp Neurol 67:417–427
Karl A, Wurm A, Pannicke T, Krügel K, Obara-Michlewska M, Wiedemann P, Reichenbach A, Albrecht J, Bringmann A (2011) Synergistic action of hypoosmolarity and glutamine in inducing acute swelling of retinal glial (Müller) cells. Glia 59:256–266
Kaur C, Sivakumar V, Yong Z, Lu J, Foulds WS, Ling EA (2007) Blood-retinal barrier disruption and ultrastructural changes in the hypoxic retina in adult rats: the beneficial effect of melatonin administration. J Pathol 212:429–439
Krishnan G, Chatterjee N (2013) Detergent resistant membrane fractions are involved in calcium signaling in Müller glial cells of retina. Int J Biochem Cell Biol 45:1758–1766
Krügel K, Wurm A, Linnertz R, Pannicke T, Wiedemann P, Reichenbach A, Bringmann A (2010) Erythropoietin inhibits osmotic swelling of retinal glial cells by Janus kinase and extracellular signal-regulated kinases1/2-mediated release of vascular endothelial growth factor. Neuroscience 165:1147–1158
Krügel K, Wurm A, Pannicke P, Hollborn M, Karl A, Wiedemann P, Reichenbach A, Kohen L, Bringmann A (2011) Involvement of oxidative stress and mitochondrial dysfunction in the osmotic swelling of retinal glial cells from diabetic rats. Exp Eye Res 92:87–93
Kumar B, Gupta SK, Srinivasan BP, Nag TC, Srivastava S, Saxena R, Jha KA (2013) Hesperetin rescues retinal oxidative stress, neuroinflammation and apoptosis in diabetic rats. Microvasc Res 87:65–74
Lambert IH, Pedersen SF, Poulsen KA (2006) Activation of PLA2 isoforms by cell swelling and ischaemia/hypoxia. Acta Physiol (Oxf) 187:75–85
Lees GJ (1991) Inhibition of sodium-potassium-ATPase: a potentially ubiquitous mechanism contributing to central nervous system neuropathology. Brain Res Rev 16:283–380
Li Y, Holtzclaw LA, Russell JT (2001) Müller cell Ca2+ waves evoked by purinergic receptor agonists in slices of rat retina. J Neurophysiol 85:986–994
Linnertz R, Wurm A, Pannicke T, Krügel K, Hollborn M, Härtig W, Iandiev I, Wiedemann P, Reichenbach A, Bringmann A (2011) Activation of voltage-gated Na+ and Ca2+ channels is required for vesicular release of glutamate from retinal glial cells implicated in cell volume regulation. Neuroscience 188:23–34
Milenkovic I, Weick M, Wiedemann P, Reichenbach A, Bringmann A (2003) P2Y receptor-mediated stimulation of Müller glial cell DNA synthesis: dependence on EGF and PDGF receptor transactivation. Invest Ophthalmol Vis Sci 44:1211–1220
Newman EA (2001) Propagation of intercellular calcium waves in retinal astrocytes and Müller cells. J Neurosci 21:2215–2223
Newman EA (2003) Glial cell inhibition of neurons by release of ATP. J Neurosci 23:1659–1666
Osborne NN, Álvarez CN, del Olmo Aguado S (2014) Targeting mitochondrial dysfunction as in aging and glaucoma. Drug Discov Today 19:1613–1622
Owada S, Larsson O, Arkhammar P, Katz AI, Chibalin AV, Berggren PO, Bertorello AM (1999) Glucose decreases Na+, K+-ATPase activity in pancreatic β-cells: an effect mediated via Ca2+-independent phospholipase A2 and protein kinase C-dependent phosphorylation of the α-subunit. J Biol Chem 274:2000–2008
Pannicke T, Iandiev I, Uckermann O, Biedermann B, Kutzera F, Wiedemann P, Wolburg H, Reichenbach A, Bringmann A (2004) A potassium channel-linked mechanism of glial cell swelling in the postischemic retina. Mol Cell Neurosci 26:493–502
Reichenbach A, Bringmann A (2015) Purinergic signaling in retinal degeneration and regeneration. Neuropharmacology, (in press)
Rothova A, Suttorp-van Schulten MS, Frits Treffers W, Kijlstra A (1996) Causes and frequency of blindness in patients with intraocular inflammatory disease. Br J Ophthalmol 80:332–336
Staub F, Winkler A, Peters J, Kempski O, Kachel V, Baethmann A (1994) Swelling, acidosis, and irreversible damage of glial cells from exposure to arachidonic acid in vitro. J Cereb Blood Flow Metab 14:1030–1039
Stepinac TK, Chamot SR, Rungger-Brändle E, Ferrez P, Munoz JL, Van Den Bergh H, Riva CE, Pournaras CJ, Wagnieres GA (2005) Light-induced retinal vascular damage by Pd-porphyrin luminescent oxygen probes. Invest Ophthalmol Vis Sci 46:956–966
Uckermann O, Vargová L, Ulbricht E, Klaus C, Weick M, Rillich K, Wiedemann P, Reichenbach A, Syková E, Bringmann A (2004) Glutamate-evoked alterations of glial and neuronal cell morphology in the guinea-pig retina. J Neurosci 24:10149–10158
Uckermann O, Wolf A, Kutzera F, Kalisch F, Beck-Sickinger A, Wiedemann P, Reichenbach A, Bringmann A (2006) Glutamate release by neurons evokes a purinergic inhibitory mechanism of osmotic glial cell swelling in the rat retina: activation by neuropeptide Y. J Neurosci Res 83:538–550
Vogler S, Pannicke T, Hollborn M, Grosche A, Busch S, Hoffmann S, Wiedemann P, Reichenbach A, Hammes HP, Bringmann A (2013) Müller cell reactivity in response to photoreceptor degeneration in rats with defective polycystin-2. PLoS One 8:e61631
Vogler S, Grosche A, Pannicke T, Ulbricht E, Wiedemann P, Reichenbach A, Bringmann A (2013) Hypoosmotic and glutamate-induced swelling of bipolar cells in the rat retina: comparison with swelling of Müller glial cells. J Neurochem 126:372–381
Voigt J, Grosche A, Vogler S, Pannicke T, Hollborn M, Kohen L, Wiedemann P, Reichenbach A, Bringmann A (2015) Nonvesicular release of ATP from rat retinal glial (Müller) cells is differentially mediated in response to osmotic stress and glutamate. Neurochem Res 40:651–660
Wurm A, Pannicke T, Wiedemann P, Reichenbach A, Bringmann A (2008) Glial cell-derived glutamate mediates autocrine cell volume regulation in the retina: activation by VEGF. J Neurochem 104:386–399
Wurm A, Lipp S, Pannicke T, Linnertz R, Krügel U, Schulz A, Färber K, Zahn D, Grosse J, Wiedemann P, Chen J, Schöneberg T, Illes P, Reichenbach A, Bringmann A (2010) Endogenous purinergic signaling is required for osmotic volume regulation of retinal glial cells. J Neurochem 112:1261–1272
Zheng W, Watts LT, Holstein DM, Prajapati SI, Keller C, Grass EH, Walter CA, Lechleiter JD (2010) Purinergic receptor stimulation reduces cytotoxic edema and brain infarcts in mouse induced by photothrombosis by energizing glial mitochondria. PLoS One 5:e14401
Zheng W, Talley Watts L, Holstein DM, Wewer J, Lechleiter JD (2013) P2Y1R-initiated, IP3R-dependent stimulation of astrocyte mitochondrial metabolism reduces and partially reverses ischemic neuronal damage in mouse. J Cereb Blood Flow Metab 33:600–611
Acknowledgments
This work was supported by grants from the Deutsche Forschungsgemeinschaft (GRK 1097 and RE 849/16-1 to A.R.; PA 615/2-1 to T.P.; GRK 1874 to H.P.H.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Vogler, S., Pannicke, T., Hollborn, M. et al. Impaired Purinergic Regulation of the Glial (Müller) Cell Volume in the Retina of Transgenic Rats Expressing Defective Polycystin-2. Neurochem Res 41, 1784–1796 (2016). https://doi.org/10.1007/s11064-016-1894-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1894-0