Abstract
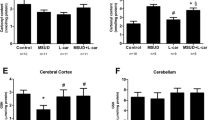
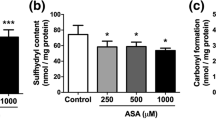
Cysteamine is a cystine-depleting drug used in the treatment of cystinosis, a metabolic disorder caused by deficiency of the lysosomal cystine carrier. As a result, cystine accumulates within lysosomes in many tissues and organs, including the nervous system. Studies with cystine dimethyl ester loaded cells suggest that cystine might induce apoptosis through oxidative stress. Our objective was to investigate the effects of co-administration of cysteamine with the oxidant cystine dimethyl ester on several parameters of oxidative stress in the brain cortex of rats. Animals were injected with 1.6 μmol/g cystine dimethyl ester and/or 0.26 μmol/g body weight cysteamine. Cystine dimethyl ester induced lipoperoxidation, protein carbonylation, and stimulated superoxide dismutase, glutathione peroxidase and catalase activities, probably through the formation of free radicals. Cysteamine prevented those effects, possibly increasing cellular thiol pool and acting as a scavenger of free radicals. These results suggest that the antioxidant effect of cysteamine may be important in the treatment of cystinosis.






Similar content being viewed by others
References
Pisoni RL, Park GY, Velilla VQ et al (1995) Detection and characterization of a transport system mediating cysteamine entry into human fibroblast lysosomes. Specificity for aminoethylthiol and aminoethylsulfide derivatives. J Biol Chem 270(3):1179–1184
Thoene JG, Oshima RG, Crawhall JC et al (1976) Cystinosis. Intracellular cystine depletion by aminothiols in vitro and in vivo. J Clin Invest 58(1):180–189
Dominy JE, Simmons CR, Hirschberger LL et al (2007) Discovery and characterization of a second mammalian thiol dioxygenase: Cysteamine dioxygenase. J Biol Chem. Jun 20; [Epub ahead of print]
Salvador RA, Davison C, Smith PK (1957) Metabolism of cysteamine. J Pharmacol Exp Ther 121(2):258–265
Lloyd JB (1986) Disulphide reduction in lysosomes. The role of cysteine. Biochem J 237(1):271–272
Gahl WA (2003) Early oral cysteamine therapy for nephropathic cystinosis. Eur J Pediatr 162(1):38–41
Tsilou ET, Rubin BI, Reed G et al (2006) Nephropathic cystinosis: posterior segment manifestations and effects of cysteamine therapy. Ophthalmology 113(6):1002–1009
Tsilou E, Zhou M, Gahl W et al (2007) Ophthalmic manifestations and histopathology of infantile nephropathic cystinosis: report of a case and review of the literature. Surv Ophthalmol 52(1):97–105
Gahl WA, Thoene JG, Schneider JA (2001) Cystinosis: a disorder of lysosomal membrane transport. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The Metabolic & Molecular Bases of Inherited Diseases. McGraw- Hill, New York, pp 5085–5108
Nichols SL, Press GA, Schneider JA et al (1990) Cortical atrophy and cognitive performance in infantile nephropathic cystinosis. Pediatr Neurol 6(6):379–381
Jonas AJ, Conley SB, Marshall R et al (1987) Nephropathic cystinosis with central nervous system involvement. Am J Med 83(5):966–970
Ballantyne AO, Scarvie KM, Trauner DA (1997) Academic achievement in individuals with infantile nephropathic cystinosis. Am J Med Genet 74(2):157–161
Vogel DG, Malekzadeh MH, Cornford ME et al (1990) Central nervous system involvement in nephropathic cystinosis. J Neuropathol Exp Neurol 49(6):591–599
Depape-Brigger D, Goldman H, Scriver CR et al (1977) The in vivo use of dithiothreitol in cystinosis. Pediatr Res 11(2):124–131
Gahl WA, Dalakas MC, Charnas L et al (1988) Myopathy and cystine storage in muscles in a patient with nephropathic cystinosis. N Engl J Med 319(22):1461–1464
Kleta R, Gahl WA (2002) Cystinosis: antibodies and healthy bodies. J Am Soc Nephrol 13(8):2189–2191
Foreman JW, Benson LL, Wellons M et al (1995) Metabolic studies of rat renal tubule cells loaded with cystine: the cystine dimethylester model of cystinosis. J Am Soc Nephrol 6(2):269–272
Park M, Helip-Wooley A, Thoene J (2002) Lysosomal cystine storage augments apoptosis in cultured human fibroblasts and renal tubular epithelial cells. J Am Soc Nephrol 13(12):2878–2887
Cantoni O, Brandi G, Albano A et al (1995) Action of cystine in the cytotoxic response of Escherichia coli cells exposed to hydrogen peroxide. Free Radic Res 22:275–283
Fleck RM, Rodrigues Junior V, Giacomazzi J et al (2005) Cysteamine prevents and reverses the inhibition of creatine kinase activity caused by cystine in rat brain cortex. Neurochem Int 46(5):391–397
Feksa LR, Cornelio A, Dutra-Filho CS (2004) Inhibition of pyruvate kinase activity by cystine in brain cortex of rats. Brain Res 1012(1–2):93–100
Foreman JW, Bowring MA, Lee J et al (1987) Effect of cystine dimethyl ester on renal solute handling and isolated renal tubule transport in the rat. A new model Fanconi syndrome Metab 36:1185–1191
Ben-Nun A, Bashan N, Potashnik R et al (1993) Cystine loading induces Fanconi’s syndrome in rats: in vivo and vesicle studies. Am J Physiol 265:839–844
Llesuy SF, Milei J, Molina H (1985) Comparison of lipid peroxidation and myocardial damage induced by adriamycin and 4′-epiadriamycin in mice. Tumori 71(3):241–249
Gonzalez Flecha B, Llesuy S, Boveris A (1991) Hydroperoxide-initiated chemiluminescence: an assay for oxidative stress in biopsies of heart, liver, and muscle Free Radic. Biol Med 10(2):93–100
LeBel CP, Ischiropoulos H, Bondy SC (1992) Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem Res Toxicol 5:227–231
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358
Kehrer JP (2000) The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 149:43–50
Reznick AZ, Packer L (1994) Oxidative damage to proteins: Spectrophotometric method for carbonyl assay. Methods Enzymol 233:357–363
Uchida K (2003) Histidine and lysine as targets of oxidative modification. Amino Acids 25:249–257
Levine RL, Garland D, Oliver CN et al (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Aebi H (1984) Catalase, in vitro. Methods Enzymol 105:121–126
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–332
Zahler WL, Cleland WW (1968) A specific and sensitive assay for disulfides. J Biol Chem 243:716–719
Lowry OH, Rosebrough NJ, Farr AL (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Leech NL, Barrett KC, Morgan GA (2005) SPSS for intermediate statistics. Use and interpretation, Lawrence Erlbaum Associates Publishers (ed), London, pp 46–62
Rech VC, Feksa LR, Arevalo do Amaral MF (2007) Promotion of oxidative stress in kidney of rats loaded with cystine dimethyl ester. Pediatr Nephrol 22(8):1121–1128
Wood PL, Khan MA, Moskal JR (2007) Cellular thiol pools are responsible for sequestration of cytotoxic reactive aldehydes: Central role of free cysteine and cysteamine. Brain Res 1158:158–163
Fox JH, Barber DS, Singh B, Zucker B, Swindell MK, Norflus F, Buzescu R, Chopra R, Ferrante RJ, Kazantsev A, Hersch SM. (2004) Cystamine increases L-cysteine levels in Huntington’s disease transgenic mouse brain and in a PC12 model of polyglutamine aggregation. J Neurochem 91:413–422
Winterbourn CC, Metodiewa CD (1999) Reactivity of biologically important thiol compounds with superoxide and hydrogen peroxide Free Radic. Biol Med 27:322–328
Rosenberg PA, Li Y, Ali S et al (1999) Intracellular redox state determines whether nitric oxide is toxic or protective to rat oligodendrocytes in culture. J Neurochem 73:476–484
Winterbourn CC, Peskin AV, Parsons-Mair HN (2002) Thiol oxidase activity of copper, zinc superoxide dismutase. J Biol Chem 277:1906–1911
Kono Y, Fridovich I (1982) Superoxide radical inhibits catalase. J Biol Chem 257:5751–5754
Jones DP (2006) Redefining oxidative stress. Antioxid Redox Signal 8:1865–1879
Jones DP (2006) Extracellular redox state: refining the definition of oxidative stress in aging. Rejuvenation Res 9:169–181
Jones DP, Go YM, Anderson CL et al (2004) Cysteine/cystine couple is a newly recognized node in the circuitry for biologic redox signaling and control. FASEB J 18:1246–1248
Liu W, Kato M, Akhand AA et al (2000) 4-hydroxynonenal induces a cellular redox status-related activation of the caspase cascade for apoptotic cell death. J Cell Sci 113:635–641
Antunes F, Cadenas E (2001) Cellular titration of apoptosis with steady state concentrations of H2O2: submicromolar levels of H2O2 induce apoptosis through Fenton chemistry independent of the cellular thiol state. Free Radic Biol Med 30:1008–1018
Tardy C, Andrieu-Abadie N, Salvayre R et al (2004) Lysosomal storage diseases: is impaired apoptosis a pathogenic mechanism? Neurochem. Res 29:871–880
Boya P, Andreau K, Poncet D et al (2003) Lysosomal membrane permeabilization induces cell death in a mitochondrion-dependent fashion. J Exp Med 197:1323–1334
Park MA, Thoene JG (2005) Potential role of apoptosis in development of the cystinotic phenotype. Pediatr Nephrol 20:441–446
Park MA, Pejovic V, Kerisit KG et al (2006) Increased apoptosis in cystinotic fibroblasts and renal proximal tubule epithelial cells results from cysteinylation of protein kinase C (delta). J Am Soc Nephrol 17:3167–3175
Jiang S, Moriarty-Craige SE, Orr M et al (2005) Oxidant-induced apoptosis in human retinal pigment epithelial cells: dependence on extracellular redox state. Invest Ophtalmol Vis Sci 46:1054–1061
Sestili P, Martinelli C, Bravi G et al (2006) Creatine supplementation affords cytoprotection in oxidatively injured cultured mammalian cells via direct antioxidant activity. Free Radic Biol Med 40:837–849
Hatano E, Tanaka A, Kanazawa A et al (2004) Inhibition of tumor necrosis factor-induced apoptosis in transgenic mouse liver expressing creatine kinase. Liver Int 24:384–393
Acknowledgements
This work was supported in part by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-Brazil), Fundação de Amparo à Pesquisa do Rio Grande do Sul (FAPERGS, RS-Brazil), Programa de Núcleos de Excelência (PRONEX-CNPq /FAPERGS -Brazil) and Rede Instituto Brasileiro de Neurociência (IBN-Net), FINEP #01.06.0842-00.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kessler, A., Biasibetti, M., da Silva Melo, D.A. et al. Antioxidant Effect of Cysteamine in Brain Cortex of Young Rats. Neurochem Res 33, 737–744 (2008). https://doi.org/10.1007/s11064-007-9486-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9486-7