Abstract
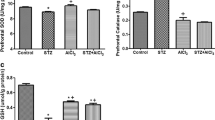
It is believed that oxidative stress (OS) plays a central role in the pathogenesis of metabolic diseases like diabetes mellitus (DM) and its complications (like peripheral neuropathy) as well as in neurodegenerative disorders like sporadic Alzheimer’s disease (sAD). Representative experimental models of these diseases are streptozotocin (STZ)-induced diabetic rats and STZ-intracerebroventricularly (STZ-icv) treated rats, in which antioxidant capacity (AC) against peroxyl (ORAC-ROO •) and hydroxyl (ORAC-OH •) free radicals (FR) was measured in three different brain regions: the hippocampus (HPC), the cerebellum (CB), and the brain stem (BS) by means of oxygen radical absorbance capacity (ORAC) assay. In the brain of both STZ-induced diabetic and STZ-icv treated rats decreased AC has been found demonstrating regionally specific distribution. In the diabetic rats these abnormalities were not associated with the development of peripheral diabetic neuropathy (PDN). Also, these abnormalities were not prevented by the intracerebroventricularly (icv) pretreatment of glucose transport inhibitor 5–thio-d-glucose (TG) in the STZ-icv treated rats, suggesting different mechanism of STZ-induced central effects from those at the periphery. Similarities of the OS alterations in the brain of STZ-icv rats and humans with sAD could be useful in the search for the new drugs in the treatment of sAD that have antioxidant activity. In the STZ-induced diabetic animals the existence of PDN was tested by the paw pressure test, 3 weeks following the diabetes induction. Mechanical nociceptive thresholds were measured three times at 10–min intervals by applying increased pressure to the hind paw until the paw-withdrawal or overt struggling was elicited. Only those diabetic animals which demonstrated decreased withdrawal threshold values in comparison with the control non-diabetic animals (C) were considered to have developed the PDN.


Similar content being viewed by others
Abbreviations
- AAPH:
-
2,2′-Azobis (2–amidino-propane) dihydrochloride
- AC:
-
Antioxidant capacity
- AD:
-
Alzheimer’s disease
- AL:
-
Alloxan
- AUC:
-
Area under curve
- BCT:
-
Betacytotoxic
- β-PE :
-
Beta-phycoerythrin
- BS:
-
Brain stem
- BSA:
-
Albumin
- C:
-
Control non-diabetic animals
- CB:
-
Cerebellum
- DM :
-
Diabetes mellitus
- DNA:
-
Deoxyribonucleic acid
- f.c. :
-
Final concentration
- FL:
-
Fluorescein
- FR:
-
Free radicals
- GLUT2:
-
Glucose transporter two
- HPC:
-
Hippocampus
- icv :
-
Intracerebroventricularly
- ip:
-
Intraperitoneal
- LC/MS :
-
Liquid chromatography/mass spectrometry
- ORAC :
-
Oxygen radical absorbance capacity
- ORAC-ROO • :
-
Antioxidant capacity against peroxyl free radicals
- ORAC–OH • :
-
Antioxidant capacity against hydroxyl free radicals
- OS:
-
Oxidative stress
- PDN :
-
Peripheral diabetic neuropathy
- ROS :
-
Reactive oxygen species
- sAD :
-
Sporadic Alzheimer’s disease
- sc :
-
Subcutaneous
- STZ :
-
Streptozotocin
- Trolox:
-
6–Hydroxy-2,5,7,8-tetramethyl-2-carboxylic acid
- TE :
-
Trolox equivalents
- TG:
-
5-Thio-d-glucose
References
Altan N, Dinçel AS, Koca C (2006) Diabetes mellitus and oxidative stress. Turk J Biochem 31:51–56
Sofic E, Rustembegovic A, Kroyer G, Cao G (2002) Serum antioxidant capacity in neurological, psychiatric, renal diseases and cardiomyopathy. J Neural Transm 109:711–719
Sharma RK, Pasqualotto FF, Nelson DR, Thomas AJ Jr, Agarwal A (1999) The reactive oxygen species-total antioxidant capacity score is a new measure of oxidative stress to predict male infertility. Hum reprod 14:2801–2807
Danova K, Dobisova A, Fischer V, Halcak L, Minarova H, Olejarova I, Pechan I (2005) Production of reactive oxygen species and antioxidant defense systems in patients after coronary artery bypass grafting: one-week follow-up study. J Clin Basic Cardiol 8:33–36
Prior RL, Hoang H, Gu L, Wu X, Bacchiocca M, Howard L, Hampsch-Woodill M, Huang D, Ou B, Jacob R (2003) Assays for hydrophilic and lipophilic antioxidant capacity [oxygen radical absorbance capacity (ORAC-FL)] of plasma and other biological and food samples. J Agric Food Chem 21:3273–3279
Biessels GJ, van der Heide LP, Kamal A, Bleys RL, Gispen WH (2002) Ageing and diabetes: implications for brain function. Eur J Pharmacol 441:1–14
Kumar JS, Menon VP (1993) Effect of diabetes on levels of lipid peroxides and glycolipids in rat brain. Metabolism 42(11):1435–1439
Ohkuwa T, Sato Y, Naoi M (1995) Hydroxylradical formation in diabetic rats induced by streptozotocin. Life Sci 56(21):1789–1798
Szkudelski T (2001) The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res 50:537–546
Ding A, Nitsch R, Hoyer S (1992) Changes in brain monoaminergic neurotransmitter concentrations in rat after intracerebroventricular injections of streptozotocin. J Cereb Blood Flow Metabol 12:103–109
Lacković Z, Šalković M (1990) Streptozocin and alloxan produce alterations in rat brain monoamines independently of pancreatic beta cells destruction. Life Sci 46:49–54
Salkovic M, Sabolic I, Lackovic Z (1995) Striatal D1 and D2 receptors after intracerebroventricular applications of alloxan and streptozotocin in rat. J Neural Transm 100:127–145
Salkovic-Petrisic M, Lackovic Z (2003) Intracerebroventricular administration of betacytotoxics alters expression of brain monoamine transporter genes. J Neural Transm 110:15–29
Lannert H, Hoyer S (1998) Intracerebroventricular administration of streptozotocin causes long-term diminutions in learning and memory abilities and in cerebral energy metabolism in adult rats. Behav Neurosci 112:1199–1208
Prickaerts J, Fahring T, Blokland A (1999) Cognitive performance and biochemical markers in septum, hippocampus and striatum of rats after an i.c.v. injection of streptozotocin: a correlation analysis. Behav Brain Res 102:73–88
Hoyer S (1998) Is sporadic Alzheimer disease the brain type of non-insulin dependent diabetes mellitus? A challenging hypothesis. J Neural Transm 105:415–422
Salkovic-Petrisic M, Tribl F, Schmidt M, Hoyer S, Riederer P (2006) Alzheimer-like changes in protein kinase B and glycogen synthase kinase-3 in rat frontal cortex and hippocampus after damage to the insulin signalling pathway. J Neurochem 96:1005–1015
Hsiao K, Chapman P, Nilsen S, Eckman C, Harigaya Y, Younkin S, Yang F, Cole G (1996) Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice. Science 274:99–102
Sharma M, Gupta YK (2001) Effect of chronic treatment of melatoninon learning, memory and oxidative deficiencies induced by intracerebroventricular streptozotocin in rats. Pharmacol Biochem Behav 70:325–331
Sharma M, Gupta YK (2002) Chronic treatment with trans resveratrol prevents intracerebroventricular streptozotocin induced cognitive impairment and oxidative stress in rats. Life Sci 71:2489–2498
Mariani E, Polidori MC, Cherubini A, Meccoci P (2005) Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J Chromatogr B Analyt Technol Biomed Life Sci 827:65–75
Cao G, Alessio HM, Cutler RG (1993) Oxygen-radical absorbance capacity assay for antioxidants. Free Radical Biol Med 14:303–311
Cao G, Verdon C, Wu AHB, Wang H, Prior RL (1995) Automated oxygen radical absorbance capacity assay using the COBAS FARA II. Clin Chem 41:1738–1744
Cao G, Sofic E, Prior RL (1996) Antioxidant capacity of tea and common vegetables. J Agric Food Chem 44(11):3426–3431
Cao G, Sofic E, Prior R (1997) Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radical Biol Med 22:749–760
Wang CC, Chu CY, Chy KO, Choy KW, Khaw KS, Rogers MS, Pang CP (2004) Trolox equivalent antioxidant capacity assay versus oxygen radical absorbance capacity assay in plasma. Clin Chem 50:952–954
Sofic E, Sapcanin A, Tahirovic I, Gavrankapetanovic I, Jellinger K, Reynolds GP, Tatschner T, Riederer P (2006) Antioxidant capacity in postmortem brain tissues of Parkinson’s and Alzheimer’s diseases. J Neural Transm Suppl 71:39–43
Wayner DDM, Burton GW, Ingold KU, Locke S (1985) Quantitative measurement of the total, peroxyl radical-trapping antioxidant capacity of human plasma by controlled peroxidation. FEBS Lett 187:33–37
Glazer AN (1990) Phycoerythrin fluorescence-based assay for reactive oxygen species. Methods Enzymol 186:161–168
Ghiselli A, Serafini M, Maiani G, Assini E, Ferro-Luzzi A (1994) A fluorescence-based method for measuring total plasma antioxidant capability. Free Radical Biol Med 18:29–36
Grisham MB (1992) Reactive metabolites of oxygen and nitrogen in biology and medicine. Landes RG Co., Austin, TX, p 10
Ou B, Hampsch-Woodill M, Prior RL (2001) Development and validation of an improved Oxygen Radical Absorbance Capacity assay using fluorescein as the fluorescent probe. J Agric Food Chem 49:4619–4626
Noble EP, Wurtman RJ, Axelrod J (1967) A simple and rapid method for injecting H3-norepinephrine into the lateral ventricle of the rat brain. Life Sci 6:281–291
Randall LO, Selitto JJ (1957) A method for measurements of analgesic activity on inflamed tissue. Arch Int Pharmacodyn Ther 111:409–419
Bach-Rojecky L, Lackovic Z (2005) Antinociceptive effect of botulinum toxin type A in rat model of carrageenan and capsaicin induced pain. Croat Med J 46:201–208
Bronstein IN, Semendjajev KA (1962) Matematički priručnik za inženjere i studente. «Tehnička knjiga», Zagreb, p S457
Gernot P (1977) Proteine. In: Clinische Chemie, Kurzlehrbuch und Kommentar zum Gegenstandskatalog für den Ersten Abschnitt der Ärztlichen Prüfung 1. Auflage, Rathgeber Verlag, München, pp 308–367
Kuyvenhoven JP, Meinders AE (1999) Oxidative stress and diabetes mellitus. Pathogenesis of long-term complications. Eur J Intern Med 10:9–19
Evans JL, Goldfine ID, Maddux BA, Grodsky GM (2002) Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocrine Rev 23:599–622
Cao G, Giovanoni M, Prior R (1996a) Antioxidant capacity in different tissues of young and old rats. Proc Soc Exp Biol Med 211:359–365
McEwen BS, Reagan LP (2004) Glucose transporter expression in the central nervous system: relationship to synaptic function. Eur J Pharmacol 490:13–24
Grünblatt E, Salkovic-Petrisic M, Osmanovic J, Riederer P, Hoyer S (2007) Brain insulin system dysfunction in streptozotocin intracerebroventricularly treated rats generates hyperphosphorylated tau protein. J Neurochem 101(3):757–770
Acknowledgments
Research was supported by the DAAD-German Academic Exchange Service, Stability Pact for South-Eastern Europe (project No. A/04/20017) and the Croatian Ministry of Science, Education and Sport (project No. 0108253). Mrs. Bozica Hrzan is thanked for the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue dedicated to Dr. Moussa Youdim.
Rights and permissions
About this article
Cite this article
Tahirovic, I., Sofic, E., Sapcanin, A. et al. Reduced Brain Antioxidant Capacity in Rat Models of Betacytotoxic-Induced Experimental Sporadic Alzheimer’s Disease and Diabetes Mellitus. Neurochem Res 32, 1709–1717 (2007). https://doi.org/10.1007/s11064-007-9410-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9410-1