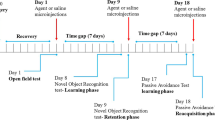
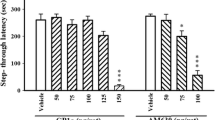
Serotonin (5-HT) is involved in memory processing via different types of its receptors within brain regions. We investigated the role of 5-HT7 receptors in acquisition, consolidation, and retrieval phases in the passive avoidance (PA) learning and memory test in mice. Adult male mice (body mass 20–35 g) were randomly divided into four experimental groups (n = 10 in each). Animals of the control group received saline, while other groups of mice received SB-269970 (a selective 5-HT7 receptor antagonist, 10 mg/kg, i.p.) 15 min before training, immediately after training, and 24 h after the acquisition trial. Numbers of trials to acquisition (NT), initial entry latency into the dark compartment in the learning phase, step-through latency, and time of stay in the dark compartment in the retention phase of the PA test (STLr and TDC, respectively) were measured. The number of trials to acquisition in the group receiving SB-269970 before training showed no significant differences in comparison with the control group. The STLr in the group receiving SB-269970 after training was significantly greater, while its TDC was shorter than in the control. There were no significant differences in the STLr and TDC between the control and SB-269970 group receiving the blocker before retrieval. It is concluded that blocking of 5-HT7 receptors could improve consolidation of the passive avoidance memory, but this procedure exerted no significant effects on acquisition and retrieval in the PA test.
Similar content being viewed by others
References
R. J. McDonald and N. S. Hong, “How does a specific learning and memory system in the mammalian brain gain control of behavior?” Hippocampus, 23, 1084–1102 (2013).
E. G. Antzoulatos and J. H. Byrne, “Learning insights transmitted by glutamate,” Trends Neurosci., 27, 555–560 (2004).
T. Myhrer, “Neurotransmitter systems involved in learning and memory in the rat: a meta-analysis based on studies of four behavioral tasks,” Brain Res. Brain Res. Rev., 41, 268–287 (2003).
M. Berger, J. A. Gray, and B. L. Roth, “The expanded biology of serotonin,” Annu. Rev. Med., 60, 355–366 (2009).
D. Hoyer, J. P. Hannon, and G. R. Martin, “Molecular, pharmacological and functional diversity of 5-HT receptors,” Pharmacol. Biochem. Behav., 71, 533–554 (2002).
J. D. McCorvy, and B. L. Roth, “Structure and function of serotonin G protein-coupled receptors,” Pharmacol. Ther., 150, 129–142 (2015).
M. Filip and M. Bader, “Overview on 5-HT receptors and their role in physiology and pathology of the central nervous system,” Pharmacol. Rep., 61, 761–777 (2009).
A. Brenchat, X. Nadal, L. Romero, et al., “Pharmacological activation of 5-HT7 receptors reduces nerve injury-induced mechanical and thermal hypersensitivity,” Pain, 149, 483–494 (2010).
J. M. Monti, M. Leopoldo, H. Jantos, and P. Lagos, “Microinjection of the 5-HT7 receptor antagonist SB-269970 into the rat brainstem and basal forebrain: site-dependent effects on REM sleep,” Pharmacol. Biochem. Behav., 102, 373–380 (2012).
S. Doly, J. Fischer, M. J. Brisorgueil, et al., “Pre- and postsynaptic localization of the 5-HT7 receptor in rat dorsal spinal cord: immunocytochemical evidence,” J. Comp. Neurol., 490, 256–269 (2005).
A. Wesołowska, A. Nikiforuk, and K. Stachowicz, “Potential anxiolytic and antidepressant effects of the selective 5-HT7 receptor antagonist SB 269970 after intrahippocampal administration to rats,” Eur. J. Pharmacol., 553, 185–190 (2006).
A. Wesołowska, A. Nikiforuk, K. Stachowicz, and E. Tatarczyńska, “Effect of the selective 5-HT7 receptor antagonist SB 269970 in animal models of anxiety and depression,” Neuropharmacology, 51, 578–586 (2006).
A. Wesołowska and M. Kowalska, “Influence of serotonin 5-HT(7) receptor blockade on the behavioral and neurochemical effects of imipramine in rats,” Pharmacol. Rep., 60, 464–474 (2008).
Q. J. Zhang, C. X. Du, H. H. Tan, et al., “Activation and blockade of serotonin7 receptors in the prelimbic cortex regulate depressive-like behaviors in a 6-hydroxy-dopamine-induced Parkinson’s disease rat model,” Neuroscience, 311, 45–55 (2015).
A. O. el-Kadi and S. I. Sharif, “The role of 5-HT in the expression of morphine withdrawal in mice,” Life Sci., 57, 511–516 (1995).
S. Shahidi and N. Hashemi-Firouzi, “The effects of a 5-HT7 receptor agonist and antagonist on morphine withdrawal syndrome in mice,” Neurosci. Lett., 578, 27–32 (2014).
A. J. Roberts, T. Krucker, C. L. Levy, et al., “Mice lacking 5-HT receptors show specific impairments in contextual learning,” Eur. J. Neurosci., 19, 1913–1922 (2004).
G. Sarkisyan and P. B. Hedlund, “The 5-HT7 receptor is involved in allocentric spatial memory information processing,” Behav. Brain Res., 202, 26–31 (2009).
L. Speranza, J. Labus, F. Volpicelli, et al., “Serotonin 5-HT7 receptor increases the density of dendritic spines and facilitates synaptogenesis in forebrain neurons,” J. Neurochem., 141, 647–661 (2017).
N. Hashemi-Firouzi, A. Komaki, S. Soleimani Asl, and S. Shahidi, “The effects of the 5-HT7 receptor on hippocampal long-term potentiation and apoptosis in a rat model of Alzheimer’s disease,” Brain Res. Bull., 135, 85–91 (2017).
G. S. Perez-Garcia and A. Meneses, “Effects of the potential 5-HT7 receptor agonist AS 19 in an autoshaping learning task,” Behav. Brain Res., 163, 136–140 (2005).
C. Carbone, A. Adinolfi, S. Cinque, et al., “Activation of 5-HT7 receptor by administration of its selective agonist, LP-211, modifies explorative-curiosity behavior in rats in two paradigms which differ in visuospatial parameters,” CNS Neurosci. Ther., 24, 712–720 (2018).
T. M. Eriksson, A. Golkar, G. C. Ekstrom, et al., “5-HT7 receptor stimulation by 8-OH-DPAT counteracts the impairing effect of 5-HT(1A) receptor stimulation on contextual learning in mice,” Eur. J. Pharmacol., 596, 107–110 (2008).
G. Perez-Garcia and A. Meneses, “Memory time-course: mRNA 5-HT1A and 5-HT7 receptors,” Behav. Brain Res., 202, No. 1, 102–113 (2009).
T. Freret, E. Paizanis, G. Beaudet, et al., “Modulation of 5-HT7 receptor: effect on object recognition performances in mice,” Psychopharmacology, 231, 393–400 (2014).
S. J. Ballaz, H. Akil, and S. J. Watson, “The 5-HT7 receptor: role in novel object discrimination and relation to novelty-seeking behavior,” Neuroscience, 149, 192–202 (2007).
A. Gasbarri, A. Cifariello, A. Pompili, and A.Meneses, “Effect of 5-HT 7 antagonist SB-269970 in the modulation of working and reference memory in the rat,” Behav. Brain Res., 195, No. 1, 164–170 (2008).
A. Komaki, S. A. Karimi, I. Salehi, et al., “The treatment combination of vitamins E and C and astaxanthin prevents high-fat diet induced memory deficits in rats,” Pharmacol. Biochem. Behav., 131, 98–103 (2015).
M. Mahmoodi, S. Shahidi, and P. Hasanein, “Involvement of the ventral tegmental area in the inhibitory avoidance memory in rats,” Physiol. Behav., 102, 542–547 (2011).
S. Shahidi, S. S. Asl, A. Komaki, and N. Hashemi-Firouzi, “The effect of chronic stimulation of serotonin receptor type 7 on recognition, passive avoidance memory, hippocampal long-term potentiation, and neuronal apoptosis in the amyloid beta protein treated rat,” Psychopharmacology, 235, 1513–1525 (2018).
F. Ghahremanitamadon, S. Shahidi, S. Zargooshnia, et al., “Protective effects of Borago officinalis extract on amyloid beta-peptide(25-35)-induced memory impairment in male rats: a behavioral study,” Biomed. Res. Int., 2014, 1–8 (2014).
S. Barzegar, A. Komaki, S. Shahidi, et al., “Effects of cannabinoid and glutamate receptor antagonists and their interactions on learning and memory in male rats,” Pharmacol. Biochem. Behav., 131, 87-90 (2015).
N. Khodamoradi, A. Komaki, I. Salehi, et al., “Effect of vitamin E on lead exposure-induced learning and memory impairment in rats,” Physiol. Behav., 144, 90–94 (2015).
S. Shahidi, S. Setareye, and M. Mahmoodi, “Effect of Prunus domestica L. (mirabelle) on learning and memory in mice,” Anc. Sci. Life., 32, 139–143 (2013).
S. Shahidi, S. Zargooshnia, S. S. Asl, et al., “Influence of N-acetylcysteine on beta-amyloid-induced Alzheimer’s disease in a rat model: A behavioral and electrophysiological study,” Brain Res. Bull., 131, 142–149 (2017).
T. Horisawa, T. Ishibashi, H. Nishikawa, et al., “The effects of selective antagonists of serotonin 5-HT7 and 5-HT1A receptors on MK-801-induced impairment of learning and memory in the passive avoidance and Morris water maze tests in rats: mechanistic implications for the beneficial effects of the novel atypical antipsychotic lurasidone,” Behav. Brain Res., 220, 83–90 (2011).
A. Meneses, “Effects of the 5-HT7 receptor antagonists SB-269970 and DR 4004 in autoshaping Pavlovian/instrumental learning task,” Behav. Brain Res., 155, No. 2, 275–282 (2004).
Y. Shen, F. J. Monsma, M. A. Metcalf, et al., “Molecular cloning and expression of a 5-hydroxytryptamine7 serotonin receptor subtype,” J. Biol. Chem., 268, 18200–18204 (1993).
M. Matsumoto, T. Kojima, H. Togashi, et al., “Differential characteristics of endogenous serotonin-mediated synaptic transmission in the hippocampal CA1 and CA3 fields of anaesthetized rats,” Naunyn Schmiedebergs Arch. Pharmacol., 366, 570–577 (2002).
K. Tokarski, A. Zelek-Molik, B. Duszynska, et al., “Acute and repeated treatment with the 5-HT7 receptor antagonist SB 269970 induces functional desensitization of 5-HT7 receptors in rat hippocampus,” Pharmacol. Rep.., 64, 256–265 (2012).
J. A. Bard, J. Zgombick, N. Adham, et al., “Cloning of a novel human serotonin receptor (5-HT7) positively linked to adenylate cyclase,” J. Biol. Chem., 268, 23422–23426 (1993).
P. B. Hedlund and J. G. Sutcliffe, “Functional, molecular and pharmacological advances in 5-HT7 receptor research,” Trends Pharmacol. Sci., 25, 481–486 (2004).
A. Meneses, “5-HT systems: emergent targets for memory formation and memory alterations,” Rev. Neurosci., 24, 629–664 (2013).
A. J. Roberts, and P. B. Hedlund, “The 5-HT(7) receptor in learning and memory,” Hippocampus, 22, 762–771 (2012).
J. F. Neumaier, T. J. Sexton, J. Yracheta, et al., “Localization of 5-HT(7) receptors in rat brain by immunocytochemistry, in situ hybridization, and agonist stimulated cFos expression,” J. Chem. Neuroanat., 21, 63–73 (2001).
T. F. Freund and G. Buzsaki, “Interneurons of the hippocampus,” Hippocampus, 6, 347–470 (1996).
K. Tokarski, M. Kusek, and G. Hess, “5-HT7 receptors modulate GABAergic transmission in rat hippocampal CA1 area,” J. Physiol. Pharmacol., 62, 535–540 (2011).
K. Tokarski, A. Zahorodna, B. Bobula, and G. Hes, “5-HT7 receptors increase the excitability of rat hippocampal CA1 pyramidal neurons,” Brain Res., 993, 230–234 (2003).
F. Kobe, D. Guseva, T. P. Jensen, et al., “5-HT7R/G12 signaling regulates neuronal morphology and function in an age-dependent manner,” J. Neurosci., 32, 2915–2930 (2012).
N. A. Otmakhova, J. Lewey, B. Asrican, and J. E. Lisman, “Inhibition of perforant path input to the CA1 region by serotonin and noradrenaline,” J. Neurophysiol., 94, 1413–1422 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shahidi, S., Mahmoodi, M. & Sadeghimehr, N. Involvement of Serotonin 5-HT7 Receptors in Learning and Memory in Mice. Neurophysiology 51, 77–82 (2019). https://doi.org/10.1007/s11062-019-09796-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-019-09796-7