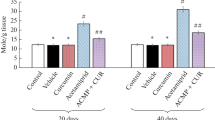
We examined effects of a plant polyphenolic compound, curcumin, against fluoride-induced oxidative stress in the rat brain. Five experimental groups of male rats (10 animals each) were compared. Animals of these experimental groups were treated with curcumin (10 and 20 mg/kg body mass), vitamin C (10 mg/kg), and sample solvent (0.5 ml) for a week prior to sodium fluoride intoxication. After treatment, rats of the experimental groups, except for the normal control group, were intoxicated with sodium fluoride (600 ppm through drinking water) for a week. Then, brains were collected and homogenized, and activities of superoxide dismutase and catalase and levels of reduced glutathione and lipid peroxidation final products were evaluated in the brain tissue homogenates. Treatment with curcumin prior to fluoride intoxication significantly normalized the above biochemical parameters; the intensity of protective effects of 20 mg/kg curcumin was close to that of vitamin C.
Similar content being viewed by others
References
M. A. Ebrahimzadeh, S. F. Nabavi, S. M. Nabavi, et al., “Antihemolytic and antioxidant activities of Allium paradoxum,” Cent. Eur. J. Biol., 5, 338–345 (2010).
M. A. Ebrahimzadeh, S. M. Nabavi, S. F. Nabavi, et al., “Antioxidant and free radical scavenging activities of culinary-medicinal mushrooms, golden chanterelle Cantharellus cibarius and angel’s wings Pleurotus porrigens,” Int. J. Med. Mushrooms, 12, 265–272 (2010).
A. Rajeswari, “Curcumin protects mouse brain from oxidative stress caused by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine,” Eur. Rev. Med. Pharmacol. Sci., 10, 157–161 (2006).
D. Sheril, An Investigation into the Neuroprotective Prospective of Curcumin, Rhodes Univ., Electronic thesis collection TR (2003).
H. Ammon and M. A. Wahl, “Pharmacology of Curcuma longa,” Planta Med., 57, 1–7 (1991).
J. Tilak, M. Banerjee, H. Mohan, et al., “Antioxidant availability of turmeric in relation to its medicinal and culinary uses,” Phytother. Res., 18, 798–804 (2004).
S. Shishodia, G. Sethi, and B. B. Aggarwal, “Curcumin: Getting back to the roots,” Ann. N. Y. Acad. Sci., 1056, 206–217 (2005).
H. Hatcher, R. Planalp, and J. Cho, “Curcumin: From ancient medicine to current clinical trials,” Cell. Mol. Life Sci., 65, 1631–1652 (2008).
J. A. Varner, K. F. Jensen, and W. Horvath, “Chronic administration of aluminum fluoride or sodium fluoride to rats in drinking water: alterations in neuronal and cerebrovascular integrity,” Brain Res., 784, 284–298 (1998).
Y. M. Shivarajashankara, A. R. Shivashankara, and P. G. Bhat, “Effect of fluoride intoxication on lipid peroxidation and antioxidant systems in rats,” Fluoride, 31, 108–113 (2001).
D. Chlubek, “Fluoride and oxidative stress,” Fluoride, 36, 217–228 (2003).
I. Inkielewicz and W. Czanowski, “Oxidative stress parameters in rats exposed to fluoride and aspirin,” Fluoride, 41, 76–82 (2008).
M. Bhatnagar, P. Rao, and J. Sushma, “Neurotoxicity of fluoride: neurodegeneration in hippocampus of female mice,” Ind. J. Exp. Biol., 40, 546–554 (2002).
B. Halliwell and J. M. Gutteridge, “Oxygen toxicity, oxygen radicals, transition metals and disease,” Biochem. J., 219, 1–14 (1984).
A. R. Shivashankara, Y. M. Shivarajashankara, P. G. Bhat, et al., “Lipid peroxidation and antioxidant defense systems in liver of rats in chronic fluoride toxicity,” Bull. Environ. Cont. Toxicol., 68, 612–616 (2002).
M. A. Ebrahimzadeh, S. F. Nabavi, S. M. Nabavi, et al., “In vitro antioxidant and antihemolytic activities of hydroalcoholic extracts of Allium scabriscapum Boiss. & Ky. aerial parts and bulbs,” Int. J. Food Prop . (accepted) (2011).
M. Sinha, P. Manna, and P. C. Sil, “A 43 kD protein from the herb, Cajanus indicus L., protects against fluoride-induced oxidative stress in mice erythrocytes,” Pathophysiology, 14, 47–54 (2007).
M. M. Bradford, “A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding,” Anal. Biochem., 72, 248–254 (1976).
H. Esterbauer and K. H. Cheeseman, “Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal,” Methods Enzymol., 186, 407–421 (1990).
H. P. Misra and I. Fridovich, “The role of superoxide anion in the autooxidation of epinephrine and simple assay for superoxide dismutase,” J. Biol. Chem., 247, 3170–3175 (1972).
J. Bonaventura, W. A. Schroeder, and S. Fang, “Human erythrocyte catalase: an improved method of isolation and a reevaluation of reported properties,” Arch. Biochem. Biophys., 150, 606–617 (1972).
G. L. Ellman, “Tissue sulfhydryl group,” Arch. Biochem. Biophys., 82, 70–77 (1959).
B. M. Babior, “The respiratory burst oxidase,” Adv. Enzymol. Rel. Area Mol. Biol., 65, 49–65 (1992).
J. M. Mates, C. P. Gomez, and C. Nunez, “Antioxidant enzymes and human diseases,” Clin. Biochem., 32, 595–603 (1999).
S. I. Yamagishi, D. Edelstein, X. L. Du, et al., “Hyperglycaemia potentiates collagen-induced platelet activation through mitochondrial superoxide overproduction,” Diabetes, 50, 1491–1494 (2001).
I. Mazzetti, B. Grigolo, R. M. Borzi, et al., “Serum copper, zinc superoxide dismutase level in patients with rheumatoid arthritis,” Int. J. Clin. Lab. Res., 26, 245–249 (1996).
H. Bartsch and J. Nair, “Ultrasensitive and specific detection method for exocyclic DNA adducts: markers for lipid peroxidation and oxidative stress,” Toxicology, 153, 105–114 (2000).
B. Halliwell and J. M. C. Guttteridge, Free Radicals in Biology and Medicine, Oxford Univ. Press (Clarendon), Oxford (2007).
M. Gul, F.Z. Kutay, S. Temocin, et al., “Cellular and clinical implication of glutathione,” Ind. J. Exp. Biol., 38, 625–634 (2000).
S. A. Sheweita, M. Abd El-Gabar, and M. Bastawy, “Carbon tetrachloride induced changes in the activity of phase II drug metabolizing enzyme in the liver of male rats: role of antioxidants,” Toxicology, 165, 217–224 (2001).
S. L. Liu, S. Degli Esposti, T. Yao, et al., “Vitamin E therapy of acute CCl4 induced hepatic injury in mice is assisted with inhibition of nuclear factor kappa B binding,” Hepatology, 22, 1474–1481 (1995).
Author information
Authors and Affiliations
Corresponding author
Additional information
Neirofiziologiya/Neurophysiology, Vol. 43, No. 4, pp. 332–336, July–August, 2011.
Rights and permissions
About this article
Cite this article
Nabavi, S.F., Eslami, S., Moghaddam, A.H. et al. Protective Effects of Curcumin against Fluoride-Induced Oxidative Stress in the Rat Brain. Neurophysiology 43, 287–291 (2011). https://doi.org/10.1007/s11062-011-9228-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-011-9228-y