Abstract
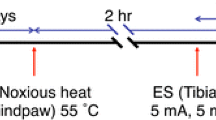
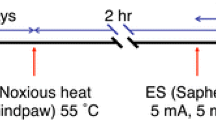
Results of electrophysiological studies suggest a significant role of the lateral spinal nucleus (LSpN) in the transmission of nociceptive signals. In our study, the presence of Fos immunoreactivity and NADPH-diaphorase positivity was observed in the rat LSpN following noxious peripheral subcutaneous stimulation. Formalin-induced unilateral hindpaw stimulation in the rat caused bilateral NADPH-d reactivity and ipsilateral Fos expression in this nucleus. In the LSpN of the L3–L5 segments of stimulated rats, on average, 4.1 ± 1.2 NADPH-d-positive, NADPH-d(+), 5.1+1.8 Fos-immunoreactive, Fos(+), and 3.0 ± 1.1 double-labeled neurons per 25-µm-thick section were found unilaterally. A close anatomical relationship between NADPH-d(+) processes and Fos(+) cell nuclei in the LSpN was also observed following noxious peripheral stimulation. These neuroanatomical findings support the hypothesis that the LSpN is involved in pain processing and suggest an important role of nitric oxide-mediated signal transduction in this nucleus.
Similar content being viewed by others
References
C. Molander and G. Grant, “Spinal cord cytoarchitecture,” in: The Rat Nervous System, G. Paxinos (ed.), Elsevier Academic Press, San Diego (2004), p. 126.
D._G. Gwyn and A. H. Waldron, “A nucleus in the dorsal lateral funiculus of the spinal cord of the rat,” Brain Res., 10, No. 3, 342–351 (1968).
M. C. Jiang, L. Liu, and G. F. Gebhart, “Cellular properties of lateral spinal nucleus neurons in the rat L6-S1 spinal cord,” J. Neurophysiol., 81, No. 6, 3078–3086 (1999).
A. S. Jansen and A. D. Loewy, “Neurons lying in the white matter of the upper cervical spinal cord project to the intermediolateral cell column,” Neuroscience, 77, No. 3, 889–898 (1997).
K. D. Cliffer, G. Urca, R. P. Elde, and G. J. Giesler, Jr., “Studies of peptidergic input to the lateral spinal nucleus,” Brain Res., 460, No. 2, 356–360 (1988).
G. Battaglia and A. Rustioni, “Substance P innervation of the rat and cat thalamus. II. Cells of origin in the spinal cord,” J. Comp. Neurol., 315, No. 4, 473–486 (1992).
X. Zhang, R. R. Ji, S. Nilsson, et al., “Neuropeptide Y and galanin-binding sites in rat and monkey lumbar dorsal root ganglia and spinal cord and effect of peripheral axotomy,” Eur. J. Neurosci., 7, No. 3, 367–380 (1995).
M. Conrath, H. Taquet, M. Pohl, et al., “Immunocytochemical evidence for calcitonin gene-related peptide-like neurons in the dorsal horn and lateral spinal nucleus of the rat cervical spinal cord,” J. Chem. Neuroanat., 2, No. 6, 335–347 (1989).
J. Leah, D. Menetrey, and J. De Pommery, “Neuropeptides in long ascending spinal tract cells in the rat: evidence for parallel processing of ascending information,” Neuroscience, 24, No. 1, 195–207 (1988).
D. Menetrey, A. Chaouch, D. Binder, et al., “The origin of the spinomesencephalic tract in the rat: an anatomical study using the retrograde transport of horseradish peroxidase,” J. Comp. Neurol., 206, No. 2, 862–867 (1982).
G. Kayalioglu, B. Robertson, K. Kristensson, et al., “Nitric oxide synthase and interferon-γ receptor immunoreactivities in relation to ascending spinal pathways to thalamus, hypothalamus, and the periaqueductal grey in the rat,” Somatosens. Mot. Res., 16, No. 4, 280–290 (1999).
R. Burstein, K. D. Cliffer, and G. J. Giesler, “Direct somatosensory projections from the spinal cord to the hypothalamus and telencephalon,” J. Neurosci., 7, No. 12, 4159–4164 (1987).
R. Burstein, R. J. Dado, and G. J. Giesler, Jr., “The cells of origin of the spinothalamic tract of the rat: a quantitative reexamination,” Brain Res., 511, No. 2, 329–337 (1990).
W. D. Willis, K. N. Westlund, and S. M. Carlton, “Pain,” in: The Rat Nervous System, G. Paxinos (ed.), Elsevier Academic Press, San Diego (2004), pp. 864–867.
D. Menetrey, A. Chaouch, and J. M. Besson, “Location and properties of dorsal horn neurons at origin of spinoreticular tract in lumbar enlargement of the rat,” J. Neurophysiol., 44, No. 5, 862–877 (1980).
E. Bullitt, “Induction of c-Fos-like protein within the lumbar spinal cord and thalamus of the rat following peripheral stimulation,” Brain Res., 493, No. 2, 391–397 (1989).
T. Herdegen, K. Kovary, J. Leah, et al., “Specific temporal and spatial distribution of JUN, FOS, and KROX-24 proteins in spinal neurons following noxious transsynaptic stimulation,” J. Comp. Neurol., 313, No. 1, 178–191 (1991).
S. P. Hunt, A. Pini, and G. Evan, “Induction of c-Fos-like protein in spinal cord neurons following sensory stimulation,” Nature, 328, No. 6131, 632–634 (1987).
J. A. Harris, “Using c-fos as a neuronal marker of pain,” Brain Res. Bull., 45, No. 1, 1–8 (1998).
A. B. Malmberg and T. L. Yaksh, “Spinal nitric oxide synthesis inhibition blocks NMDA-induced thermal hyperalgesia and produces antinociception in the formalin test in rats,” Pain, 54, No. 3, 291–300 (1993).
S. T. Meller and G. F. Gebhart, “Nitric oxide (NO) and nociceptive processing in the spinal cord,” Pain, 52, No. 2, 127–136 (1993).
B. T. Hope, G. J. Michael, K. M. Knigge, et al., “Neuronal NADPH diaphorase is a nitric oxide synthase,” Proc. Natl. Acad. Sci. USA, 88, No. 7, 2811–2814 (1991).
R. J. Traub, A. Solodkin, and G. F. Gebhart, “NADPH-diaphorase histochemistry provides evidence for a bilateral, somatotopically inappropriate response to unilateral hindpaw inflammation in the rat,” Brain Res., 647, No. 1, 113–123 (1994).
M. Zimmermann, “Ethical guidelines for investigations of experimental pain in conscious animals,” Pain, 16, No. 2, 109–110 (1983).
C. Molander, Q. Xu, and G. Grant, “The cytoarchitectonic organization of the spinal cord in the rat. I. The lower thoracic and lumbosacral cord,” J. Comp. Neurol., 230, No. 1, 133–141 (1984).
D. Menetrey, A. Gannon, J. D. Levine, et al., “Expression of c-Fos protein in interneurons and projection neurons of the rat spinal cord in response to noxious somatic, articular, and visceral stimulation,” J. Comp. Neurol., 285, No. 2, 177–195 (1989).
G. E. Marshall, S. A. S. Shehab, R. C. Spike, et al., “Neurokinin-1 receptors on lumbar spinothalamic neurons in the rat,” Neuroscience, 72, No. 1, 255–263 (1996).
J. L. Li, T. Kaneko, R. Shigemoto, et al., “Distribution of trigeminohypothalamic and spinohypothalamic tract neurons displaying substance P receptor-like immunoreactivity in the rat,” J. Comp. Neurol., 378, No. 4, 508–521 (1997).
J. L. Li, Y.Q. Ding, K. H. Xiong, et al., “Substance P receptor (NK-1)-immunoreactive neurons projecting to the periaqueductal gray: distribution in the spinal trigeminal nucleus and the spinal cord of the rat,” Neurosci. Res., 30, No. 3, 219–225 (1998).
R. Benoliel, M. Tanaka, R. M. Caudle, et al., “Co-localization of N-methyl-D-aspartate receptors and substance P (neurokinin-1) receptors in rat spinal cord,” Neurosci. Lett., 291, No. 2, 61–64 (2000).
K. I. Rusin, D. Bleakman, P. S. Chard, et al., “Tachykinins potentiate N-methyl-d-aspartate responses in acutely isolated neurons from the dorsal horn,” J. Neurochem., 60, No. 3, 952–960 (1993).
K. Vikman, B. Robertson, A. Liljeborg, et al., “Interferon-γ receptors are expressed at synapses in the rat superficial dorsal horn and lateral spinal nucleus,” J. Neurocytol., 27, No. 10, 749–760 (1998).
T. Herdegen, S. Rüdiger, B. Mayer, et al., “Expression of nitric oxide synthase and colocalisation with Jun, Fos and Krox transcription factors in spinal cord neurons following noxious stimulation of the rat hindpaw,” Mol. Brain Res., 22, Nos. 1/4, 245–258 (1994).
M. Salter, P. J. Strijbos, S. Neale, et al., “The nitric oxide-cyclic GMP pathway is required for nociceptive signalling at specific loci within the somatosensory pathway,” Neuroscience, 73, No. 3, 649–655 (1996).
J. H. Park, V. A. Straub, and M. O’Shea, “Anterograde signalling by nitric oxide: characterization and in vitro reconstitution of an identified nitrergic synapse,” J. Neurosci., 18, No. 14, 5463–5476 (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Neirofiziologiya/Neurophysiology, Vol. 40, No. 1, pp. 38–42, January–February, 2008.
Rights and permissions
About this article
Cite this article
Dagci, T., Okur, B.E. & Kayalioglu, G. Lateral spinal nucleus of the rat: NADPH-diaphorase activity and Fos expression after noxious peripheral stimulation. Neurophysiology 40, 34–38 (2008). https://doi.org/10.1007/s11062-008-9018-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-008-9018-3