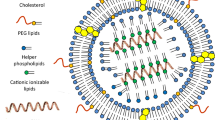
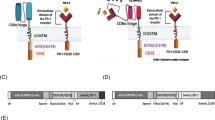
Abstract
Purpose
Exosomes are nano-vesicular carriers capable of delivering cargoes for intercellular communication, which holds potential as biocompatible and high efficiency systems for drug delivery. In this study, we evaluated the potential effect of T7 peptide-decorated exosome-loaded Galectin-9 siRNA (T7-Exo/siGalectin-9) in the M1 polarization of macrophages and immunosuppression of glioblastoma (GBM).
Methods
Differentially expressed genes in GBM were in silico predicted and then experimentally verified. Galectin-9 was knocked down by siRNA to assess its role in tumor-bearing mice. T7 peptide-decorated exosomes (derived from human embryonic kidney [HEK]-293T cells) targeting GBM were prepared, and loaded with Galectin-9 siRNA by electroporation to prepare nanoformulations (T7-Exo/siGalectin-9). The role of T7-Exo/siGalectin-9 in CD8+ T cell cytotoxicity to target GBM cells and polarization of macrophages was evaluated after artificial modulation of Galectin-9 expression. Anti-tumor effects of T7-Exo/siGalectin-9 were elucidated in vitro and in vivo.
Results
Galectin-9 was highly expressed in GBM tissues and cell lines. The siRNA-mediated knockdown of Galectin-9 repressed the growth of xenografts of GBM cells in C57BL/6 mice and activated immune response in the tumor microenvironment. T7-Exo/siGalectin-9 effectively delivered siGalectin-9 to GBM cells. T7-Exo/siGalectin-9 contributed to activation of the TLR7-IRF5 pathway, which polarized macrophages to M1 phenotype. By this mechanism, phagocytosis of GBM cells by macrophages was increased, the anti-tumor effect of CD8+ T cells was enhanced and the inflammatory responses were suppressed.
Conclusion
Overall, T7-Exo/siGalectin-9 promotes macrophage repolarization and restricts the immunosuppression of GBM, thus providing novel insights into and drug delivery system of immunotherapy for GBM.
Graphical abstract






Similar content being viewed by others
Data availability
The datasets generated/analyzed during the current study are available.
References
Omuro A, DeAngelis LM (2013) Glioblastoma and other malignant gliomas: a clinical review. JAMA 310:1842–1850. https://doi.org/10.1001/jama.2013.280319
Lah TT, Novak M, Breznik B (2020) Brain malignancies: Glioblastoma and brain metastases. Semin Cancer Biol 60:262–273. https://doi.org/10.1016/j.semcancer.2019.10.010
Venkataramani V, Yang Y, Schubert MC, Reyhan E, Tetzlaff SK, Wissmann N, Botz M, Soyka SJ, Beretta CA, Pramatarov RL, Fankhauser L, Garofano L, Freudenberg A, Wagner J, Tanev DI, Ratliff M, Xie R, Kessler T, Hoffmann DC, Hai L, Dorflinger Y, Hoppe S, Yabo YA, Golebiewska A, Niclou SP, Sahm F, Lasorella A, Slowik M, Doring L, Iavarone A, Wick W, Kuner T, Winkler F (2022) Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell 185:2899–2917e2831. https://doi.org/10.1016/j.cell.2022.06.054
Le Rhun E, Preusser M, Roth P, Reardon DA, van den Bent M, Wen P, Reifenberger G, Weller M (2019) Molecular targeted therapy of glioblastoma. Cancer Treat Rev 80:101896. https://doi.org/10.1016/j.ctrv.2019.101896
Campos B, Olsen LR, Urup T, Poulsen HS (2016) A comprehensive profile of recurrent glioblastoma. Oncogene 35:5819–5825. https://doi.org/10.1038/onc.2016.85
Ma R, Taphoorn MJB, Plaha P (2021) Advances in the management of glioblastoma. J Neurol Neurosurg Psychiatry 92:1103–1111. https://doi.org/10.1136/jnnp-2020-325334
Hernandez A, Domenech M, Munoz-Marmol AM, Carrato C, Balana C (2021) Glioblastoma: relationship between metabolism and immunosuppressive microenvironment. Cells 10:3529. https://doi.org/10.3390/cells10123529
Nduom EK, Weller M, Heimberger AB (2015) Immunosuppressive mechanisms in glioblastoma. Neuro Oncol 17 Suppl 7:vii9–vii14. https://doi.org/10.1093/neuonc/nov151
Cui X, Morales RT, Qian W, Wang H, Gagner JP, Dolgalev I, Placantonakis D, Zagzag D, Cimmino L, Snuderl M, Lam RHW, Chen W (2018) Hacking macrophage-associated immunosuppression for regulating glioblastoma angiogenesis. Biomaterials 161:164–178. https://doi.org/10.1016/j.biomaterials.2018.01.053
Desland FA, Hormigo A (2020) The CNS and the brain tumor microenvironment: implications for glioblastoma immunotherapy. Int J Mol Sci 21:7358. https://doi.org/10.3390/ijms21197358
Kim G, Kim M, Lee Y, Byun JW, Hwang DW, Lee M (2020) Systemic delivery of microRNA-21 antisense oligonucleotides to the brain using T7-peptide decorated exosomes. J Control Release 317:273–281. https://doi.org/10.1016/j.jconrel.2019.11.009
Liu X, Cao Z, Liu N, Gao G, Du M, Wang Y, Cheng B, Zhu M, Jia B, Pan L, Zhang W, Jiang Y, He W, Xu L, Zhang W, An Q, Guo Q, Gu J (2022) Kill two birds with one stone: Engineered exosome-mediated delivery of cholesterol modified YY1-siRNA enhances chemoradiotherapy sensitivity of glioblastoma. Front Pharmacol 13:975291. https://doi.org/10.3389/fphar.2022.975291
Zhang Y, Zhai M, Chen Z, Han X, Yu F, Li Z, Xie X, Han C, Yu L, Yang Y, Mei X (2017) Dual-modified liposome codelivery of doxorubicin and vincristine improve targeting and therapeutic efficacy of glioma. Drug Deliv 24:1045–1055. https://doi.org/10.1080/10717544.2017.1344334
Zhou W, Zhou Y, Chen X, Ning T, Chen H, Guo Q, Zhang Y, Liu P, Zhang Y, Li C, Chu Y, Sun T, Jiang C (2021) Pancreatic cancer-targeting exosomes for enhancing immunotherapy and reprogramming tumor microenvironment. Biomaterials 268:120546. https://doi.org/10.1016/j.biomaterials.2020.120546
Yuan F, Ming H, Wang Y, Yang Y, Yi L, Li T, Ma H, Tong L, Zhang L, Liu P, Li J, Lin Y, Yu S, Ren B, Yang X (2020) Molecular and clinical characterization of Galectin-9 in glioma through 1,027 samples. J Cell Physiol 235:4326–4334. https://doi.org/10.1002/jcp.29309
Zhang F, Parayath NN, Ene CI, Stephan SB, Koehne AL, Coon ME, Holland EC, Stephan MT (2019) Genetic programming of macrophages to perform anti-tumor functions using targeted mRNA nanocarriers. Nat Commun 10:3974. https://doi.org/10.1038/s41467-019-11911-5
Panda SK, Facchinetti V, Voynova E, Hanabuchi S, Karnell JL, Hanna RN, Kolbeck R, Sanjuan MA, Ettinger R, Liu YJ (2018) Galectin-9 inhibits TLR7-mediated autoimmunity in murine lupus models. J Clin Invest 128:1873–1887. https://doi.org/10.1172/JCI97333
Han Z, Liu S, Lin H, Trivett AL, Hannifin S, Yang, Oppenheim JJ (2019) Inhibition of murine hepatoma tumor growth by cryptotanshinone involves TLR7-dependent activation of macrophages and induction of adaptive antitumor immune defenses. Cancer Immunol Immunother 68:1073–1085. https://doi.org/10.1007/s00262-019-02338-4
Stathopoulos A, Pretto C, Devillers L, Pierre D, Hofman FM, Kruse C, Jadus M, Chen TC, Schijns VE (2012) Development of immune memory to glial brain tumors after tumor regression induced by immunotherapeutic toll-like receptor 7/8 activation. Oncoimmunology 1:298–305. https://doi.org/10.4161/onci.19068
Fu B, Zhao M, Wang L, Patil G, Smith JA, Juncadella IJ, Zuvela-Jelaska L, Dorf ME, Li S (2017) RNAi screen and proteomics reveal NXF1 as a novel regulator of IRF5 signaling. Sci Rep 7:2683. https://doi.org/10.1038/s41598-017-02857-z
Biray Avci C, Kurt CC, Tepedelen BE, Ozalp O, Goker B, Mutlu Z, Dodurga Y, Elmas L, Gunduz C (2016) Zoledronic acid induces apoptosis via stimulating the expressions of ERN1, TLR2, and IRF5 genes in glioma cells. Tumour Biol 37:6673–6679. https://doi.org/10.1007/s13277-015-4519-3
Zhou J, Li X, Wu X, Zhang T, Zhu Q, Wang X, Wang H, Wang K, Lin Y, Wang X (2018) Exosomes released from tumor-associated macrophages transfer miRNAs that induce a Treg/Th17 cell imbalance in epithelial ovarian cancer. Cancer Immunol Res 6:1578–1592. https://doi.org/10.1158/2326-6066.CIR-17-0479
Kooijmans SAA, Stremersch S, Braeckmans K, de Smedt SC, Hendrix A, Wood MJA, Schiffelers RM, Raemdonck K, Vader P (2013) Electroporation-induced siRNA precipitation obscures the efficiency of siRNA loading into extracellular vesicles. J Control Release 172:229–238. https://doi.org/10.1016/j.jconrel.2013.08.014
Li HY, He HC, Song JF, Du YF, Guan M, Wu CY (2020) Bone marrow-derived mesenchymal stem cells repair severe acute pancreatitis by secreting miR-181a-5p to target PTEN/Akt/TGF-beta1 signaling. Cell Signal 66:109436. https://doi.org/10.1016/j.cellsig.2019.109436
Wang M, Cai Y, Peng Y, Xu B, Hui W, Jiang Y (2020) Exosomal LGALS9 in the cerebrospinal fluid of glioblastoma patients suppressed dendritic cell antigen presentation and cytotoxic T-cell immunity. Cell Death Dis 11:896. https://doi.org/10.1038/s41419-020-03042-3
Fabie A, Mai LT, Dagenais-Lussier X, Hammami A, van Grevenynghe J, Stager S (2018) IRF-5 promotes cell death in CD4 T cells during chronic infection. Cell Rep 24:1163–1175. https://doi.org/10.1016/j.celrep.2018.06.107
Johnston WF, Salmon M, Su G, Lu G, Stone ML, Zhao Y, Owens GK, Upchurch GR Jr, Ailawadi G (2013) Genetic and pharmacologic disruption of interleukin-1beta signaling inhibits experimental aortic aneurysm formation. Arterioscler Thromb Vasc Biol 33:294–304. https://doi.org/10.1161/ATVBAHA.112.300432
Xiong W, MacTaggart J, Knispel R, Worth J, Persidsky Y, Baxter BT (2009) Blocking TNF-alpha attenuates aneurysm formation in a murine model. J Immunol 183:2741–2746. https://doi.org/10.4049/jimmunol.0803164
Batra R, Suh MK, Carson JS, Dale MA, Meisinger TM, Fitzgerald M, Opperman PJ, Luo J, Pipinos II, Xiong W, Baxter BT (2018) IL-1beta (Interleukin-1beta) and TNF-alpha (tumor necrosis Factor-alpha) impact abdominal aortic aneurysm formation by differential effects on macrophage polarization. Arterioscler Thromb Vasc Biol 38:457–463. https://doi.org/10.1161/ATVBAHA.117.310333
Hume DA (2006) The mononuclear phagocyte system. Curr Opin Immunol 18:49–53. https://doi.org/10.1016/j.coi.2005.11.008
McKenzie MD, Dudek NL, Mariana L, Chong MM, Trapani JA, Kay TW, Thomas HE (2006) Perforin and Fas induced by IFNgamma and TNFalpha mediate beta cell death by OT-I CTL. Int Immunol 18:837–846. https://doi.org/10.1093/intimm/dxl020
Kalia V, Sarkar S (2018) Regulation of effector and memory CD8 T cell differentiation by IL-2-A balancing act. Front Immunol 9:2987. https://doi.org/10.3389/fimmu.2018.02987
Smith KA (1988) Interleukin-2: inception, impact, and implications. Science 240:1169–1176. https://doi.org/10.1126/science.3131876
Cheng LE, Ohlen C, Nelson BH, Greenberg PD (2002) Enhanced signaling through the IL-2 receptor in CD8+ T cells regulated by antigen recognition results in preferential proliferation and expansion of responding CD8+ T cells rather than promotion of cell death. Proc Natl Acad Sci U S A 99:3001–3006. https://doi.org/10.1073/pnas.052676899
Luu M, Riester Z, Baldrich A, Reichardt N, Yuille S, Busetti A, Klein M, Wempe A, Leister H, Raifer H, Picard F, Muhammad K, Ohl K, Romero R, Fischer F, Bauer CA, Huber M, Gress TM, Lauth M, Danhof S, Bopp T, Nerreter T, Mulder IE, Steinhoff U, Hudecek M, Visekruna A (2021) Microbial short-chain fatty acids modulate CD8(+) T cell responses and improve adoptive immunotherapy for cancer. Nat Commun 12:4077. https://doi.org/10.1038/s41467-021-24331-1
Liang T, Wang X, Wang F, Feng E, You G (2019) Galectin-9: a predictive biomarker negatively regulating Immune response in glioma patients. World Neurosurg 132:e455–e462. https://doi.org/10.1016/j.wneu.2019.08.117
Knudsen AM, Rudkjobing SJ, Sorensen MD, Dahlrot RH, Kristensen BW (2021) Expression and prognostic value of the immune checkpoints Galectin-9 and PD-L1 in glioblastomas. J Neuropathol Exp Neurol 80:541–551. https://doi.org/10.1093/jnen/nlab041
Querol Cano L, Tagit O, Dolen Y, van Duffelen A, Dieltjes S, Buschow SI, Niki T, Hirashima M, Joosten B, van den Dries K, Cambi A, Figdor CG, van Spriel AB (2019) Intracellular Galectin-9 controls dendritic cell function by maintaining plasma. Memb Rigid iSci 22:240–255. https://doi.org/10.1016/j.isci.2019.11.019
Ma Y, Shurin GV, Gutkin DW, Shurin MR (2012) Tumor associated regulatory dendritic cells. Semin Cancer Biol 22:298–306. https://doi.org/10.1016/j.semcancer.2012.02.010
Zhu S, Yang N, Wu J, Wang X, Wang W, Liu YJ, Chen J (2020) Tumor microenvironment-related dendritic cell deficiency: a target to enhance tumor immunotherapy. Pharmacol Res 159:104980. https://doi.org/10.1016/j.phrs.2020.104980
Zhang Y, Bi J, Huang J, Tang Y, Du S, Li P (2020) Exosome: a review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int J Nanomed 15:6917–6934. https://doi.org/10.2147/IJN.S264498
Ghoreishy A, Khosravi A, Ghaemmaghami A (2019) Exosomal microRNA and stroke: a review. J Cell Biochem 120:16352–16361. https://doi.org/10.1002/jcb.29130
Liang M, Gao C, Wang Y, Gong W, Fu S, Cui L, Zhou Z, Chu X, Zhang Y, Liu Q, Zhao X, Zhao B, Yang M, Li Z, Yang C, Xie X, Yang Y, Gao C (2018) Enhanced blood-brain barrier penetration and glioma therapy mediated by T7 peptide-modified low-density lipoprotein particles. Drug Deliv 25:1652–1663. https://doi.org/10.1080/10717544.2018.1494223
He GZ, Lin WJ (2021) Peptide-functionalized nanoparticles-encapsulated cyclin-dependent kinases inhibitor seliciclib in transferrin receptor overexpressed Cancer cells. Nanomaterials (Basel) 11:772. https://doi.org/10.3390/nano11030772
Lv R, Bao Q, Li Y (2017) Regulation of M1type and M2type macrophage polarization in RAW264.7 cells by Galectin9. Mol Med Rep 16:9111–9119. https://doi.org/10.3892/mmr.2017.7719
Zhang M, Hutter G, Kahn SA, Azad TD, Gholamin S, Xu CY, Liu J, Achrol AS, Richard C, Sommerkamp P, Schoen MK, McCracken MN, Majeti R, Weissman I, Mitra SS, Cheshier SH (2016) Anti-CD47 treatment stimulates phagocytosis of Glioblastoma by M1 and M2 polarized Macrophages and promotes M1 polarized macrophages in vivo. PLoS ONE 11:e0153550. https://doi.org/10.1371/journal.pone.0153550
Kurachi M (2019) CD8(+) T cell exhaustion. Semin Immunopathol 41:327–337. https://doi.org/10.1007/s00281-019-00744-5
Walter U, Santamaria P (2005) CD8+ T cells in autoimmunity. Curr Opin Immunol 17:624–631. https://doi.org/10.1016/j.coi.2005.09.014
Daley D, Mani VR, Mohan N, Akkad N, Ochi A, Heindel DW, Lee KB, Zambirinis CP, Pandian GSB, Savadkar S, Torres-Hernandez A, Nayak S, Wang D, Hundeyin M, Diskin B, Aykut B, Werba G, Barilla RM, Rodriguez R, Chang S, Gardner L, Mahal LK, Ueberheide B, Miller G (2017) Dectin 1 activation on macrophages by galectin 9 promotes pancreatic carcinoma and peritumoral immune tolerance. Nat Med 23:556–567. https://doi.org/10.1038/nm.4314
Rodell CB, Arlauckas SP, Cuccarese MF, Garris CS, Li R, Ahmed MS, Kohler RH, Pittet MJ, Weissleder R (2018) TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat Biomed Eng 2:578–588. https://doi.org/10.1038/s41551-018-0236-8
Liu Z, Xie Y, Xiong Y, Liu S, Qiu C, Zhu Z, Mao H, Yu M, Wang X (2020) TLR 7/8 agonist reverses oxaliplatin resistance in colorectal cancer via directing the myeloid-derived suppressor cells to tumoricidal M1-macrophages. Cancer Lett 469:173–185. https://doi.org/10.1016/j.canlet.2019.10.020
Chen PM, Pan WY, Wu CY, Yeh CY, Korupalli C, Luo PK, Chou CJ, Chia WT, Sung HW (2020) Modulation of tumor microenvironment using a TLR-7/8 agonist-loaded nanoparticle system that exerts low-temperature hyperthermia and immunotherapy for in situ cancer vaccination. Biomaterials 230:119629. https://doi.org/10.1016/j.biomaterials.2019.119629
Okazaki S, Stintzing S, Sunakawa Y, Cao S, Zhang W, Yang D, Ning Y, Matsusaka S, Berger MD, Miyamoto Y, Suenaga M, Schirripa M, West JD, Gopez R, Akihito T, Ichikawa W, Heinemann V, DePaolo RW, Lenz HJ (2017) Predictive value of TLR7 polymorphism for cetuximab-based chemotherapy in patients with metastatic colorectal cancer. Int J Cancer 141:1222–1230. https://doi.org/10.1002/ijc.30810
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
CL, NG and FL designed the study. CL, NG and FL collated the data, designed and developed the database, carried out data analyses and produced the initial draft of the manuscript. CL, NG and FL contributed to drafting the manuscript. All authors have read and approved the final submitted manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflict of interests to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
11060_2023_4257_MOESM1_ESM.eps
Figure S1 Flow cytometric analysis of macrophage polarization in TILs (A, for panel 1I), IFN-γ+ CD8+ T cell percentage in TILs (B, for panel 1J), and IFN-γ+ CD8+ T cells treated with Galectin-9 alone or combined with oe-IRF5/oe-TLR7 (C, for panel 5C) (EPS 2444 KB)
11060_2023_4257_MOESM2_ESM.eps
Figure S2 Detection of the effect of electroporation on exosomes. A, The protein expression of CD63, CD9, and CD81 in T7-Exos and T7-Exo/siGalectin-9 detected by Western blot analysis. B, Observation of T7-Exos and T7-Exo/siGalectin-9 by TEM. C, The size of T7-Exos and T7-Exo/siGalectin-9 (EPS 7485 KB)
11060_2023_4257_MOESM3_ESM.eps
Figure S3 Flow cytometric analysis of CD68+ cells in THP-1 cells stimulated by PMA (A, for panel 4D), as well as CD11b+, CD206+, CD11b+ and CD80+ cells (B and C, for panel 4G) and CFSE+ eFluor®670+ cells (D, for panel 4I) after Galectin-9 supplement alone or combined with oe-IRF5/oe-TLR7 (EPS 3567 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, C., Guan, N. & Liu, F. T7 peptide-decorated exosome-based nanocarrier system for delivery of Galectin-9 siRNA to stimulate macrophage repolarization in glioblastoma. J Neurooncol 162, 93–108 (2023). https://doi.org/10.1007/s11060-023-04257-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04257-y