Abstract
Purpose
Pilocytic astrocytoma (PA) is a circumscribed low-grade astrocytic glioma, generally considered to be associated with a good prognosis. However, a subset of PA patients shows unfavorable outcomes. In this study, we retrospectively reviewed PA patients and performed further molecular analysis, such as DNA methylation profiling, to identify prognostic factors.
Methods
We analyzed 29 histologically-confirmed PA patients from a single center from 2002 to 2021 and conducted integrated molecular analyses among elderly PA patients since age was an independent prognostic factor for poor outcomes.
Results
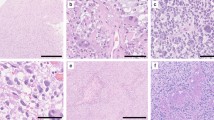
The median age at diagnosis was 14 years (range 3–82 years) and 4 patients (14%) were elderly (patients ≥ 60 years old). Age over 60 was associated with poor progression-free survival and overall survival. We performed DNA methylation analysis on 2 of the 4 elderly patients. Both cases were histologically diagnosed as PA, but DNA methylation profiling revealed one as high-grade astrocytoma with piloid features (all methylation class scores were below 0.3 in both v11b4 and v12.5) and the other as glioblastoma, IDH-wildtype (score was over 0.5 in both v11b4 and v12.5), using the German Cancer Research Center methylation profiling classifiers and t-SNE analysis.
Conclusions
Elderly patients with PA morphology showed unfavorable outcomes in this cohort. In those patients, further molecular analysis and DNA methylation profiling revealed the possibility of high-grade astrocytic tumors, including newly defined entities.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- PA:
-
Pilocytic astrocytoma
- MC AAP:
-
Methylation-class anaplastic astrocytoma with piloid features
- HGAP:
-
High-grade astrocytoma with piloid features
- GTR:
-
Gross total resection
- STR:
-
Subtotal resection
- PR:
-
Partial resection
- PFS:
-
Progression-free survival
- OS:
-
Overall survival
- HE:
-
Hematoxylin and eosin
- IHC:
-
Immunohistochemistry
- t-SNE:
-
T-distributed stochastic neighbor embedding
- KPS:
-
Karnofsky Performance Score
- NF1:
-
Neurofibromatosis 1
- SEER:
-
Surveillance Epidemiology and End Results
- AA:
-
Anaplastic astrocytoma
References
Ostrom QT, Cioffi G, Waite K, et al. (2021) CBTRUS Statistical Report: primary brain and other central nervous system tumors diagnosed in the United States in 2014–2018. Neuro Oncol 23: iii1-iii105. https://doi.org/10.1093/neuonc/noab200
Board WCoTE (2021) Central nervous system tumours. International Agency for Research on Cancer, Lyon
Tomita Y, Hibler EA, Suruga Y et al (2022) Age is a major determinant for poor prognosis in patients with pilocytic astrocytoma: a SEER population study. Clin Exp Med. https://doi.org/10.1007/s10238-022-00882-5
Dodgshun AJ, Maixner WJ, Hansford JR et al (2016) Low rates of recurrence and slow progression of pediatric pilocytic astrocytoma after gross-total resection: justification for reducing surveillance imaging. J Neurosurg Pediatr 17:569–572. https://doi.org/10.3171/2015.9.Peds15449
Jungk C, Reinhardt A, Warta R et al (2019) Extent of resection, MGMT promoter methylation status and tumor location independently predict progression-free survival in adult sporadic pilocytic astrocytoma. Cancers (Basel). https://doi.org/10.3390/cancers11081072
Nelson AJ, Zakaria R, Jenkinson MD et al (2019) Extent of resection predicts risk of progression in adult pilocytic astrocytoma. Br J Neurosurg 33:343–347. https://doi.org/10.1080/02688697.2018.1549315
Mair MJ, Wöhrer A, Furtner J et al (2020) Clinical characteristics and prognostic factors of adult patients with pilocytic astrocytoma. J Neurooncol 148:187–198. https://doi.org/10.1007/s11060-020-03513-9
Bond KM, Hughes JD, Porter AL et al (2018) Adult pilocytic astrocytoma: an institutional series and systematic literature review for extent of resection and recurrence. World Neurosurg 110:276–283. https://doi.org/10.1016/j.wneu.2017.11.102
Theeler BJ, Ellezam B, Sadighi ZS et al (2014) Adult pilocytic astrocytomas: clinical features and molecular analysis. Neuro Oncol 16:841–847. https://doi.org/10.1093/neuonc/not246
Jones DT, Gronych J, Lichter P et al (2012) MAPK pathway activation in pilocytic astrocytoma. Cell Mol Life Sci 69:1799–1811. https://doi.org/10.1007/s00018-011-0898-9
Kurani H, Gurav M, Shetty O et al (2019) Pilocytic astrocytomas: BRAFV600E and BRAF fusion expression patterns in pediatric and adult age groups. Childs Nerv Syst 35:1525–1536. https://doi.org/10.1007/s00381-019-04282-1
Capper D, Jones DTW, Sill M et al (2018) DNA methylation-based classification of central nervous system tumours. Nature 555:469–474. https://doi.org/10.1038/nature26000
Kameda M, Otani Y, Ichikawa T et al (2017) Congenital glioblastoma with distinct clinical and molecular characteristics: case reports and a literature review. World Neurosurg. https://doi.org/10.1016/j.wneu.2017.02.026
Reinhardt A, Stichel D, Schrimpf D et al (2018) Anaplastic astrocytoma with piloid features, a novel molecular class of IDH wildtype glioma with recurrent MAPK pathway, CDKN2A/B and ATRX alterations. Acta Neuropathol 136:273–291. https://doi.org/10.1007/s00401-018-1837-8
Johnson DR, Brown PD, Galanis E et al (2012) Pilocytic astrocytoma survival in adults: analysis of the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute. J Neurooncol 108:187–193. https://doi.org/10.1007/s11060-012-0829-0
Stuer C, Vilz B, Majores M et al (2007) Frequent recurrence and progression in pilocytic astrocytoma in adults. Cancer 110:2799–2808. https://doi.org/10.1002/cncr.23148
Voronina N, Aichmuller C, Kolb T et al (2021) The age of adult pilocytic astrocytoma cells. Oncogene 40:2830–2841. https://doi.org/10.1038/s41388-021-01738-0
Rodriguez FJ, Scheithauer BW, Burger PC et al (2010) Anaplasia in pilocytic astrocytoma predicts aggressive behavior. Am J Surg Pathol 34:147–160. https://doi.org/10.1097/PAS.0b013e3181c75238
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Bender K, Perez E, Chirica M et al (2021) High-grade astrocytoma with piloid features (HGAP): the Charite experience with a new central nervous system tumor entity. J Neurooncol 153:109–120. https://doi.org/10.1007/s11060-021-03749-z
Giannini C, Scheithauer BW, Burger PC et al (1999) Cellular proliferation in pilocytic and diffuse astrocytomas. J Neuropathol Exp Neurol 58:46–53. https://doi.org/10.1097/00005072-199901000-00006
Collins VP, Jones DT, Giannini C (2015) Pilocytic astrocytoma: pathology, molecular mechanisms and markers. Acta Neuropathol 129:775–788. https://doi.org/10.1007/s00401-015-1410-7
Ono T, Takahashi M, Hatakeyama J et al (2019) Clinical significance of molecular diagnosis of pilocytic astrocytoma: a case report. NMC Case Rep J 6:95–99. https://doi.org/10.2176/nmccrj.cr.2018-0282
Shibui S, Narita Y, Mizusawa J et al (2013) Randomized trial of chemoradiotherapy and adjuvant chemotherapy with nimustine (ACNU) versus nimustine plus procarbazine for newly diagnosed anaplastic astrocytoma and glioblastoma (JCOG0305). Cancer Chemother Pharmacol 71:511–521. https://doi.org/10.1007/s00280-012-2041-5
Funding
This study was supported by grants-in-aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science and Technology to Y.O. (No.21K20803 and 22K16687) and K.S. (No.22K06951) and the National Cancer Center Rare Cancer Grant (Award Number G007, AY).
Author information
Authors and Affiliations
Contributions
Conception and design: YS, KS, YO, KI. Development of the methodology and acquisition of experimental data: YS, KS, YO, YM, YH, KI. Collection of clinical samples and data: YS, YO, KF, JI, AU, NT, KM, SH, NK, RI, RM, YT, TY, KW, HY. Writing, review, and/or revision of the manuscript: YS, KS, YO, KI. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
DC declares that he holds a patent on a method for DNA methylation based tumor classification. The other authors declare that they have no competing interests.
Ethical approval
All procedures in involving human participants were performed in accordance with the ethical standards of the institutional research committee (IRB#1911–023).
Informed consent
Written consent for participation and publication was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suruga, Y., Satomi, K., Otani, Y. et al. The utility of DNA methylation analysis in elderly patients with pilocytic astrocytoma morphology. J Neurooncol 160, 179–189 (2022). https://doi.org/10.1007/s11060-022-04131-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04131-3