Abstract
Introduction
The so-called radiation-induced glioma (RIG, a secondary glioma after cranial irradiation), is a serious late effect after cranial radiation therapy. The clinical characteristics of and ideal treatment for these tumors are unclear. We analyzed our case series and conducted a comprehensive literature review to reveal the precise characteristics of RIGs.
Methods
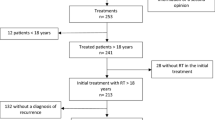
We analyzed the cases of six patients with RIGs treated at our institution and 354 patients with RIGs from the literature. The latency period from irradiation to the development of each RIG and the median overall survival of the patients were subjected to Kaplan–Meier analyses. Spearman’s correlation test was used to determine the relationship between age at irradiation and the latency period.
Results
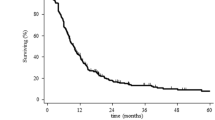
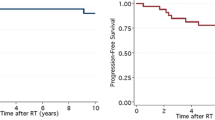
The mean age of the 360 patients at the development of RIG was 27.42 ± 17.87 years. The mean latency period was 11.35 ± 8.58 years. Multiple gliomas were observed in 28.4%. WHO grade 3 and 4 RIGs accounted for 93.3%. The latency periods were significant shorter in the higher WHO grade group (p = 0.0366) and the concomitant systemic chemotherapy group (p < 0.0001). Age at irradiation was negatively associated with the latency period (r =− 0.2287, p = 0.0219). The patients treated with radiotherapy achieved significantly longer survival compared to those treated without radiotherapy (p = 0.0011).
Conclusions
Development in younger age, multiplicity, and high incidence of grade 3 and 4 are the clinical characteristics of RIGs. Cranial irradiation at older ages and concomitant chemotherapy were associated with shorter latency for the development of RIG. Radiation therapy may be the feasible treatment option despite radiation-induced gliomas.



Similar content being viewed by others
References
Ostrom QT, Cioffi G, Gittleman H et al (2019) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2012–2016. Neuro Oncol 21:V1–V100. https://doi.org/10.1093/neuonc/noz150
Ostrom QT, Bauchet L, Davis FG et al (2014) The epidemiology of glioma in adults: a state of the science review. Neuro Oncol 16:896–913. https://doi.org/10.1093/neuonc/nou087
Brenner AV, Sugiyama H, Preston DL et al (2020) Radiation risk of central nervous system tumors in the life span study of atomic bomb survivors, 1958–2009. Eur J Epidemiol 35:591–600. https://doi.org/10.1007/s10654-019-00599-y
Terziev R, Psimaras D, Marie Y et al (2021) Cumulative incidence and risk factors for radiation induced leukoencephalopathy in high grade glioma long term survivors. Sci Rep 11:1–9. https://doi.org/10.1038/s41598-021-89216-1
Yamasaki F, Takayasu T, Nosaka R et al (2017) Development of cystic malacia after high-dose cranial irradiation of pediatric CNS tumors in long-term follow-up. Child’s Nerv Syst 33:957–964. https://doi.org/10.1007/s00381-017-3400-7
Greene-Schloesser D, Robbins ME (2012) Radiation-induced cognitive impairment-from bench to bedside. Neuro Oncol. https://doi.org/10.1093/neuonc/nos196
Vatner RE, Niemierko A, Misra M et al (2018) Endocrine deficiency as a function of radiation dose to the hypothalamus and pituitary in pediatric and young adult patients with brain tumors. I J Clin Oncol 36:2854–2862. https://doi.org/10.1200/JCO.2018.78.1492
Desai SS, Paulino AC, Mai WY, Teh BS (2006) Radiation-induced moyamoya syndrome. Int J Radiat Oncol Biol Phys 65:1222–1227. https://doi.org/10.1016/j.ijrobp.2006.01.038
Lew SM, Morgan JN, Psaty E et al (2006) Cumulative incidence of radiation-induced cavernomas in long-term survivors of medulloblastoma. J Neurosurg. https://doi.org/10.3171/ped.2006.104.2.103
Tsui K, Gajjar A, Li C et al (2015) Subsequent neoplasms in survivors of childhood central nervous system tumors: risk after modern multimodal therapy. Neuro Oncol 17:448–456. https://doi.org/10.1093/neuonc/nou279
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Cahan WG, Woodard HQ (1948) Sarcoma arising in irradiated bone; report of 11 cases. Cancer 1:3–29. https://doi.org/10.1002/1097-0142(194805)1:1%3c3::aid-cncr2820010103%3e3.0.co;2-7
Doskaliyev A, Yamasaki F, Kenjo M et al (2008) Secondary anaplastic oligodendroglioma after cranial irradiation: a case report. J Neurooncol 88:299–303. https://doi.org/10.1007/s11060-008-9564-y
Neglia JP, Robison LL, Stovall M et al (2006) New primary neoplasms of the central nervous system in survivors of childhood cancer: a report from the childhood cancer survivor study. J Natl Cancer Inst 98:1528–1537. https://doi.org/10.1093/jnci/djj411
Di Carlo DT, Cagnazzo F, Benedetto N et al (2019) Multiple high-grade gliomas: epidemiology, management, and outcome. a systematic review and meta-analysis. Neurosurg Rev 42:263–275. https://doi.org/10.1007/s10143-017-0928-7
Mascelli S, Nozza P, Sak K et al (2016) Distinctive genetic profile with IDH1, TP53, and MLH1 mutations in a radiation-induced anaplastic astrocytoma. Pediatr Blood Cancer 63:179–179. https://doi.org/10.1002/pbc.25671
Whitehouse JP, Howlett M, Federico A et al (2021) Defining the molecular features of radiation-induced glioma: a systematic review and meta-analysis. Neuro-Oncology Adv 3:1–16. https://doi.org/10.1093/noajnl/vdab109
Yamanaka R, Hayano A, Kanayama T (2018) Radiation-induced gliomas: a comprehensive review and meta-analysis. Neurosurg Rev 41:719–731. https://doi.org/10.1007/s10143-016-0786-8
Blagosklonny MV (2005) Carcinogenesis, cancer therapy and chemoprevention. Cell Death Differ 12:592–602. https://doi.org/10.1038/sj.cdd.4401610
Borgmann A, Zinn C, Hartmann R et al (2008) Secondary malignant neoplasms after intensive treatment of relapsed acute lymphoblastic leukaemia in childhood. Eur J Cancer 44:257–268. https://doi.org/10.1016/j.ejca.2007.09.019
Minniti G, Niyazi M, Alongi F et al (2021) Current status and recent advances in reirradiation of glioblastoma. Radiat Oncol. https://doi.org/10.1186/s13014-021-01767-9
Monje ML, Ramakrishna NR, Young G et al (2007) Durable response of a radiation-induced, high-grade cerebellar glioma to temozolomide. J Neurooncol 84:179–183. https://doi.org/10.1007/s11060-007-9354-y
Kon T (2013) Radiation-induced glioblastoma following radiotherapy for pituitary adenomas: marked response to chemotherapy. J Neurol Neurophysiol 04:3–5. https://doi.org/10.4172/2155-9562.1000155
Funding
The authors declare that no funds, grants, or other support were received for the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The material preparation and the data collection and analyses were performed by SO and FY. The first draft of the manuscript was written by SO, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Onishi, S., Yamasaki, F., Amatya, V.J. et al. Characteristics and therapeutic strategies of radiation-induced glioma: case series and comprehensive literature review. J Neurooncol 159, 531–538 (2022). https://doi.org/10.1007/s11060-022-04090-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04090-9