Abstract
Purpose
Although radiation therapy (RT) is a common treatment for pediatric brain tumors, it is associated with detrimental long-term effects such as impaired cognition, vascular injury, and increased stroke risk. This study aimed to develop metrics that describe vascular injury and relate them to the presence of cerebral microbleeds (CMBs) and cognitive performance scores.
Methods
Twenty-five young adult survivors of pediatric brain tumors treated with either whole-brain (n = 12), whole-ventricular (n = 7), or no RT (n = 6) underwent 7T MRI and neurocognitive testing. Simultaneously acquired MR angiography and susceptibility-weighted images were used to segment CMBs and vessels and quantify their radii and volume.
Results
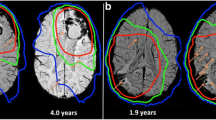
Patients treated with whole-brain RT had significantly lower arterial volumes (p = 0.003) and a higher proportion of smaller vessels (p = 0.003) compared to the whole-ventricular RT and non-irradiated control patients. Normalized arterial volume decreased with increasing CMB count (R = − 0.66, p = 0.003), and decreasing trends were observed with time since RT and at longitudinal follow-up. Global cognition and verbal memory significantly decreased with smaller normalized arterial volume (p ≤ 0.05).
Conclusions
Arterial volume is reduced with increasing CMB presence and is influenced by the total brain volume exposed to radiation. This work highlights the potential use of vascular-derived metrics as non-invasive markers of treatment-induced injury and cognitive impairment in pediatric brain tumor patients.





Similar content being viewed by others
References
https://www.cancer.org/cancer/cancer-in-children/types-of-childhood-cancers.html
https://www.childrensoncologygroup.org/index.php/braintumors
Knab B, Connell PP (2007) Radiotherapy for pediatric brain tumors: when and how. Expert Rev Anticancer Ther 7(sup1):S69–S77
Taphoorn MJ, Klein M (2004) Cognitive deficits in adult patients with brain tumors. Lancet Neurol 3(3):159–168
Lupo JM, Chuang CF, Chang SM, Barani IJ, Jimenez B, Hess CP, Nelson SJ (2012) 7-Tesla susceptibility-weighted imaging to assess the effects of radiotherapy on normal-appearing brain in patients with glioma. Int J RadiatOncolBiolPhys 82(3):e493-500
Greene-Schloesser D, Robbins ME, Peiffer AM, Shaw EG, Wheeler KT, Chan MD (2012) Radiation-induced brain injury: a review. Front Oncol 2:73
Campen CJ, Kranick SM, Kasner SE et al (2012) Cranial irradiation increases risk of stroke in pediatric brain tumor survivors. Stroke J CerebCirc 43(11):3035–3040
Wahl M, Anwar M, Hess CP, Chang SM, Lupo JM (2017) Relationship between radiation dose and microbleed formation in patients with malignant glioma. RadiatOncol 12(1):12
Lupo JM, Molinaro AM, Essock-Burns E, Butowski N, Chang SM, Cha S, Nelson SJ (2016) The effects of anti-angiogenic therapy on the formation of radiation-induced microbleeds in normal brain tissue of patients with glioma. NeuroOncol 18(1):87–95
Morrison MA, Hess CP, Clarke JL, Butowski N, Chang SM, Molinaro AM, Lupo JM (2019) Risk factors of radiotherapy-induced cerebral microbleeds and serial analysis of their size compared with white matter changes: A 7T MRI study in 113 adult patients with brain tumors. J MagnReson Imaging 50(3):868–877
Deshpande SS, Donneys A, Farberg AS, Tchanque-Fossuo CN, Felice PA, Buchman SR (2014) Quantification and characterization of radiation-induced changes to mandibular vascularity using micro-computed tomography. Ann PlastSurg 72(1):100–103
Gutierrez J, Cheung K, Bagci A et al (2015) Brain arterial diameters as a risk factor for vascular events. J Am Heart Assoc 4(8):e002289
Mazighi M, Labreuche J, Gongora-Rivera F, Duyckaerts C, Hauw JJ, Amarenco P (2009) Autopsy prevalence of proximal extracranial atherosclerosis in patients with fatal stroke. Stroke 40(3):713–718
Roth NM, Sontag MR, Kiani MF (1999) Early effects of ionizing radiation on the microvascular networks in normal tissue. Radiat Res 151(3):270–277
Cole FM, Yates P (1967) Intracerebralmicroaneurysms and small cerebrovascular lesions. Brain 90:759–768
Haacke EM, Xu Y, Cheng YC, Reichenbach JR (2004) Susceptibility weighted imaging (SWI). MagnReson Med 52(3):612–618
Lupo JM, Banerjee S, Hammond KE, Kelley DA, Xu D, Chang SM, Vigneron DB, Majumdar S, Nelson SJ (2009) GRAPPA-based susceptibility-weighted imaging of normal volunteers and patients with brain tumor at 7 T. MagnReson Imaging 27(4):480–488
Bian W, Hess CP, Chang SM, Nelson SJ, Lupo JM (2014) Susceptibility-weighted MR imaging of radiation therapy-induced cerebral microbleeds in patients with glioma: a comparison between 3T and 7T. Neuroradiology 56(2):91–96
Roddy E, Sear K, Felton E et al (2016) Presence of cerebral microbleeds is associated with worse executive function in pediatric brain tumor survivors. NeuroOncol 18(11):1548–1558
Akers A, Al-Shahi Salman R, Awad IA et al (2017) Synopsis of guidelines for the clinical management of cerebral cavernous malformations: consensus recommendations based on systematic literature review by the angioma alliance scientific advisory board clinical experts panel. Neurosurgery 80:665–680
Salehi A, Zhang JH, Obenaus A (2017) Response of the cerebral vasculature following traumatic brain injury. J Cereb Blood Flow Metab 37(7):2320–2339
Poels MMF, Ikram MA, Van Der Lugt A, Hofman A, Niessen WJ, Krestin GP, Breteler MM, Vernooij MW (2012) Cerebral microbleeds are associated with worse cognitive function: the rotterdam scan study. Neurology 78(5):326–333
Maruff P, Thomas E, Cysique L et al (2009) Validity of the CogState brief battery: relationship to standardized tests and sensitivity to cognitive impairment in mild traumatic brain injury, schizophrenia, and AIDS dementia complex. Arch ClinNeuropsychol 24(2):165–178
Morrison MA, Mueller S, Felton E et al (2021) Rate of radiation-induced microbleed formation on 7T MRI relates to cognitive impairment in young patients treated with radiation therapy for a brain tumor. Radiother Oncol 154(1):145–153
Bian W, Banerjee S, Kelly DA et al (2015) Simultaneous imaging of radiation-induced cerebral microbleeds, arteries and veins, using a multiple gradient echo sequence at 7 Tesla. J MagnReson Imaging 42(2):269–279
Smith SM (2002) Fast robust automated brain extraction. Ann Meet Organ Hum Brain Mapp 17(3):143–155
Avadiappan S, Payabvash S, Morrison MA et al (2020) A fully automated method for segmenting arteries and quantifying vessel radii on magnetic resonance angiography images of varying projection thickness. Frontiers Neurosci Brain Imaging Methods 14:537
Frangi AF, Niessen WJ, Vincken KL, Viergever MA (1998) Multiscale vessel enhancement filtering. In: Proceedings of medical image computing and computer-assisted intervention (MICCAI.), pp 130–137
Sethian JA (1996) A fast marching level set method for monotonically advancing fronts. ProcNatlAcadSci USA 93(4):1591–1595
Bullitt E, Zeng D, Mortamet B et al (2010) The effects of healthy aging on intracerebral blood vessels visualized by magnetic resonance angiography. Neurobiol Aging 31(2):290–300
Bian W, Hess CP, Chang SM, Nelson SJ, Lupo JM (2013) Computer-aided detection of radiation-induced cerebral microbleeds on susceptibility-weighted MR images. NeuroimageClin 2(1):282–290
Bullitt E, Lin NU, Smith KJ et al (2009) Blood vessel morphological changes as visualized by MRA during treatment of brain metastases: a feasibility study. Radiology 245(3):824–830
Acknowledgements
This work was supported by NIH-NICHD grant RO1HD079568 and funding from the La Roche family. We thank all the patients and families for their participation in this study.
Funding
This work was supported by NIH-NICHD grant RO1HD079568 as well as the La Roche family.
Author information
Authors and Affiliations
Contributions
Study design: JML, SM. Data collection: AJ, EF, SS. Image processing and analysis: SA, MAM. Statistical analysis: SA, AMM, MAM. Data interpretation: JML, SA, SM, AMM, SEB, CPH. Manuscript preparation: SA, MAM, JML. All authors approved the final manuscript content.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to report with respect to this study.
Ethical approval
This study was approved by our institutional review board and performed in accordance with the ethical standards of the institutional and national research committees or comparable ethical standards. Written informed consent, or assent when appropriate, was obtained from all individual participants and additional parental consent was obtained for all minors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
All work was performed while at UCSF.
Rights and permissions
About this article
Cite this article
Avadiappan, S., Morrison, M.A., Jakary, A. et al. Relationship between 7T MR-angiography features of vascular injury and cognitive decline in young brain tumor patients treated with radiation therapy. J Neurooncol 153, 143–152 (2021). https://doi.org/10.1007/s11060-021-03753-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-021-03753-3