Abstract
Background
Carmustine wafers (CW) are approved to treat newly or recurrent high-grade gliomas (HGG). Widespread use has been limited regarding some doubtful uncertainties about their efficacy, related increased risk of infection and expensive cost.
Objective
To describe the epidemiology of CW implantation, search for related complications, long-term survival and associated prognostic factors.
Methods
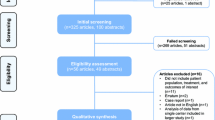
We processed the French medico-administrative national database to retrieve appropriate cases operated between 2010 and 2018. A survival analysis was conducted.
Results
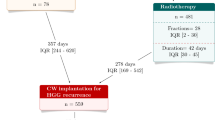
We identified 1659 patients treated in 39 institutions. Median age at CW implantation was 61 years and there was an over-representation of male (63.5%). 491 patients (29.6%) had previous diagnosis of glioma. Time between the first surgery and CW implantation was 0.9 years, IQR[0.6, 1.6]. The frontal lobe was the most frequently involved 29%. 131 patients (7.9%) had to be re operated on for a complication of which 121 for surgical site infection. At one year, 514 patients (31%) had died. Median overall survival (OS) was 1.4 years, 95% CI [1.3, 1.5]. OS at 1 and 2 year was 66%, 95%CI [63.7, 68.5], 32.3%, 95%CI [29.9, 35]. In the adjusted Cox regression, male gender & age at CW implantation were established as independent factors of OS in all three groups. Patients with recurrent HGG have a significant worse prognosis (HR = 0.71, 95% CI [0.62, 0.80] p < 0.001). A post-operative diagnosis of infection or intracranial bleeding eventually leading to a redo surgery was not associated with a decrease OS.
Conclusion
Over the past 9 years, there is a significant decrease utilisation of CW in France. OS after CW implantation is significantly variable as influenced by many factors such as age, gender or recurrent disease but not by post-operative complications. Compare to previous results, CW may increase the OS and this effect seems more pronounced when adjuvant RT/TMZ is given.


Similar content being viewed by others
Abbreviations
- CCAM:
-
Common classification of medical acts
- CI:
-
Confidence interval
- CSF:
-
Cerebro spinal fluid
- HR:
-
Hazard ratio
- HGG:
-
High grade glioma
- IQR:
-
Inter quartile range
- LGG:
-
Low grade glioma
- MGMT:
-
Methyl guanine methyl transferase
- NA:
-
Not applicable or not available
- NS:
-
Not significant
- PMSI:
-
Programme de Médicalisation des Systèmes d’Information
- OS:
-
Overall survival
- RT:
-
Radiotherapy
- TMZ:
-
Temozolomide
- RT/TMZ:
-
Concomitant radio-chemotherapy according the Stupp protocol
- WHO:
-
World Health Organization
References
Bauchet L, Mathieu-Daudé H, Fabbro-Peray P et al (2010) Oncological patterns of care and outcome for 952 patients with newly diagnosed glioblastoma in 2004. Neuro Oncol 12:725–735. https://doi.org/10.1093/neuonc/noq030
Koshy M, Villano JL, Dolecek TA et al (2012) Improved survival time trends for glioblastoma using the SEER 17 population-based registries. J Neurooncol 107:207–212. https://doi.org/10.1007/s11060-011-0738-7
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Harrell FE (2015) Regression modeling strategies. Springer-Verlag New York, Inc., Secaucus, NJ
Therneau TM, Grambsch PM (2000) Modeling survival data: extending the Cox model. Springer, New York
R Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
R Studio Team (2015) RStudio: Integrated development environment for r. RStudio Inc, Boston, MA
Nicholls SG, Quach P, von Elm E et al (2015) The REporting of studies conducted using observational routinely-collected health data (RECORD) statement: methods for arriving at consensus and developing reporting guidelines. PLoS ONE 10:e0125620. https://doi.org/10.1371/journal.pone.0125620
Lang TA, Altman DG (2015) Basic statistical reporting for articles published in biomedical journals: The "Statistical Analyses and Methods in the Published Literature" or the SAMPL Guidelines. Int J Nurs Studies 52:5–9. https://doi.org/10.1016/j.ijnurstu.2014.09.006
Champeaux C, Weller J, Katsahian S (2019) Epidemiology of meningiomas. A nationwide study of surgically treated tumours on french medico-administrative data. Cancer Epidemiol 58:63–70. https://doi.org/10.1016/j.canep.2018.11.004
Champeaux C, Weller J (2019) Long-term survival after decompressive craniectomy for malignant brain infarction: A 10-year nationwide study. Neurocrit Care. https://doi.org/10.1007/s12028-019-00774-9
Sage W, Guilfoyle M, Luney C et al (2018) Local alkylating chemotherapy applied immediately after 5-ALA guided resection of glioblastoma does not provide additional benefit. J Neurooncol 136:273–280. https://doi.org/10.1007/s11060-017-2649-8
Roux A, Caire F, Guyotat J et al (2017) Carmustine wafer implantation for high-grade gliomas: evidence-based safety efficacy and practical recommendations from the neuro-oncology club of the french society of neurosurgery. Neurochirurgie 63:433–443. https://doi.org/10.1016/j.neuchi.2017.07.003
Roux A, Peeters S, Zanello M et al (2017) Extent of resection and carmustine wafer implantation safely improve survival in patients with a newly diagnosed glioblastoma: A single center experience of the current practice. J Neurooncol 135:83–92. https://doi.org/10.1007/s11060-017-2551-4
Lechapt-Zalcman E, Levallet G, Dugué AE et al (2012) O(6) -methylguanine-DNA methyltransferase (MGMT) promoter methylation and low MGMT-encoded protein expression as prognostic markers in glioblastoma patients treated with biodegradable carmustine wafer implants after initial surgery followed by radiotherapy with concomitant and adjuvant temozolomide. Cancer 118:4545–4554. https://doi.org/10.1002/cncr.27441
Metellus P, Coulibaly B, Nanni I et al (2009) Prognostic impact of o6-methylguanine-DNA methyltransferase silencing in patients with recurrent glioblastoma multiforme who undergo surgery and carmustine wafer implantation: A prospective patient cohort. Cancer 115:4783–4794. https://doi.org/10.1002/cncr.24546
Chowdhary SA, Ryken T, Newton HB (2015) Survival outcomes and safety of carmustine wafers in the treatment of high-grade gliomas: A meta-analysis. J Neurooncol 122:367–382. https://doi.org/10.1007/s11060-015-1724-2
Ashby LS, Smith KA, Stea B (2016) Gliadel wafer implantation combined with standard radiotherapy and concurrent followed by adjuvant temozolomide for treatment of newly diagnosed high-grade glioma: A systematic literature review. World J Surg Oncol 14:225. https://doi.org/10.1186/s12957-016-0975-5
Fabbro-Peray P, Zouaoui S, Darlix A et al (2019) Association of patterns of care, prognostic factors, and use of radiotherapy-temozolomide therapy with survival in patients with newly diagnosed glioblastoma: A french national population-based study. J Neurooncol 142:91–101. https://doi.org/10.1007/s11060-018-03065-z
Price SJ, Whittle IR, Ashkan K et al (2012) NICE guidance on the use of carmustine wafers in high grade gliomas: A national study on variation in practice. Br J Neurosurg 26:331–335. https://doi.org/10.3109/02688697.2012.673651
Sun MZ, Oh T, Ivan ME et al (2015) Survival impact of time to initiation of chemoradiotherapy after resection of newly diagnosed glioblastoma. J Neurosurg 122:1144–1150. https://doi.org/10.3171/2014.9.JNS14193
Katsigiannis S, Krischek B, Barleanu S et al (2019) Impact of time to initiation of radiotherapy on survival after resection of newly diagnosed glioblastoma. Radiat Oncol. https://doi.org/10.1186/s13014-019-1272-6
Menei P, Metellus P, Parot-Schinkel E et al (2010) Biodegradable carmustine wafers (gliadel) alone or in combination with chemoradiotherapy: The french experience. Ann Surg Oncol 17:1740–1746. https://doi.org/10.1245/s10434-010-1081-5
Pallud J, Audureau E, Noel G et al (2015) Long-term results of carmustine wafer implantation for newly diagnosed glioblastomas: A controlled propensity-matched analysis of a french multicenter cohort. Neuro-oncology 17:1609–1619. https://doi.org/10.1093/neuonc/nov126
Hart MG, Garside R, Rogers G et al (2011) Chemotherapy wafers for high grade glioma. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD007294.pub2
Chaichana KL, Kone L, Bettegowda C et al (2015) Risk of surgical site infection in 401 consecutive patients with glioblastoma with and without carmustine wafer implantation. Neurol Res 37:717–726. https://doi.org/10.1179/1743132815Y.0000000042
Duntze J, Litré C-F, Eap C et al (2013) Implanted carmustine wafers followed by concomitant radiochemotherapy to treat newly diagnosed malignant gliomas: Prospective, observational, multicenter study on 92 cases. Ann Surg Oncol 20:2065–2072. https://doi.org/10.1245/s10434-012-2764-x
Barr JG, Grundy PL (2012) The effects of the NICE technology appraisal 121 (gliadel and temozolomide) on survival in high-grade glioma. Br J Neurosurg 26:818–822. https://doi.org/10.3109/02688697.2012.697221
Lillehei KO, Kalkanis SN, Liau LM et al (2018) Rationale and design of the 500-patient, 3-year, and prospective vigilant ObservatIon of GlIadeL WAfer ImplaNT registry. CNS Oncol 7:CNS08. https://doi.org/10.2217/cns-2017-0036
Xing W-K, Shao C, Qi Z-y et al (2015) The role of gliadel wafers in the treatment of newly diagnosed GBM: a meta-analysis. Drug Des Devel Ther 9:3341–3348. https://doi.org/10.2147/DDDT.S85943
Acknowledgements
We thank the ATIH for its assistance and Miss Rebecca Grant for her review of the manuscript.
Funding
This research received no grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
CC: Conceived and designed the analysis/Collected the data/Performed the analysis/Wrote the paper/Revision of the work, JW: Conceived and designed the analysis/Collected the data/Revision of the work/Final approval.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Champeaux, C., Weller, J. Implantation of carmustine wafers (Gliadel®) for high-grade glioma treatment. A 9-year nationwide retrospective study. J Neurooncol 147, 159–169 (2020). https://doi.org/10.1007/s11060-020-03410-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03410-1