Abstract
Introduction
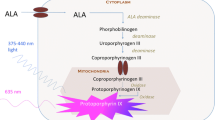
Fluorescence guided surgery has developed over the last 2 decades as a formidable augmentation strategy to promote maximal safe resection and diagnostic accuracy within gliomas. The majority of the literature evidence supporting this modality utilizes 5-aminolevulinic acid in the setting of high-grade gliomas. The role for fluorescence guided surgery in low-grade gliomas is less well defined.
Results
This review describes the existing literature discussing the utilization of 5-aminolevulinic acid for fluorescence guided surgery in low-grade gliomas, including its pertinence in identification of anaplastic foci and potential role in guiding resection following combination with augmentation strategies for detection.
Conclusion
The advance in operative technology and growth of research analyzing 5-aminolevulinic acid will continue to enhance the role of fluorescence guided surgery within the standard of surgical management for low-grade gliomas.
Similar content being viewed by others
References
Hervey-Jumper SL, Berger MS (2016) Maximizing safe resection of low- and high-grade glioma. J Neurooncol 130:269–282
Ahmadi R, Dictus C, Hartmann C et al (2009) Long-term outcome and survival of surgically treated supratentorial low-grade glioma in adult patients. Acta Neurochir (Wien) 151:1359–1365
Keles GE, Lamborn KR, Berger MS (2001) Low-grade hemispheric gliomas in adults: a critical review of extent of resection as a factor influencing outcome. J Neurosurg 95:735–745
McGirt MJ, Chaichana KL, Attenello FJ et al (2008) Extent of surgical resection is independently associated with survival in patients with hemispheric infiltrating low-grade gliomas. Neurosurgery 63:700–707 (author reply 707–708)
Stummer W, Reulen HJ, Novotny A et al (2003) Fluorescence-guided resections of malignant gliomas—an overview. Acta Neurochir Suppl 88:9–12
Li Y, Rey-Dios R, Roberts DW et al (2014) Intraoperative fluorescence-guided resection of high-grade gliomas: a comparison of the present techniques and evolution of future strategies. World Neurosurg 82:175–185
Stummer W, Pichlmeier U, Meinel T et al (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7:392–401
Lau D, Hervey-Jumper SL, Chang S et al (2016) A prospective Phase II clinical trial of 5-aminolevulinic acid to assess the correlation of intraoperative fluorescence intensity and degree of histologic cellularity during resection of high-grade gliomas. J Neurosurg 124:1300–1309
Zhao S, Wu J, Wang C et al (2013) Intraoperative fluorescence-guided resection of high-grade malignant gliomas using 5-aminolevulinic acid-induced porphyrins: a systematic review and meta-analysis of prospective studies. PLoS ONE 8:e63682
Teixidor P, Arraez MA, Villalba G et al (2016) Safety and efficacy of 5-aminolevulinic acid for high grade glioma in usual clinical practice: a prospective cohort study. PLoS ONE 11:e0149244
Schatlo B, Fandino J, Smoll NR et al (2015) Outcomes after combined use of intraoperative MRI and 5-aminolevulinic acid in high-grade glioma surgery. Neuro Oncol 17:1560–1567
Stummer W, Novotny A, Stepp H et al (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93:1003–1013
Aldave G, Tejada S, Pay E et al (2013) Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic Acid-guided surgery. Neurosurgery 72:915–920 (discussion 920-911)
Cordova JS, Gurbani SS, Holder CA et al (2016) Semi-automated volumetric and morphological assessment of glioblastoma resection with fluorescence-guided surgery. Mol Imaging Biol 18:454–462
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820
Nishikawa R (2011) Fluorescence illuminates the way. Neuro Oncol 13:805
Jaber M, Wolfer J, Ewelt C et al (2016) The value of 5-aminolevulinic acid in low-grade gliomas and high-grade gliomas lacking glioblastoma imaging features: an analysis based on fluorescence, magnetic resonance imaging, 18F-fluoroethyl tyrosine positron emission tomography, and tumor molecular factors. Neurosurgery 78:401–411 (discussion 411)
Widhalm G, Kiesel B, Woehrer A et al (2013) 5-Aminolevulinic acid induced fluorescence is a powerful intraoperative marker for precise histopathological grading of gliomas with non-significant contrast-enhancement. PLoS ONE 8:e76988
Belykh E, Miller EJ, Hu D et al (2018) Scanning fiber endoscope improves detection of 5-aminolevulinic acid-induced protoporphyrin IX fluorescence at the boundary of infiltrative glioma. World Neurosurg 113:e51–e69
Xie Y, Thom M, Ebner M et al (2017) Wide-field spectrally resolved quantitative fluorescence imaging system: toward neurosurgical guidance in glioma resection. J Biomed Opt 22:1–14
Sanai N, Snyder LA, Honea NJ et al (2011) Intraoperative confocal microscopy in the visualization of 5-aminolevulinic acid fluorescence in low-grade gliomas. J Neurosurg 115:740–748
Ferraro N, Barbarite E, Albert TR et al (2016) The role of 5-aminolevulinic acid in brain tumor surgery: a systematic review. Neurosurg Rev 39:545–555
Hadjipanayis CG, Widhalm G, Stummer W (2015) What is the surgical benefit of utilizing 5-aminolevulinic acid for fluorescence-guided surgery of malignant gliomas? Neurosurgery 77:663–673
Stummer W, Stepp H, Wiestler OD et al (2017) Randomized, prospective double-blinded study comparing 3 different doses of 5-aminolevulinic acid for fluorescence-guided resections of malignant gliomas. Neurosurgery 81:230–239
Halani SH, Adamson DC (2016) Clinical utility of 5-aminolevulinic acid HCl to better visualize and more completely remove gliomas. Onco Targets Ther 9:5629–5642
Eljamel S (2015) 5-ALA fluorescence image guided resection of glioblastoma multiforme: a meta-analysis of the literature. Int J Mol Sci 16:10443–10456
Maugeri R, Villa A, Pino M et al (2018) With a little help from my friends: the role of intraoperative fluorescent dyes in the surgical management of high-grade gliomas. Brain Sci 8:31
Coons SW, Johnson PC (1993) Regional heterogeneity in the proliferative activity of human gliomas as measured by the Ki-67 labeling index. J Neuropathol Exp Neurol 52:609–618
Stummer W, Stocker S, Wagner S et al (1998) Intraoperative detection of malignant gliomas by 5-aminolevulinic acid-induced porphyrin fluorescence. Neurosurgery 42:518–525 (discussion 525-516)
Paulus W, Peiffer J (1989) Intratumoral histologic heterogeneity of gliomas. A quantitative study. Cancer 64:442–447
Hatakeyama T, Kawai N, Nishiyama Y et al (2008) 11C-methionine (MET) and 18F-fluorothymidine (FLT) PET in patients with newly diagnosed glioma. Eur J Nucl Med Mol Imaging 35:2009–2017
Sadeghi N, Salmon I, Decaestecker C et al (2007) Stereotactic comparison among cerebral blood volume, methionine uptake, and histopathology in brain glioma. AJNR Am J Neuroradiol 28:455–461
Singhal T, Narayanan TK, Jain V et al (2008) 11C-L-methionine positron emission tomography in the clinical management of cerebral gliomas. Mol Imaging Biol 10:1–18
Nabavi A, Black PM, Gering DT et al (2001) Serial intraoperative magnetic resonance imaging of brain shift. Neurosurgery 48:787–797 (discussion 797-788)
Widhalm G, Wolfsberger S, Minchev G et al (2010) 5-Aminolevulinic acid is a promising marker for detection of anaplastic foci in diffusely infiltrating gliomas with nonsignificant contrast enhancement. Cancer 116:1545–1552
Valdes PA, Leblond F, Kim A et al (2011) Quantitative fluorescence in intracranial tumor: implications for ALA-induced PpIX as an intraoperative biomarker. J Neurosurg 115:11–17
Stummer W, Stocker S, Novotny A et al (1998) In vitro and in vivo porphyrin accumulation by C6 glioma cells after exposure to 5-aminolevulinic acid. J Photochem Photobiol B 45:160–169
Tsugu A, Ishizaka H, Mizokami Y et al (2011) Impact of the combination of 5-aminolevulinic acid-induced fluorescence with intraoperative magnetic resonance imaging-guided surgery for glioma. World Neurosurg 76:120–127
Marbacher S, Klinger E, Schwyzer L et al (2014) Use of fluorescence to guide resection or biopsy of primary brain tumors and brain metastases. Neurosurg Focus 36:E10
Jaber AEC, Wölfer J, Brokinkel B, Thoams C, Hasselblatt M, Grauer O, Stummer W (2018) Is visible ALA-induced fluorescence an independent biomarker for prognosis in histologically confirmed (WHO 2016) low grade gliomas? Neurosurgery. https://doi.org/10.1093/neuros/nyy365
Saito K, Hirai T, Takeshima H et al (2017) Genetic factors affecting intraoperative 5-aminolevulinic acid-induced fluorescence of diffuse gliomas. Radiol Oncol 51:142–150
Chan DTM, Yi-Pin Sonia H, Poon WS (2017) 5-Aminolevulinic acid fluorescence guided resection of malignant glioma: Hong Kong experience. Asian J Surg 41(5):467–472
Ewelt C, Floeth FW, Felsberg J et al (2011) Finding the anaplastic focus in diffuse gliomas: the value of Gd-DTPA enhanced MRI, FET-PET, and intraoperative, ALA-derived tissue fluorescence. Clin Neurol Neurosurg 113:541–547
Valdes PA, Jacobs V, Harris BT et al (2015) Quantitative fluorescence using 5-aminolevulinic acid-induced protoporphyrin IX biomarker as a surgical adjunct in low-grade glioma surgery. J Neurosurg 123:771–780
Administration USFaD: Aminolevulinic acid hydrochloride, known as ALA HCl (Gleolan, NX Development Corp.) as an optical imaging agent indicated in patients with gliomas. https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm562645.htm
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hendricks, B.K., Sanai, N. & Stummer, W. Fluorescence-guided surgery with aminolevulinic acid for low-grade gliomas. J Neurooncol 141, 13–18 (2019). https://doi.org/10.1007/s11060-018-03026-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-03026-6