Abstract
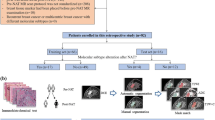
Current biopsy planning based on contrast-enhanced T1W (CET1W) or FLAIR sequences frequently delivers biopsy samples that are not in concordance with the gross tumor diagnosis. This study investigates whether the quantitative information of transfer constant Ktrans maps derived from T1W dynamic contrast-enhanced MRI (DCE-MRI) can help enhance the quality of biopsy target selection in glioma. 28 patients with suspected glioma received MRI including DCE-MRI and a standard neuronavigation protocol of 3D FLAIR- and CET1W data sets (0.1 mmol/kg gadobutrol) at 3.0 T. After exclusion of five cases with no Ktrans-elevation, 2–6 biopsy targets were independently selected by a neurosurgeon (samples based on standard imaging) and a neuroradiologist (samples based on kinetic parameter Ktrans) per case and tissue samples corresponding to these targets were collected by a separate independent neurosurgeon. Standard technique and Ktrans-based samples were rated for diagnostic concordance with the gross tumor resection reference diagnosis (67 WHO IV; 24 WHO III and II) by a neuropathologist blinded for selection mode. Ktrans-based sample targets differed from standard technique sample targets in 90/91 cases. More Ktrans-based than standard imaging-based samples could be extracted. Diagnoses from Ktrans-based samples were more frequently concordant with the reference gross tumor diagnoses than those from standard imaging-based samples (WHO IV: 30/39 vs. 11/20; p = 0.08; WHO III/II: 12/13 vs. 6/11; p = 0.06). In 4/5 non-contrast-enhancing gliomas, Ktrans-based selection revealed significantly more accurate samples than standard technique sample-selection (10/12 vs. 2/8 samples; p = 0.02). If Ktrans elevation is present, Ktrans-based biopsy targeting provides significantly more diagnostic tissue samples in non-contrast-enhancing glioma than selection based on CET1W and FLAIR-weighted images alone.



Similar content being viewed by others
Abbreviations
- CET1W MRI:
-
Contrast-enhanced T1-weighted MRI
- DSC-MRI:
-
T2*-dynamic susceptibility-weighted MRI
- DCE-MRI:
-
T1W dynamic contrast-enhanced MRI
- DTS:
-
presumed diagnosis based on target tissue sample only
- GTRD:
-
reference diagnosis from the completely resected gross tumor
- kep :
-
reflux volume transfer constant (of contrast agent)
- Ktrans :
-
efflux volume transfer constant (of contrast agent)
- MRS:
-
MR spectroscopy
- ve :
-
extracellular-extravascular volume fraction (of contrast agent)
- WHO:
-
World Health Organization
References
Paulus W, Peiffer J (1989) Intratumoral histologic heterogeneity of gliomas. A quantitative study. Cancer 64:442–447
Coons SW, Johnson PC, Scheithauer BW, Yates AJ, Pearl DK (1997) Improving diagnostic accuracy and interobserver concordance in the classification and grading of primary gliomas. Cancer 79:1381–1393
Kunz M, Thon N, Eigenbrod S, Hartmann C, Egensperger R, Herms J, Geisler J, La Fougere C, Lutz J, Linn J, Kreth S, von Deimling A, Tonn JC, Kretzschmar HA, Popperl G, Kreth FW (2011) Hot spots in dynamic (18)FET-PET delineate malignant tumor parts within suspected WHO grade II gliomas. Neuro Oncol 13:307–316. doi:10.1093/neuonc/noq196
Sottoriva A, Spiteri I, Piccirillo SG, Touloumis A, Collins VP, Marioni JC, Curtis C, Watts C, Tavare S (2013) Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proc Natl Acad Sci USA 110:4009–4014. doi:10.1073/pnas.1219747110
Ganslandt O, Stadlbauer A, Fahlbusch R, Kamada K, Buslei R, Blumcke I, Moser E, Nimsky C (2005) Proton magnetic resonance spectroscopic imaging integrated into image-guided surgery: correlation to standard magnetic resonance imaging and tumor cell density. Neurosurgery 56:291–298 (discussion 291–298)
Muragaki Y, Chernov M, Maruyama T, Ochiai T, Taira T, Kubo O, Nakamura R, Iseki H, Hori T, Takakura K (2008) Low-grade glioma on stereotactic biopsy: how often is the diagnosis accurate? Minim Invasive Neurosurg 51:275–279. doi:10.1055/s-0028-1082322
Hermann EJ, Hattingen E, Krauss JK, Marquardt G, Pilatus U, Franz K, Setzer M, Gasser T, Tews DS, Zanella FE, Seifert V, Lanfermann H (2008) Stereotactic biopsy in gliomas guided by 3-tesla 1H-chemical-shift imaging of choline. Stereotact Funct Neurosurg 86:300–307. doi:10.1159/000155232
Weber MA, Henze M, Tuttenberg J, Stieltjes B, Meissner M, Zimmer F, Burkholder I, Kroll A, Combs SE, Vogt-Schaden M, Giesel FL, Zoubaa S, Haberkorn U, Kauczor HU, Essig M (2010) Biopsy targeting gliomas: do functional imaging techniques identify similar target areas? Invest Radiol 45:755–768. doi:10.1097/RLI.0b013e3181ec9db0
Pafundi DH, Laack NN, Youland RS, Parney IF, Lowe VJ, Giannini C, Kemp BJ, Grams MP, Morris JM, Hoover JM, Hu LS, Sarkaria JN, Brinkmann DH (2013) Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: results of a prospective pilot study. Neuro Oncol 15:1058–1067. doi:10.1093/neuonc/not002
Lopez WO, Cordeiro JG, Albicker U, Doostkam S, Nikkhah G, Kirch RD, Trippel M, Reithmeier T (2015) Correlation of (18)F-fluoroethyl tyrosine positron-emission tomography uptake values and histomorphological findings by stereotactic serial biopsy in newly diagnosed brain tumors using a refined software tool. Onco Targets Ther 8:3803–3815. doi:10.2147/OTT.S87126
Dammers R, Schouten JW, Haitsma IK, Vincent AJ, Kros JM, Dirven CM (2010) Towards improving the safety and diagnostic yield of stereotactic biopsy in a single centre. Acta Neurochir (Wien) 152:1915–1921. doi:10.1007/s00701-010-0752-0
Roessler K, Gatterbauer B, Becherer A, Paul M, Kletter K, Prayer D, Hoeftberger R, Hainfellner J, Asenbaum S, Knosp E (2007) Surgical target selection in cerebral glioma surgery: linking methionine (MET) PET image fusion and neuronavigation. Minim Invasive Neurosurg 50:273–280. doi:10.1055/s-2007-991143
Chaskis C, Stadnik T, Michotte A, Van Rompaey K, D’Haens J (2006) Prognostic value of perfusion-weighted imaging in brain glioma: a prospective study. Acta Neurochir (Wien) 148: 277–285. doi:10.1007/s00701-005-0718-9 (discussion 285)
Son BC, Kim MC, Choi BG, Kim EN, Baik HM, Choe BY, Naruse S, Kang JK (2001) Proton magnetic resonance chemical shift imaging (1H CSI)-directed stereotactic biopsy. Acta Neurochir (Wien) 143:45–49 (discussion 49–50)
Lefranc M, Monet P, Desenclos C, Peltier J, Fichten A, Toussaint P, Sevestre H, Deramond H, Le Gars D (2012) Perfusion MRI as a neurosurgical tool for improved targeting in stereotactic tumor biopsies. Stereotact Funct Neurosurg 90:240–247. doi:10.1159/000338092
Knopp EA, Cha S, Johnson G, Mazumdar A, Golfinos JG, Zagzag D, Miller DC, Kelly PJ, Kricheff II (1999) Glial neoplasms: dynamic contrast-enhanced T2*-weighted MR imaging. Radiology 211:791–798. doi:10.1148/radiology.211.3.r99jn46791
Tofts PS, Brix G, Buckley DL, Evelhoch JL, Henderson E, Knopp MV, Larsson HB, Lee TY, Mayr NA, Parker GJ, Port RE, Taylor J, Weisskoff RM (1999) Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging 10:223–232
Roberts HC, Roberts TP, Brasch RC, Dillon WP (2000) Quantitative measurement of microvascular permeability in human brain tumors achieved using dynamic contrast-enhanced MR imaging: correlation with histologic grade. AJNR Am J Neuroradiol 21:891–899
Provenzale JM, Mukundan S, Dewhirst M (2005) The role of blood-brain barrier permeability in brain tumor imaging and therapeutics. AJR Am J Roentgenol 185:763–767. doi:10.2214/ajr.185.3.01850763
Zhang N, Zhang L, Qiu B, Meng L, Wang X, Hou BL (2012) Correlation of volume transfer coefficient Ktrans with histopathologic grades of gliomas. J Magn Reson Imaging 36:355–363. doi:10.1002/jmri.23675
Dujardin MI, Sourbron SP, Chaskis C, Verellen D, Stadnik T, de Mey J, Luypaert R (2012) Quantification of cerebral tumour blood flow and permeability with T1-weighted dynamic contrast enhanced MRI: a feasibility study. J Neuroradiol 39:227–235. doi:10.1016/j.neurad.2011.07.001
Jia ZZ, Geng DY, Liu Y, Chen XR, Zhang J (2013) Microvascular permeability of brain astrocytoma with contrast-enhanced magnetic resonance imaging: correlation analysis with histopathologic grade. Chin Med J (Engl) 126:1953–1956
Tan CH, Hobbs BP, Wei W, Kundra V (2015) Dynamic contrast-enhanced MRI for the detection of prostate cancer: meta-analysis. AJR Am J Roentgenol 204:W439–W448. doi:10.2214/AJR.14.13373
An YS, Kang DK, Jung YS, Han S, Kim TH (2015) Tumor metabolism and perfusion ratio assessed by 18F-FDG PET/CT and DCE-MRI in breast cancer patients: correlation with tumor subtype and histologic prognostic factors. Eur J Radiol 84:1365–1370. doi:10.1016/j.ejrad.2015.03.026
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. doi:10.1007/s00401-016-1545-1
Wang HZ, Riederer SJ, Lee JN (1987) Optimizing the precision in T1 relaxation estimation using limited flip angles. Magn Reson Med 5:399–416
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann HJ (2005) Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Invest Radiol 40:715–724
Della Puppa A, Persano L, Masi G, Rampazzo E, Sinigaglia A, Pistollato F, Denaro L, Barzon L, Palu G, Basso G, Scienza R, d’Avella D (2012) MGMT expression and promoter methylation status may depend on the site of surgical sample collection within glioblastoma: a possible pitfall in stratification of patients? J Neurooncol 106:33–41. doi:10.1007/s11060-011-0639-9
Aker FV, Hakan T, Karadereler S, Erkan M (2005) Accuracy and diagnostic yield of stereotactic biopsy in the diagnosis of brain masses: comparison of results of biopsy and resected surgical specimens. Neuropathology 25:207–213
Pedeutour-Braccini Z, Burel-Vandenbos F, Goze C, Roger C, Bazin A, Costes-Martineau V, Duffau H, Rigau V (2015) Microfoci of malignant progression in diffuse low-grade gliomas: towards the creation of an intermediate grade in glioma classification? Virchows Arch 466:433–444. doi:10.1007/s00428-014-1712-5
Widhalm G, Kiesel B, Woehrer A, Traub-Weidinger T, Preusser M, Marosi C, Prayer D, Hainfellner JA, Knosp E, Wolfsberger S (2013) 5-Aminolevulinic acid induced fluorescence is a powerful intraoperative marker for precise histopathological grading of gliomas with non-significant contrast-enhancement. PLoS ONE 8:e76988. doi:10.1371/journal.pone.0076988
Cordova JS, Shu HK, Liang Z, Gurbani SS, Cooper LA, Holder CA, Olson JJ, Kairdolf B, Schreibmann E, Neill SG, Hadjipanayis CG, Shim H (2016) Whole-brain spectroscopic MRI biomarkers identify infiltrating margins in glioblastoma patients. Neuro Oncol 18:1180–1189. doi:10.1093/neuonc/now036
Jia ZZ, Gu HM, Zhou XJ, Shi JL, Li MD, Zhou GF, Wu XH (2015) The assessment of immature microvascular density in brain gliomas with dynamic contrast-enhanced magnetic resonance imaging. Eur J Radiol 84:1805–1809. doi:10.1016/j.ejrad.2015.05.035
van Osch MJ, van der Grond J, Bakker CJ (2005) Partial volume effects on arterial input functions: shape and amplitude distortions and their correction. J Magn Reson Imaging 22:704–709. doi:10.1002/jmri.20455
van der Schaaf I, Vonken EJ, Waaijer A, Velthuis B, Quist M, van Osch T (2006) Influence of partial volume on venous output and arterial input function. AJNR Am J Neuroradiol 27:46–50
Calamante F (2013) Arterial input function in perfusion MRI: a comprehensive review. Prog Nucl Magn Reson Spectrosc 74:1–32. doi:10.1016/j.pnmrs.2013.04.002
Hansen AE, Pedersen H, Rostrup E, Larsson HB (2009) Partial volume effect (PVE) on the arterial input function (AIF) in T1-weighted perfusion imaging and limitations of the multiplicative rescaling approach. Magn Reson Med 62:1055–1059. doi:10.1002/mrm.22098
Haroon HA, Buckley DL, Patankar TA, Dow GR, Rutherford SA, Baleriaux D, Jackson A (2004) A comparison of Ktrans measurements obtained with conventional and first pass pharmacokinetic models in human gliomas. J Magn Reson Imaging 19:527–536. doi:10.1002/jmri.20045
Heisen M, Fan X, Buurman J, van Riel NA, Karczmar GS, ter Haar Romeny BM (2010) The influence of temporal resolution in determining pharmacokinetic parameters from DCE-MRI data. Magn Reson Med 63:811–816. doi:10.1002/mrm.22171
Lavini C, Verhoeff JJ (2010) Reproducibility of the gadolinium concentration measurements and of the fitting parameters of the vascular input function in the superior sagittal sinus in a patient population. Magn Reson Imaging 28:1420–1430. doi:10.1016/j.mri.2010.06.017
Sourbron SP, Buckley DL (2013) Classic models for dynamic contrast-enhanced MRI. NMR Biomed 26:1004–1027. doi:10.1002/nbm.2940
van Niekerk CG, van der Laak JA, Hambrock T, Huisman HJ, Witjes JA, Barentsz JO, Hulsbergen-van de Kaa CA (2014) Correlation between dynamic contrast-enhanced MRI and quantitative histopathologic microvascular parameters in organ-confined prostate cancer. Eur Radiol 24:2597–2605. doi:10.1007/s00330-014-3301-z
Jia Z, Geng D, Xie T, Zhang J, Liu Y (2012) Quantitative analysis of neovascular permeability in glioma by dynamic contrast-enhanced MR imaging. J Clin Neurosci 19:820–823. doi:10.1016/j.jocn.2011.08.030
Jackson A, Jayson GC, Li KL, Zhu XP, Checkley DR, Tessier JJ, Waterton JC (2003) Reproducibility of quantitative dynamic contrast-enhanced MRI in newly presenting glioma. Br J Radiol 76:153–162. doi:10.1259/bjr/70653746
Lavini C, Verhoeff JJ, Majoie CB, Stalpers LJ, Richel DJ, Maas M (2011) Model-based, semiquantitative and time intensity curve shape analysis of dynamic contrast-enhanced MRI: a comparison in patients undergoing antiangiogenic treatment for recurrent glioma. J Magn Reson Imaging 34:1303–1312. doi:10.1002/jmri.22742
Weber MA, Zoubaa S, Schlieter M, Juttler E, Huttner HB, Geletneky K, Ittrich C, Lichy MP, Kroll A, Debus J, Giesel FL, Hartmann M, Essig M (2006) Diagnostic performance of spectroscopic and perfusion MRI for distinction of brain tumors. Neurology 66:1899–1906. doi:10.1212/01.wnl.0000219767.49705.9c
Funding
Neither of the authors did receive any funding from the company or any third parties for this study nor did anybody received payments related to this study. The study is investigator-initiated and received no funding from third parties.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
V.C.K. received compensation for an invited talk at a Philips User Meeting in Switzerland in 2015. J.G. is employed by Philips Healthcare, the company providing both MRI and software used in this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Keil, V.C., Pintea, B., Gielen, G.H. et al. Biopsy targeting with dynamic contrast-enhanced versus standard neuronavigation MRI in glioma: a prospective double-blinded evaluation of selection benefits. J Neurooncol 133, 155–163 (2017). https://doi.org/10.1007/s11060-017-2424-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-017-2424-x