Abstract
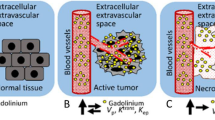
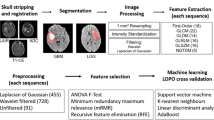
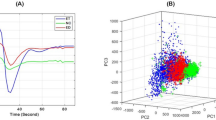
This study proposes an automatic method for identification and quantification of different tissue components: the non-enhanced infiltrative tumor, vasogenic edema and enhanced tumor areas, at the subject level, in patients with glioblastoma (GB) based on dynamic contrast enhancement (DCE) and dynamic susceptibility contrast (DSC) MRI. Nineteen MR data sets, obtained from 12 patients with GB, were included. Seven patients were scanned before and 8 weeks following bevacizumab initiation. Segmentation of the tumor area was performed based on the temporal data of DCE and DSC at the group-level using k-means algorithm, and further at the subject-level using support vector machines algorithm. The obtained components were associated to different tissues types based on their temporal characteristics, calculated perfusion and permeability values and MR-spectroscopy. The method enabled the segmentation of the tumor area into the enhancing permeable component; the non-enhancing hypoperfused component, associated with vasogenic edema; and the non-enhancing hyperperfused component, associated with infiltrative tumor. Good agreement was obtained between the group-level, unsupervised and subject-level, supervised classification results, with significant correlation (r = 0.93, p < 0.001) and average symmetric root-mean-square surface distance of 2.5 ± 5.1 mm. Longitudinal changes in the volumes of the three components were assessed alongside therapy. Tumor area segmentation using DCE and DSC can be used to differentiate between vasogenic edema and infiltrative tumors in patients with GB, which is of major clinical importance in therapy response assessment.




Similar content being viewed by others
References
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, Degroot J, Wick W, Gilbert MR, Lassman AB, Tsien C, Mikkelsen T, Wong ET, Chamberlain MC, Stupp R, Lamborn KR, Vogelbaum MA, van den Bent MJ, Chang SM (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972. doi:10.1200/JCO.2009.26.3541
Demir MK, Hakan T, Akinci O, Berkman Z (2005) Primary cerebellar glioblastoma multiforme. Diagn Interv Radiol 11:83–86
Ostergaard L, Sorensen AG, Kwong KK, Weisskoff RM, Gyldensted C, Rosen BR (1996) High resolution measurement of cerebral blood flow using intravascular tracer bolus passages. Part II: experimental comparison and preliminary results. Magn Reson Med 36:726–736
Ostergaard L, Weisskoff RM, Chesler DA, Gyldensted C, Rosen BR (1996) High resolution measurement of cerebral blood flow using intravascular tracer bolus passages. Part I: mathematical approach and statistical analysis. Magn Reson Med 36:715–725
Sourbron S, Ingrisch M, Siefert A, Reiser M, Herrmann K (2009) Quantification of cerebral blood flow, cerebral blood volume, and blood-brain-barrier leakage with DCE-MRI. Magn Reson Med 62:205–217. doi:10.1002/mrm.22005
Al-Okaili RN, Krejza J, Wang S, Woo JH, Melhem ER (2006) Advanced MR imaging techniques in the diagnosis of intraaxial brain tumors in adults. Radiographics 26(Suppl 1):S173–S189. doi:10.1148/rg.26si065513
Cha S (2006) Update on brain tumor imaging: from anatomy to physiology. AJNR Am J Neuroradiol 27:475–487
Fatterpekar GM, Galheigo D, Narayana A, Johnson G, Knopp E (2012) Treatment-related change versus tumor recurrence in high-grade gliomas: a diagnostic conundrum–use of dynamic susceptibility contrast-enhanced (DSC) perfusion MRI. AJR Am J Roentgenol 198:19–26. doi:10.2214/AJR.11.7417
Sourbron SP, Buckley DL (2013) Classic models for dynamic contrast-enhanced MRI. NMR Biomed 26:1004–1027. doi:10.1002/nbm.2940
Zhang W, Kreisl TN, Solomon J, Reynolds RC, Glen DR, Cox RW, Fine HA, Butman JA (2009) Acute Effects of Bevacizumab on Glioblastoma Vascularity Assessed with DCE-MRI and Relation to Patient Survival. The International Society for Magnetic Resonance in Medicine, Honolulu
Sorensen AG, Batchelor TT, Zhang WT, Chen PJ, Yeo P, Wang M, Jennings D, Wen PY, Lahdenranta J, Ancukiewicz M, di Tomaso E, Duda DG, Jain RK (2009) A “vascular normalization index” as potential mechanistic biomarker to predict survival after a single dose of cediranib in recurrent glioblastoma patients. Cancer Res 69:5296–5300. doi:10.1158/0008-5472.CAN-09-0814
Huo J, Okada K, van Rikxoort EM, Kim HJ, Alger JR, Pope WB, Goldin JG, Brown MS (2013) Ensemble segmentation for GBM brain tumors on MR images using confidence-based averaging. Med Phys 40:093502. doi:10.1118/1.4817475
Liberman G, Louzoun Y, Aizenstein O, Blumenthal DT, Bokstein F, Palmon M, Corn BW, Ben Bashat D (2013) Automatic multi-modal MR tissue classification for the assessment of response to bevacizumab in patients with glioblastoma. Eur J Radiol 82:e87–e94. doi:10.1016/j.ejrad.2012.09.001
Wu W, Chen AY, Zhao L, Corso JJ (2013) Brain tumor detection and segmentation in a CRF (conditional random fields) framework with pixel-pairwise affinity and superpixel-level features. Int J Comput Assist Radiol Surg. doi:10.1007/s11548-013-0922-7
Zhu Y, Young GS, Xue Z, Huang RY, You H, Setayesh K, Hatabu H, Cao F, Wong ST (2012) Semi-automatic segmentation software for quantitative clinical brain glioblastoma evaluation. Acad Radiol 19:977–985. doi:10.1016/j.acra.2012.03.026
Assefa D, Keller H, Menard C, Laperriere N, Ferrari RJ, Yeung I (2010) Robust texture features for response monitoring of glioblastoma multiforme on T1-weighted and T2-FLAIR MR images: a preliminary investigation in terms of identification and segmentation. Med Phys 37:1722–1736
Zollner FG, Emblem KE, Schad LR (2010) Support vector machines in DSC-based glioma imaging: suggestions for optimal characterization. Magn Reson Med 64:1230–1236. doi:10.1002/mrm.22495
Emblem KE, Nedregaard B, Hald JK, Nome T, Due-Tonnessen P, Bjornerud A (2009) Automatic glioma characterization from dynamic susceptibility contrast imaging: brain tumor segmentation using knowledge-based fuzzy clustering. J Magn Reson Imaging 30:1–10. doi:10.1002/jmri.21815
Singh A, Rathore R, Gupta R, Haris M, Rathore DK, Verma SK, Purwar A, Bayu G, Sarma MK, Singh JK (2007) Segmentation of Gd-DTPA Enhancing Lesion of Brain using Time to Peak of Concentration Time Curve and its Pharmacokinetic Analysis in Dynamic Contrast Enhanced (DCE) MRI. The International Society for Magnetic Resonance in Medicine, Berlin
Vonken EP, van Osch MJ, Bakker CJ, Viergever MA (2000) Simultaneous quantitative cerebral perfusion and Gd-DTPA extravasation measurement with dual-echo dynamic susceptibility contrast MRI. Magn Reson Med 43:820–827. doi:10.1002/1522-2594(200006)43:6<820:AID-MRM7>3.0.CO;2-F
Bagher-Ebadian H, Jain R, Nejad-Davarani SP, Mikkelsen T, Lu M, Jiang Q, Scarpace L, Arbab AS, Narang J, Soltanian-Zadeh H, Paudyal R, Ewing JR (2012) Model selection for DCE-T1 studies in glioblastoma. Magn Reson Med 68:241–251. doi:10.1002/mrm.23211
Nadav G, Liberman G, Artzi M, Kiryati N, Ben Bashat D (2014) Flow and permeability estimation from DCE data: 2-compartment exchange and Tofts models comparison. The International Society for Magnetic Resonance in Medicine, Milan
Calamante F (2013) Arterial input function in perfusion MRI: a comprehensive review. Prog Nucl Magn Reson Spectrosc 74:1–32. doi:10.1016/j.pnmrs.2013.04.002
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TE, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, De Stefano N, Brady JM, Matthews PM (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23(Suppl 1):S208–S219. doi:10.1016/j.neuroimage.2004.07.051
Artzi M, Aizenstein O, Jonas-Kimchi T, Myers V, Hallevi H, Ben Bashat D (2013) FLAIR lesion segmentation: application in patients with brain tumors and acute ischemic stroke. Eur J Radiol 82:1512–1518. doi:10.1016/j.ejrad.2013.05.029
Artzi M, Aizenstein O, Hendler T, Ben Bashat D (2011) Unsupervised multiparametric classification of dynamic susceptibility contrast imaging: study of the healthy brain. Neuroimage 56:858–864. doi:10.1016/j.neuroimage.2011.03.027
Tofts PS, Kermode AG (1991) Measurement of the blood-brain barrier permeability and leakage space using dynamic MR imaging. 1. Fundamental concepts. Magn Reson Med 17:357–367
Liberman G, Louzoun Y, Colliot O, BB D (2011) T1 mapping, AIF and pharmacokinetic parameter extraction from dynamic contrast enhancement MRI data. Multimodal Brain Image Anal 7012:76–83
Gerig G, Jomier M, Chakos M (2001) Valmet: a new validation tool for assessing and improving 3D object segmentation. MICCAI, Beijing
Hossman KA, Bloink M (1981) Blood flow and regulation of blood flow in experimental peritumoral edema. Stroke 12:211–217
Law M, Cha S, Knopp EA, Johnson G, Arnett J, Litt AW (2002) High-grade gliomas and solitary metastases: differentiation by using perfusion and proton spectroscopic MR imaging. Radiology 222:715–721. doi:10.1148/radiol.2223010558
Law M, Yang S, Wang H, Babb JS, Johnson G, Cha S, Knopp EA, Zagzag D (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. AJNR Am J Neuroradiol 24:1989–1998
Croteau D, Scarpace L, Hearshen D, Gutierrez J, Fisher JL, Rock JP, Mikkelsen T (2001) Correlation between magnetic resonance spectroscopy imaging and image-guided biopsies: semiquantitative and qualitative histopathological analyses of patients with untreated glioma. Neurosurgery 49:823–829
Law M, Yang S, Babb JS, Knopp EA, Golfinos JG, Zagzag D, Johnson G (2004) Comparison of cerebral blood volume and vascular permeability from dynamic susceptibility contrast-enhanced perfusion MR imaging with glioma grade. AJNR Am J Neuroradiol 25:746–755
Lu S, Ahn D, Johnson G, Law M, Zagzag D, Grossman RI (2004) Diffusion-tensor MR imaging of intracranial neoplasia and associated peritumoral edema: introduction of the tumor infiltration index. Radiology 232:221–228. doi:10.1148/radiol.2321030653
Kinoshita M, Goto T, Okita Y, Kagawa N, Kishima H, Hashimoto N, Yoshimine T (2010) Diffusion tensor-based tumor infiltration index cannot discriminate vasogenic edema from tumor-infiltrated edema. J Neurooncol 96:409–415. doi:10.1007/s11060-009-9979-0
Pope WB, Young JR, Ellingson BM (2011) Advances in MRI assessment of gliomas and response to anti-VEGF therapy. Curr Neurol Neurosci Rep 11:336–344. doi:10.1007/s11910-011-0179-x
LaViolette PS, Cohen AD, Rand SD, Mueller W, Schmainda KM (2011) Independent component analysis of dynamic susceptibility contrast MRI in brain tumor: a new biomarker for measuring tumor perfusion patterns. The International Society for Magnetic Resonance in Medicine, Montreal
LaViolette PS, Cohen AD, Prah MA, Rand SD, Connelly J, Malkin MG, Mueller WM, Schmainda KM (2013) Vascular change measured with independent component analysis of dynamic susceptibility contrast MRI predicts bevacizumab response in high-grade glioma. Neuro Oncol 15:442–450. doi:10.1093/neuonc/nos323
Acknowledgments
To Guy Nadav for technical support and to Vicki Myers for editorial assistance. This work was performed in partial fulfillment of the requirements for a Ph.D. degree of Artzi Moran, Sackler Faculty of Medicine, Tel Aviv University, Israel.
Conflict of interest
We declare that there is no conflict of interest for any of the authors.
Funding
This work was supported by the James S. McDonnell Foundation number 220020176.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Artzi, M., Blumenthal, D.T., Bokstein, F. et al. Classification of tumor area using combined DCE and DSC MRI in patients with glioblastoma. J Neurooncol 121, 349–357 (2015). https://doi.org/10.1007/s11060-014-1639-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-014-1639-3