Abstract
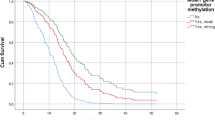
DNA methylation plays an important role in cancer biology and methylation events are important prognostic and predictive markers in many tumor types. We have used methylation-specific multiplex ligation-dependent probe amplification to survey the methylation status of MGMT and 25 tumor suppressor genes in 73 glioblastoma cases. The data obtained was correlated with overall survival and response to treatment. The study revealed that methylation of promoter regions in TP73 (seven patients), THBS1 (eight patients) and PYCARD (nine patients) was associated with improved outcome, whereas GATA5 (21 patients) and WT1 (24 patients) promoter methylation were associated with poor outcome. In patients treated with temozolomide and radiation MGMT and PYCARD promoter methylation events remained associated with improved survival whereas GATA5 was associated with a poor outcome. The identification of GATA5 promoter methylation in glioblastoma has not previously been reported. Furthermore, a cumulative methylation score separated patients into survival groups better than any single methylation event. In conclusion, we have identified specific methylation events associated with patient outcome and treatment response in glioblastoma, and these may be of functional and predictive/prognostic significance. This study therefore provides novel candidates and approaches for future prospective validation.


Similar content being viewed by others
References
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) WHO classification of tumours of the central nervous system. IARC Press, Lyon
Stupp R, Bent MJ, Weller M, Fisher B, Taphoorn MJ, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia SK, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Baylin SB, Jones PA (2011) A decade of exploring the cancer epigenome—biological and translational implications. Nat Rev Cancer 11:726–734
Esteller M, Garcia-Foncillas J, Andion E, Goodman SN, Hidalgo OF, Vanaclocha V, Baylin SB, Herman JG (2000) Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med 343:1350–1354
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003
Noushmehr H, Weisenberger DJ, Diefes K, Phillips HS, Pujara K, Berman BP, Pan F, Pelloski CE, Sulman EP, Bhat KP, Verhaak RG, Hoadley KA, Hayes DN, Perou CM, Schmidt HK, Ding L, Wilson RK, Van Den Berg D, Shen H, Bengtsson H, Neuvial P, Cope LM, Buckley J, Herman JG, Baylin SB, Laird PW, Aldape K, Cancer Genome Atlas Research Network (2010) Identification of a CpG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell 17:510–522
Turcan S, Rohle D, Goenka A, Walsh LA, Fang F, Yilmaz E, Campos C, Fabius AW, Lu C, Ward PS, Thompson CB, Kaufman A, Guryanova O, Levine R, Heguy A, Viale A, Morris LG, Huse JT, Mellinghoff IK, Chan TA (2012) IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature 483:479–483
Malzkorn B, Wolter M, Riemenschneider MJ, Reifenberger G (2011) Unraveling the glioma epigenome: from molecular mechanisms to novel biomarkers and therapeutic targets. Brain Pathol 21:619–632
Nygren AO, Ameziane N, Duarte HM, Vijzelaar RN, Waisfisz Q, Hess CJ, Schouten JP, Errami A (2005) Methylation-specific MLPA (MS-MLPA): simultaneous detection of CpG methylation and copy number changes of up to 40 sequences. Nucleic Acids Res 33:e128
Jeuken JW, Cornelissen SJ, Vriezen M, Dekkers MM, Errami A, Sijben A, Boots-Sprenger SH, Wesseling P (2007) MS-MLPA: an attractive alternative laboratory assay for robust, reliable, and semiquantitative detection of MGMT promoter hypermethylation in gliomas. Lab Invest 87:1055–1065
NICE guidance, TA121 ‘Carmustine implants and temozolomide for the treatment of newly diagnosed high-grade glioma; publications.nice.org.uk, June 2007
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbour Laboratory Press, New York
Grambsch PM, Therneau TM (1994) Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 81:515–526
Ellingson BM, Cloughesy TF, Pope WB, Zaw TM, Phillips H, Lalezari S, Nghiemphu PL, Ibrahim H, Naeini KM, Harris RJ, Lai A (2012) Anatomic localization of O6-methylguanine DNA methyltransferase (MGMT) promoter methylated and unmethylated tumors: a radiographic study in 358 de novo human glioblastomas. Neuroimage 59:908–916
Weller M, Stupp R, Reifenberger G, Brandes AA, van den Bent MJ, Wick W, Hegi ME (2010) MGMT promoter methylation in malignant gliomas: Ready for personalized medicine? Nat Rev Neurol 6:39–51
Etcheverry A, Aubry M, de Tayrac M, Vauleon E, Boniface R, Guenot F, Saikali S, Hamlat A, Riffaud L, Menei P, Quillien V, Mosser J (2010) DNA methylation in glioblastoma: impact on gene expression and clinical outcome. BMC Genomics 14:701
Qian XC, Brent TP (1997) Methylation hot spots in the 5′ flanking region denote silencing of the O6-methylguanine-DNA methyltransferase gene. Cancer Res 57:3672–3677
van Nifterik KA, van den Berg J, van der Meide WF, Ameziane N, Wedekind LE, Steenbergen RDM, Leenstra S, Lafleur MVM, Slotman BJ, Stalpers LJA, Sminia P (2010) Absence of the MGMT protein as well as methylation of the MGMT promoter predict the sensitivity for temozolomide. Br J Cancer 103:29–35
Christians A, Hartmann C, Benner A, Meyer J, von Deimling A, Weller M, Wick W, Weiler M (2012) Prognostic value of three different methods of mgmt promoter methylation analysis in a prospective trial on newly diagnosed glioblastoma. PLoS ONE 7(3):e33449
Zheng S, Houseman EA, Morrison Z, Wrensch MR, Patoka JS, Ramos C, Haas-Kogan DA, McBride S, Marsit CJ, Christensen BC, Nelson HH, Stokoe D, Wiemels JL, Chang SM, Prados MD, Tihan T, Vandenberg SR, Kelsey KT, Berger MS, Wiencke JK (2011) DNA hypermethylation profiles associated with glioma subtypes and EZH2 and IGFBP2 mRNA expression. Neurooncology 13:280–289
Courtois S, Hainaut P, de Fromentel CC. TP63, TP73: The Guardian’s Elder Brothers. In: Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000. Available from: http://www.ncbi.nlm.nih.gov/books/NBK6163/
Agundez M, Grau L, Palou J, Algaba F, Villavicencio H, Sanchez-Carbayo M (2011) Evaluation of the methylation status of tumour suppressor genes for predicting bacillus Calmette–Guérin response in patients with T1G3 high-risk bladder tumours. Eur Urol 60:131–140
Seeber LM, Zweemer RP, Marchionni L, Massuger LF, Smit VT, van Baal WM, Verheijen RH, van Diest PJ (2010) Methylation profiles of endometrioid and serous endometrial cancers. Endocr Relat Cancer 17:663–673
Stephen JK, Chen KM, Raitanen M, Grénman S, Worsham MJ (2009) DNA hypermethylation profiles in squamous cell carcinoma of the vulva. Int J Gynecol Pathol 28:63–75
Worsham MJ, Chen KM, Meduri V, Nygren AO, Errami A, Schouten JP, Benninger MS (2006) Epigenetic events of disease progression in head and neck squamous cell carcinoma. Arch Otolaryngol Head Neck Surg 132:668–677
Dong S, Pang JC, Hu J, Zhou LF, Ng HK (2002) Transcriptional inactivation of TP73 expression in oligodendroglial tumors. Int J Cancer 98:370–375
Lomas J, Amiñoso C, Gonzalez-Gomez P, Eva Alonso M, Arjona D, Lopez-Marin I, de Campos JM, Isla A, Vaquero J, Gutierrez M, Sarasa JL, Josefa Bello M, Rey JA (2004) Methylation status of TP73 in meningiomas. Cancer Genet Cytogenet 148:148–151
Li Q, Ahuja N, Burger PC, Issa JP (1999) Methylation and silencing of the Thrombospondin-1 promoter in human cancer. Oncogene 18:3284–3289
Christensen BC, Smith AA, Zheng S, Koestler DC, Houseman EA, Marsit CJ, Wiemels JL, Nelson HH, Karagas MR, Wrensch MR, Kelsey KT, Wiencke JK (2011) DNA methylation, isocitrate dehydrogenase mutation, and survival in glioma. J Natl Cancer Inst 103:1–11
Martinez R, Schackert G, Esteller M (2007) Hypermethylation of the proapoptotic gene TMS1/ASC: prognostic importance in glioblastoma multiforme. J Neurooncol 82:133–139
Yu J, Zhang H, Gu J, Lin S, Li J, Lu W, Wang Y, Zhu J (2004) Methylation profiles of thirty four promoter-CpG islands and concordant methylation behaviours of sixteen genes that may contribute to carcinogenesis of astrocytoma. BMC Cancer 4:65
Fu B, Guo M, Wang S, Campagna D, Luo M, Herman JG, Iacobuzio-Donahue CA (2007) Evaluation of GATA-4 and GATA-5 methylation profiles in human pancreatic cancers indicate promoter methylation patterns distinct from other human tumor types. Cancer Biol Ther 6:1546–1552
Acknowledgments
We would like to thank our colleagues within the Yorkshire Regional Genetics Service and Leeds Teaching Hospitals NHS Trust for their support in providing the diagnostic MGMT methylation service with special thanks to Ian Cook and Lampros Mavrogiannis. DAC is supported by a UK Medical Research Council Career Development Fellowship [G0802416]. SEL is supported by a grant from Brain Tumour Research and Support across Yorkshire and Candlelighters.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The experiments described in this paper comply with the current laws of the United Kingdom regarding ethics.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rankeillor, K.L., Cairns, D.A., Loughrey, C. et al. Methylation-specific multiplex ligation-dependent probe amplification identifies promoter methylation events associated with survival in glioblastoma. J Neurooncol 117, 243–251 (2014). https://doi.org/10.1007/s11060-014-1372-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-014-1372-y