Abstract
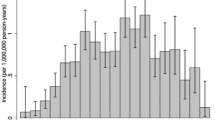
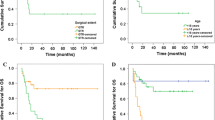
Patients with World Health Organization (WHO) grade II supratentorial ependymomas are commonly observed after gross total resection (GTR), although supporting data are limited. We sought to characterize the natural history of such tumors. We used the Surveillance, Epidemiology, and End Results program to identify 112 patients ages 0–77 diagnosed with WHO grade II ependymomas between 1988 and 2007, of whom 63 (56 %) and 49 (44 %) had supratentorial and infratentorial primaries, respectively. Inclusion criteria were strict to ensure patient homogeneity. Of 33 patients with supratentorial tumors after GTR, 18 (55 %) received adjuvant radiation therapy and 15 (45 %) did not. Ependymoma-specific mortality (ESM) was the primary endpoint. With a median follow up of 4.5 years, only 1 of 33 patients with supratentorial ependymoma died of their disease after GTR; the 5-year estimate of ESM in this population was 3.3 % (95 % CI 0.2–14.8 %). Among patients with infratentorial ependymomas after GTR, the 5-year estimate of ESM was 8.7 % (95 % CI 1.4–24.6 %). In patients with subtotally resected tumors, 5-year estimates of ESM in patients with supratentorial and infratentorial primaries were 20.1 % (95 % CI 8.0–36.2 %) and 12.3 % (95 % CI 2.9–28.8 %), respectively. Among the whole cohort, on both univariable and multivariable regression, extent of resection was predictive of ESM, while tumor location and use of radiation were not. After GTR, patients with WHO grade II supratentorial ependymomas have a very favorable natural history with low associated cancer-specific mortality. Observation, with radiation reserved as a salvage option, may be a reasonable postoperative strategy in this population.

Similar content being viewed by others
References
Merchant TE, Li C, Xiong X, Kun LE, Boop FA, Sanford RA (2009) Conformal radiotherapy after surgery for paediatric ependymoma: a prospective study. Lancet Oncol 10:258–266
Rogers L, Pueschel J, Spetzler R, Shapiro W, Coons S, Thomas T, Speiser B (2005) Is gross-total resection sufficient treatment for posterior fossa ependymomas? J Neurosurg 102:629–636. doi:10.3171/jns.2005.102.4.0629
Korshunov A, Golanov A, Sycheva R, Timirgaz V (2004) The histologic grade is a main prognostic factor for patients with intracranial ependymomas treated in the microneurosurgical era: an analysis of 258 patients. Cancer 100:1230–1237. doi:10.1002/cncr.20075
Awaad YM, Allen JC, Miller DC, Schneider SJ, Wisoff J, Epstein FJ (1996) Deferring adjuvant therapy for totally resected intracranial ependymoma. Pediatr Neurol 14:216–219
Hukin J, Epstein F, Lefton D, Allen J (1998) Treatment of intracranial ependymoma by surgery alone. Pediatr Neurosurg 29:40–45
Palma L, Celli P, Mariottini A, Zalaffi A, Schettini G (2000) The importance of surgery in supratentorial ependymomas. Long-term survival in a series of 23 cases. Childs Nerv Syst 16:170–175
Metellus P, Figarella-Branger D, Guyotat J, Barrie M, Giorgi R, Jouvet A, Chinot O (2008) Supratentorial ependymomas: prognostic factors and outcome analysis in a retrospective series of 46 adult patients. Cancer 113:175–185. doi:10.1002/cncr.23530
Mansur DB, Perry A, Rajaram V, Michalski JM, Park TS, Leonard JR, Luchtman-Jones L, Rich KM, Grigsby PW, Lockett MA, Wahab SH, Simpson JR (2005) Postoperative radiation therapy for grade II and III intracranial ependymoma. Int J Radiat Oncol Biol Phys 61:387–391. doi:10.1016/j.ijrobp.2004.06.002
Amirian ES, Armstrong TS, Gilbert MR, Scheurer ME (2012) Predictors of survival among older adults with ependymoma. J Neurooncol 107:183–189. doi:10.1007/s11060-011-0730-2
McGuire CS, Sainani KL, Fisher PG (2009) Both location and age predict survival in ependymoma: a SEER study. Pediatr Blood Cancer 52:65–69. doi:10.1002/pbc.21806
Children’s Oncology Group Protocol Homepage. ACNS0831. Phase III randomized trial of post-radiation chemotherapy in patients with newly diagnosed ependymoma ages 1 to 21 years. Available at http://www.childrensoncologygroup.org. Accessed 2 Oct 2013
Surveillance, epidemiology, and end results (SEER) program (www.seer.cancer.gov) research data (1973–2009), National Cancer Institute, DCCPS, surveillance research program, surveillance systems branch, released April 2012, based on the November 2011 submission
Iezzoni LI, Ngo LH, Li D, Roetzheim RG, Drews RE, McCarthy EP (2008) Early stage breast cancer treatments for younger medicare beneficiaries with different disabilities. Health Serv Res 43:1752–1767. doi:10.1111/j.1475-6773.2008.00853.x
Gaynor JJ, Feuer EJ, Tan CC, Wu DH, Little CR, Straus DJ, Clarkson BD, Brennan MF (1993) On the use of cause-specific failure and conditional failure probabilities: examples from clinical oncology data. J Am Stat Assoc 88:400–409
Gray RJ (1988) A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat 16:1141–1154
Fine JP, Gray RJ (1999) A proportional hazards model for the sub distribution of a competing risk. J Am Stat Assoc 94:496–509
Scrucca L, Santucci A, Aversa F (2007) Competing risk analysis using R: an easy guide for clinicians. Bone Marrow Transplant 40:381–387. doi:10.1038/sj.bmt.1705727
Conklin HM, Li C, Xiong X, Ogg RJ, Merchant TE (2008) Predicting change in academic abilities after conformal radiation therapy for localized ependymoma. J Clin Oncol 26:3965–3970. doi:10.1200/JCO.2007.15.9970
Hua C, Bass JK, Khan R, Kun LE, Merchant TE (2008) Hearing loss after radiotherapy for pediatric brain tumors: effect of cochlear dose. Int J Radiat Oncol Biol Phys 72:892–899. doi:10.1016/j.ijrobp.2008.01.050
Merchant TE, Rose SR, Bosley C, Wu S, Xiong X, Lustig RH (2011) Growth hormone secretion after conformal radiation therapy in pediatric patients with localized brain tumors. J Clin Oncol 29:4776–4780. doi:10.1200/JCO.2011.37.9453
Duffner PK, Krischer JP, Horowitz ME, Cohen ME, Burger PC, Friedman HS, Kun LE (1998) Second malignancies in young children with primary brain tumors following treatment with prolonged postoperative chemotherapy and delayed irradiation: a pediatric oncology group study. Ann Neurol 44:313–316. doi:10.1002/ana.410440305
Armstrong GT, Liu Q, Yasui Y, Huang S, Ness KK, Leisenring W, Hudson MM, Donaldson SS, King AA, Stovall M, Krull KR, Robison LL, Packer RJ (2009) Long-term outcomes among adult survivors of childhood central nervous system malignancies in the childhood cancer survivor study. J Natl Cancer Inst 101:946–958. doi:10.1093/jnci/djp148djp148
Galloway TJ, Indelicato DJ, Amdur RJ, Morris CG, Swanson EL, Marcus RB (2012) Analysis of dose at the site of second tumor formation after radiotherapy to the central nervous system. Int J Radiat Oncol Biol Phys 82:90–94. doi:10.1016/j.ijrobp.2010.10.062
St Clair WH, Adams JA, Bues M, Fullerton BC, La Shell S, Kooy HM, Loeffler JS, Tarbell NJ (2004) Advantage of protons compared to conventional X-ray or IMRT in the treatment of a pediatric patient with medulloblastoma. Int J Radiat Oncol Biol Phys 58:727–734. doi:10.1016/S0360-3016(03)01574-8
MacDonald SM, Safai S, Trofimov A, Wolfgang J, Fullerton B, Yeap BY, Bortfeld T, Tarbell NJ, Yock T (2008) Proton radiotherapy for childhood ependymoma: initial clinical outcomes and dose comparisons. Int J Radiat Oncol Biol Phys 71:979–986. doi:10.1016/j.ijrobp.2007.11.065
Kuhlthau KA, Pulsifer MB, Yeap BY, Rivera Morales D, Delahaye J, Hill KS, Ebb D, Abrams AN, Macdonald SM, Tarbell NJ, Yock TI (2012) Prospective study of health-related quality of life for children with brain tumors treated with proton radiotherapy. J Clin Oncol 30:2079–2086. doi:10.1200/JCO.2011.37.0577
MacDonald SM, Trofimov A, Safai S, Adams J, Fullerton B, Ebb D, Tarbell NJ, Yock TI (2011) Proton radiotherapy for pediatric central nervous system germ cell tumors: early clinical outcomes. Int J Radiat Oncol Biol Phys 79:121–129. doi:10.1016/j.ijrobp.2009.10.069
Grill J, Le Deley MC, Gambarelli D, Raquin MA, Couanet D, Pierre-Kahn A, Habrand JL, Doz F, Frappaz D, Gentet JC, Edan C, Chastagner P, Kalifa C (2001) Postoperative chemotherapy without irradiation for ependymoma in children under 5 years of age: a multicenter trial of the French Society of Pediatric Oncology. J Clin Oncol 19:1288–1296
Zacharoulis S, Levy A, Chi SN, Gardner S, Rosenblum M, Miller DC, Dunkel I, Diez B, Sposto R, Ji L, Asgharzadeh S, Hukin J, Belasco J, Dubowy R, Kellie S, Termuhlen A, Finlay J (2007) Outcome for young children newly diagnosed with ependymoma, treated with intensive induction chemotherapy followed by myeloablative chemotherapy and autologous stem cell rescue. Pediatr Blood Cancer 49:34–40. doi:10.1002/pbc.20935
Grundy RG, Wilne SA, Weston CL, Robinson K, Lashford LS, Ironside J, Cox T, Chong WK, Campbell RH, Bailey CC, Gattamaneni R, Picton S, Thorpe N, Mallucci C, English MW, Punt JA, Walker DA, Ellison DW, Machin D (2007) Primary postoperative chemotherapy without radiotherapy for intracranial ependymoma in children: the UKCCSG/SIOP prospective study. Lancet Oncol 8:696–705. doi:10.1016/S1470-2045(07)70208-5
Massimino M, Gandola L, Barra S, Giangaspero F, Casali C, Potepan P, Di Rocco C, Nozza P, Collini P, Viscardi E, Bertin D, Biassoni V, Cama A, Milanaccio C, Modena P, Balter R, Tamburrini G, Peretta P, Mascarin M, Scarzello G, Fidani P, Milano GM, Sardi I, Genitori L, Garre ML (2011) Infant ependymoma in a 10-year AIEOP (Associazione Italiana Ematologia Oncologia Pediatrica) experience with omitted or deferred radiotherapy. Int J Radiat Oncol Biol Phys 80:807–814. doi:10.1016/j.ijrobp.2010.02.048
Merchant TE, Mulhern RK, Krasin MJ, Kun LE, Williams T, Li C, Xiong X, Khan RB, Lustig RH, Boop FA, Sanford RA (2004) Preliminary results from a phase II trial of conformal radiation therapy and evaluation of radiation-related CNS effects for pediatric patients with localized ependymoma. J Clin Oncol 22:3156–3162. doi:10.1200/JCO.2004.11.142
Needle MN, Goldwein JW, Grass J, Cnaan A, Bergman I, Molloy P, Sutton L, Zhao H, Garvin JH Jr, Phillips PC (1997) Adjuvant chemotherapy for the treatment of intracranial ependymoma of childhood. Cancer 80:341–347. doi:10.1002/(SICI)1097-0142(19970715)80:2<341:AID-CNCR23>3.0.CO;2-T
Timmermann B, Kortmann RD, Kuhl J, Meisner C, Slavc I, Pietsch T, Bamberg M (2000) Combined postoperative irradiation and chemotherapy for anaplastic ependymomas in childhood: results of the German prospective trials HIT 88/89 and HIT 91. Int J Radiat Oncol Biol Phys 46:287–295
Children’s Oncology Group Protocol Homepage. ACNS0121. A Phase II trial of conformal RT for pediatric patients with Localized ependymoma, chemotherapy prior to 2nd surgery for incompletely resected ependymoma and observations for completely resected, differentiated, supratentorial ependymoma. Available at http://www.childrensoncologygroup.org. Accessed 2 Oct 2013
Taylor MD, Poppleton H, Fuller C, Su X, Liu Y, Jensen P, Magdaleno S, Dalton J, Calabrese C, Board J, Macdonald T, Rutka J, Guha A, Gajjar A, Curran T, Gilbertson RJ (2005) Radial glia cells are candidate stem cells of ependymoma. Cancer Cell 8:323–335. doi:10.1016/j.ccr.2005.09.001
Rodriguez D, Cheung MC, Housri N, Quinones-Hinojosa A, Camphausen K, Koniaris LG (2009) Outcomes of malignant CNS ependymomas: an examination of 2408 cases through the surveillance, epidemiology, and end results (SEER) database (1973–2005). J Surg Res 156:340–351. doi:10.1016/j.jss.2009.04.024
McGuire CS, Sainani KL, Fisher PG (2009) Incidence patterns for ependymoma: a surveillance, epidemiology, and end results study. J Neurosurg 110:725–729. doi:10.3171/2008.9.JNS08117
Evans AE, Anderson JR, Lefkowitz-Boudreaux IB, Finlay JL (1996) Adjuvant chemotherapy of childhood posterior fossa ependymoma: cranio-spinal irradiation with or without adjuvant CCNU, vincristine, and prednisone: a Childrens Cancer Group study. Med Pediatr Oncol 27:8–14. doi:10.1002/(SICI)1096-911X(199607)27:1<8:AID-MPO3>3.0.CO;2-K
Souweidane MM, Bouffet E, Finlay J (1998) The role of chemotherapy in newly diagnosed ependymoma of childhood. Pediatr Neurosurg 28:273–278
Niazi TN, Jensen EM, Jensen RL (2009) WHO Grade II and III supratentorial hemispheric ependymomas in adults: case series and review of treatment options. J Neurooncol 91:323–328. doi:10.1007/s11060-008-9717-z
Metellus P, Guyotat J, Chinot O, Durand A, Barrie M, Giorgi R, Jouvet A, Figarella-Branger D (2010) Adult intracranial WHO grade II ependymomas: long-term outcome and prognostic factor analysis in a series of 114 patients. Neuro Oncol 12:976–984. doi:10.1093/neuonc/noq047noq047
Venkatramani R, Dhall G, Patel M, Grimm J, Hawkins C, McComb G, Krieger M, Wong K, O’Neil S, Finlay JL (2012) Supratentorial ependymoma in children: to observe or to treat following gross total resection? Pediatr Blood Cancer 58:380–383. doi:10.1002/pbc.23086
Acknowledgments
The authors have no acknowledgements to report. This study has not been presented elsewhere.
Conflict of interest
Author Paul Nguyen MD has received research funding from Varian and consulted for Ferring Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aizer, A.A., Ancukiewicz, M., Nguyen, P.L. et al. Natural history and role of radiation in patients with supratentorial and infratentorial WHO grade II ependymomas: results from a population-based study. J Neurooncol 115, 411–419 (2013). https://doi.org/10.1007/s11060-013-1237-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1237-9