Abstract
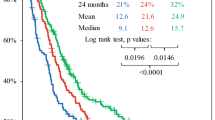
To evaluate in a single center retrospectively the efficacy and tolerability of a weekly regimen, which alternates temozolomide (TMZ) in patients with recurrent or progressive high-grade glioma (HGG). From January 2005 until June 2011, 54 patients with recurrent or progressive HGG were treated with TMZ 150 mg/m²/day on days 1–7 and 15–21 of a 28-day cycle (“one week on–one week off” scheme; TMZ 7/14) with individual dose adjustment depending on toxicity. The majority of patients (n = 48, 89 %) was treated at first tumor recurrence or progression. All patients had received prior radiotherapy with or without concomitantly administered TMZ and, optionally, adjuvant chemotherapy. After initiation of TMZ 7/14, MRI was obtained every 8–12 weeks. Tumor response or progression was assessed according to Macdonald criteria. Blood examinations were performed weekly. Toxicity was evaluated according to Common Terminology Criteria for Adverse Events (CTCAE; version 3.0). A total of 434 treatment weeks with TMZ 7/14 were delivered. The median number of treatment weeks was 7 (range, 1–41 weeks). No grade 4 hematological toxicity and no opportunistic infections occurred. Patients with neutropenia were not observed. Two patients developed grade 3 and 4 patients grade 2 leukocytopenia. Thrombocytopenia grade 3 and grade 2 occurred in 4 patients and 6 patients, respectively. The progression-free survival (PFS) rate at 6 months was 43 %. Median PFS from treatment initiation was 18 weeks (95 % CI, 14–22 weeks) and median overall survival (OS) was 37 weeks (95 % CI, 31–42 weeks). The rates for PFS and OS at 1 year were 24 and 28 %, respectively. Our data suggest that treatment with TMZ 7/14 is safe and effective in patients with recurrent or progressive HGG.
Similar content being viewed by others
References
Easaw JC, Mason WP, Perry J, Laperriere N, Eisenstat DD, Del Maestro R, Belanger K, Fulton D, Macdonald D (2011) Canadian recommendations for the treatment of recurrent or progressive glioblastoma multiforme. Curr Oncol 18:126–136
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Wick W, Hartmann C, Engel C, Stoffels M, Felsberg J, Stockhammer F, Sabel MC, Koeppen S, Ketter R, Meyermann R, Rapp M, Meisner C, Kortmann RD, Pietsch T, Wiestler OD, Ernemann U, Bamberg M, Reifenberger G, von Deimling A, Weller M (2009) NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine, and vincristine or temozolomide. J Clin Oncol 27:5874–5880
Chamberlain MC (2010) Emerging clinical principles on the use of bevacizumab for the treatment of malignant gliomas. Cancer 116:3988–3999
Desjardins A, Reardon DA, Coan A, Marcello J, Herndon JE 2nd, Bailey L, Peters KB, Friedman HS, Vredenburgh JJ (2012) Bevacizumab and daily temozolomide for recurrent glioblastoma. Cancer 118:1302–1312
Wick A, Pascher C, Wick W, Jauch T, Weller M, Bogdahn U, Hau P (2009) Rechallenge with temozolomide in patients with recurrent gliomas. J Neurol 256:734–741
Wick W, Steinbach JP, Kuker WM, Dichgans J, Bamberg M, Weller M (2004) One week on/one week off: a novel active regimen of temozolomide for recurrent glioblastoma. Neurology 62:2113–2115
Wick A, Felsberg J, Steinbach JP, Herrlinger U, Platten M, Blaschke B, Meyermann R, Reifenberger G, Weller M, Wick W (2007) Efficacy and tolerability of temozolomide in an alternating weekly regimen in patients with recurrent glioma. J Clin Oncol 25:3357–3361
Wick W, Weller M (2005) How lymphotoxic is dose-intensified temozolomide? The glioblastoma experience. J Clin Oncol 23:4235–4236
Brandes AA, Tosoni A, Cavallo G, Bertorelle R, Gioia V, Franceschi E, Biscuola M, Blatt V, Crino L, Ermani M (2006) Temozolomide 3 weeks on and 1 week off as first-line therapy for recurrent glioblastoma: phase II study from gruppo italiano cooperativo di neuro-oncologia (GICNO). Br J Cancer 95:1155–1160
Abacioglu U, Caglar HB, Yumuk PF, Akgun Z, Atasoy BM, Sengoz M (2011) Efficacy of protracted dose-dense temozolomide in patients with recurrent high-grade glioma. J Neurooncol 103:585–593
Neyns B, Chaskis C, Joosens E, Menten J, D’Hondt L, Branle F, Sadones J, Michotte A (2008) A multicenter cohort study of dose-dense temozolomide (21 of 28 days) for the treatment of recurrent anaplastic astrocytoma or oligoastrocytoma. Cancer Investig 26:269–277
Perry JR, Belanger K, Mason WP, Fulton D, Kavan P, Easaw J, Shields C, Kirby S, Macdonald DR, Eisenstat DD, Thiessen B, Forsyth P, Pouliot JF (2010) Phase II trial of continuous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. J Clin Oncol 28:2051–2057
Kong DS, Lee JI, Kim JH, Kim ST, Kim WS, Suh YL, Dong SM, Nam DH (2010) Phase II trial of low-dose continuous (metronomic) treatment of temozolomide for recurrent glioblastoma. Neuro Oncol 12:289–296
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003
Esteller M, Garcia-Foncillas J, Andion E, Goodman SN, Hidalgo OF, Vanaclocha V, Baylin SB, Herman JG (2000) Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med 343:1350–1354
Krex D, Klink B, Hartmann C, von Deimling A, Pietsch T, Simon M, Sabel M, Steinbach JP, Heese O, Reifenberger G, Weller M, Schackert G (2007) Long-term survival with glioblastoma multiforme. Brain 130:2596–2606
Tolcher AW, Gerson SL, Denis L, Geyer C, Hammond LA, Patnaik A, Goetz AD, Schwartz G, Edwards T, Reyderman L, Statkevich P, Cutler DL, Rowinsky EK (2003) Marked inactivation of O6-alkylguanine-DNA alkyltransferase activity with protracted temozolomide schedules. Br J Cancer 88:1004–1011
Beier D, Schriefer B, Brawanski K, Hau P, Weis J, Schulz JB, Beier CP (2012) Efficacy of clinically relevant temozolomide dosing schemes in glioblastoma cancer stem cell lines. J Neurooncol 109:45–52
Taal W, Segers-van Rijn JM, Kros JM, van Heuvel I, van der Rijt CC, Bromberg JE, Sillevis Smitt PA, van den Bent MJ (2012) Dose dense 1 week on/1 week off temozolomide in recurrent glioma: a retrospective study. J Neurooncol 108:195–200
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Brandes AA, Ermani M, Basso U, Paris MK, Lumachi F, Berti F, Amista P, Gardiman M, Iuzzolino P, Turazzi S, Monfardini S (2002) Temozolomide in patients with glioblastoma at second relapse after first line nitrosourea-procarbazine failure: a phase II study. Oncology 63:38–41
Brada M, Hoang-Xuan K, Rampling R, Dietrich PY, Dirix LY, Macdonald D, Heimans JJ, Zonnenberg BA, Bravo-Marques JM, Henriksson R, Stupp R, Yue N, Bruner J, Dugan M, Rao S, Zaknoen S (2001) Multicenter phase II trial of temozolomide in patients with glioblastoma multiforme at first relapse. Ann Oncol 12:259–266
Yung WK, Albright RE, Olson J, Fredericks R, Fink K, Prados MD, Brada M, Spence A, Hohl RJ, Shapiro W, Glantz M, Greenberg H, Selker RG, Vick NA, Rampling R, Friedman H, Phillips P, Bruner J, Yue N, Osoba D, Zaknoen S, Levin VA (2000) A phase II study of temozolomide versus procarbazine in patients with glioblastoma multiforme at first relapse. Br J Cancer 83:588–593
Brada M, Stenning S, Gabe R, Thompson LC, Levy D, Rampling R, Erridge S, Saran F, Gattamaneni R, Hopkins K, Beall S, Collins VP, Lee SM (2010) Temozolomide versus procarbazine, lomustine, and vincristine in recurrent high-grade glioma. J Clin Oncol 28:4601–4608
Lee SM, Thatcher N, Dougal M, Margison GP (1993) Dosage and cycle effects of dacarbazine (DTIC) and fotemustine on O6-alkylguanine-DNA alkyltransferase in human peripheral blood mononuclear cells. Br J Cancer 67:216–221
Gilbert MR, Wang M, Aldape KD, Stupp R, Hegi M, Jaeckle KA, Armstrong TS, Wefel JS, Won M, Blumenthal DT, Mahajan A, Schultz CJ, Erridge SC, Brown PD, Chakravarti A, Curran WJ, Mehta MP: RTOG 0525: a randomized phase III trial comparing standard adjuvant temozolomide (TMZ) with a dose-dense (dd) schedule in newly diagnosed glioblastoma (GBM). J Clin Oncol 29: (suppl; abstr 2006), 2011
Friedman HS, Johnson SP, Dong Q, Schold SC, Rasheed BK, Bigner SH, Ali-Osman F, Dolan E, Colvin OM, Houghton P, Germain G, Drummond JT, Keir S, Marcelli S, Bigner DD, Modrich P (1997) Methylator resistance mediated by mismatch repair deficiency in a glioblastoma multiforme xenograft. Cancer Res 57:2933–2936
Maxwell JA, Johnson SP, McLendon RE, Lister DW, Horne KS, Rasheed A, Quinn JA, Ali-Osman F, Friedman AH, Modrich PL, Bigner DD, Friedman HS (2008) Mismatch repair deficiency does not mediate clinical resistance to temozolomide in malignant glioma. Clin Cancer Res 14:4859–4868
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Norbert Galldiks and Theresa Berhorn contributed equally to this work.
Rights and permissions
About this article
Cite this article
Galldiks, N., Berhorn, T., Blau, T. et al. “One week on–one week off”: efficacy and side effects of dose-intensified temozolomide chemotherapy: experiences of a single center. J Neurooncol 112, 209–215 (2013). https://doi.org/10.1007/s11060-013-1048-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1048-z