Abstract
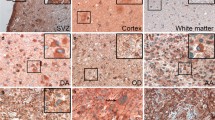
Gliomas are the most common primary brain tumors in adults and have a poor prognosis. Galectin-3 is a β-galactosidase-binding lectin which is important in pre-mRNA splicing, regulation of cell proliferation, cell adhesion and apoptosis. Although galectin-3 has been shown as a glioma related marker and expression of galectin-3 has been reported to correlate with WHO grade in human gliomas, expression of galectin-3 in early neoplastic lesions such as early neoplastic proliferation (ENP) and microtumor is still far from fully understood. In the present study, expression of galectin-3 in ethylnitrosourea-induced rat gliomas including preneoplastic and neoplastic lesions was examined by immunohistochemistry for galectin-3, Iba-1 (a specific microglial cell marker), GFAP (a specific astrocyte cell marker), and conventional hematoxylin and eosin staining (for morphological observation). The results showed that exact location of ENP was detected clearly by galectin-3 immunohistochemistry whereas normal brain tissues were negative. In ENP and microtumor, galectin-3 was expressed in neoplastic astrocytic cells but rarely in microglia. In malignant glioma, however, galectin-3 was expressed in both neoplastic astrocytic cells and microglia. This suggests that galectin-3 is activated in microglia and macrophages according to the progression of glioma. Galectin-3 was not expressed in oligodendrocytic cells. Our results indicate that galectin-3 is a good specific marker indicating the early stage of glioma tumorigenesis.






Similar content being viewed by others
References
Adey WR, Byus CV, Cain CD, Higgins RJ, Jones RA, Kean CJ, Kuster N, MacMurray A, Stagg RB, Zimmerman G (2000) Spontaneous and nitrosourea-induced primary tumors of the central nervous system in Fischer 344 rats exposed to frequency-modulated microwave fields. Cancer Res 60:1857–1863
Bresalier RS, Yan PS, Byrd JC, Lotan R, Raz A (1997) Expression of the endogenous galactose-binding protein galectin-3 correlates with the malignant potential of tumors in the central nervous system. Cancer 80:776–787
Dagher SF, Wang JL, Patterson RJ (1995) Identification of galectin-3 as a factor in pre-mRNA splicing. Proc Natl Acad Sci 92:1213–1217
Deininger MH, Trautmann K, Meyermann R, Schluesener HJ (2002) Galectin-3 labeling correlates positively in tumor cells and negatively in endothelial cells with malignancy and poor prognosis in oligodendroglioma patients. Anticancer Res 22:1585–1592
Fukumori T, Kanayama HO, Raz A (2007) The role of galectin-3 in cancer drug resistance. Drug Resist Update 10:101–108
Fukushi J, Makagiansar IT, Stallcup WB (2004) NG2 proteoglycan promotes endothelial cell motility and angiogenesis via engagement of galectin-3 and alpha 3 beta 1 integrin. Mol Biol Cell 15:3580–3590
Gordower L, Decaestecker C, Kacem Y, Lemmers A, Gusman J, Burchert M (1999) Galectin-3 and galectin-3-binding site expression in human adult astrocytic tumours and related angiogenesis. Neuropathol Appl Neurobiol 25:319–330
Hara A, Yamada H, Sakai N, Hirayama H, Tanaka T, Mori H (1990) Immunohistochemical demonstration of the placental form of glutathione S-transferase, a detoxifying enzyme in human gliomas. Cancer 66:2563–2568
Hara A (1991) Placental form of glutathione S-tranferase a detoxifying enzyme in gliomas. Acta Sch Med Univ Gifu 39:587–597
Koestner A, Swenberg JA, Wechsler W (1971) Transplacental production with ethylnitrosourea of neoplasm of the nervous system in Spraqua–Dawley rat. Am J Pathol 63:37–56
Kuwabara I, Liu FT (1996) Galectin-3 promotes adhesion of human neutrophils to laminin. J Immunol 156:3939–3944
Le Mercier M, Fortin S, Mathieu V, Kiss R, Lefranc F (2009) Galectins and gliomas. Brain Pathol 20:17–27
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (2007) WHO classification of tumours of the central nervous system. International Agency for Research on Cancer (IARC), Lyon
Oberto G, Rolfo K, Yu P, Carbonatto M, Peano S, Kuster N, Ebert S, Tofani S (2007) Carcinogenicity study of 217 Hz pulsed 900 MHz electromagnetic fields in Pim1 transgenic mice. Radiat Res 168:316–326
Ohgaki H, Kleihues P (2005) Epidemiology and etiology of gliomas. Acta Neuropathol 109:93–108
Preusser M, de Ribaupierre S, Wöhrer A, Erridge SC, Hegi M, Weller M, Stupp R (2011) Current concepts and management of glioblastoma. Ann Neurol 70:9–21
Reifenberger G, Bilzer T, Seitz RJ, Wechsler W (1989) Expression of vimentin and glial fibrillary acidic protein in ethylnitrosourea-induced rat gliomas and glioma cell lines. Acta Neuropathol 78:270–282
Satoh K, Niwa M, Goda W, Binh NH, Nakashima M, Takamatsu M, Hara A (2011) Galectin-3 expression in delayed neuronal death of hippocampal CA1 following transient forebrain ischemia, and its inhibition by hypothermia. Brain Res 1382:266–274
Strik HM, Deininger MH, Frank B, Schluesener HJ, Meyermann R (2001) Galectin-3: cellular distribution and correlation with WHO-grade in human gliomas. J Neurooncol 53:13–20
Vladimirova V, Waha A, Lückerath K, Pesheva P, Probstmeier R (2008) Runx2 is expressed in human glioma cells and mediates the expression of galectin-3. J Neurosci Res 86:2450–2461
Wei J, Barr J, Kong LY, Wang Y, Wu A, Sharma AK, Gumin J, Henry V, Colman H, Sawaya R, Lang FF, Heimberger AB (2010) Glioma-associated cancer-initiating cells induce immunosuppression. Clin Cancer Res 16:461–473
Yang RY, Hsu DK, Liu FT (1996) Expression of galectin-3 modulates T-cell growth and apoptosis. Proc Natl Acad Sci USA 93:6737–6742
Zeng Y, Danielson KG, Albert TJ, Shapiro IM, Risbud MV (2007) HIF-1 alpha is a regulator of galectin-3 expression in the intervertebral disc. J Bone Miner Res 22:1851–1861
Zook BC, Simmens SJ, Jones RV (2000) Evaluation of ENU-induced gliomas in rats: nomenclature, immunochemistry, and malignancy. Toxicol Pathol 28:193–201
Acknowledgments
The authors are indebted to Ms. Kyoko Takahashi for her excellent technical assistance. This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Ethical standards
We fully complied with the ‘Guidelines Concerning Experimental Animals’ issued by the Japanese Association for Laboratory Animal Science and exercised due consideration so as not to cause any ethical problems.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Binh, N.H., Satoh, K., Kobayashi, K. et al. Galectin-3 in preneoplastic lesions of glioma. J Neurooncol 111, 123–132 (2013). https://doi.org/10.1007/s11060-012-1005-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-012-1005-2