Abstract
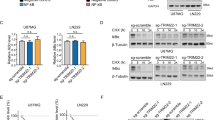
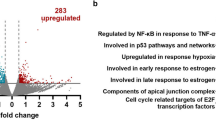
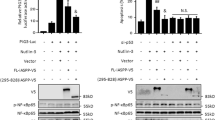
Malignant gliomas are diffusively infiltrative and remain among the deadliest of all cancers. NF-κB is a transcription factor that mediates cell growth, migration and invasion, angiogenesis and resistance to apoptosis. Normally, the activity of NF-κB is tightly regulated by numerous mechanisms. However, in many cancers, NF-κB is constitutively activated and may function as a tumor promoter. Herein, we show that in gliomas, NF-κB is constitutively activated and the levels of cIAP2, Bcl-2, Bcl-xL and Survivin are elevated. These genes are regulated by NF-κB and can inhibit apoptosis. To understand the potential role of NF-κB p65 in suppressing apoptosis, we generated human glioma cell lines that inducibly express shRNA molecules specific for p65. We demonstrate that in the absence of p65, TNF-α induced cIAP2 expression is significantly reduced while the levels of Bcl-2, Bcl-xL and Survivin are not affected. These data suggest that of these genes, only cIAP2 is a direct target of p65, which was confirmed using RT-PCR and chromatin immunoprecipitation (ChIP) assays. By reducing the levels of p65 and/or cIAP2 levels, we demonstrate that the levels of RIP poly-ubiquitination are reduced, and that p65-deficient glioma cells are more sensitive to the cytotoxic effects of TNF-α. Specifically, in the presence of TNF-α glioma cells lacking p65 and/or cIAP2 showed cellular proliferation defects and underwent cell death. These data suggest that NF-κB and/or cIAP2 may be therapeutically relevant targets for the treatment of malignant gliomas.







Similar content being viewed by others
References
Naugler WE, Karin M (2008) NF-κB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev 18:19–26
Meffert MK, Baltimore D (2005) Physiological functions for brain NF-κB. Trends Neurosci 28:37–43
Widera D, Kaus A, Kaltschmidt C, Kaltschmidt B (2008) Neural stem cells, inflammation and NF-κB: basic principle of maintenance and repair or origin of brain tumours? J Cell Mol Med 12:459–470
Widera D, Mikenberg I, Kaltschmidt B, Kaltschmidt C (2006) Potential role of NF-κB in adult neural stem cells: the underrated steersman? Int J Dev Neurosci 24:91–102
Gilmore T (2008) www.NF-κB.org
Furnari FB, Fenton T, Bachoo RM, Mukasa A, Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, Chin L, DePinho RA, Cavenee WK (2007) Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev 21:2683–2710
Maher EA, Furnari FB, Bachoo RM, Rowitch DH, Louis DN, Cavenee WK, DePinho RA (2001) Malignant glioma: genetics and biology of a grave matter. Genes Dev 15:1311–1333
Reifenberger G, Collins VP (2004) Pathology and molecular genetics of astrocytic gliomas. J Mol Med 82:656–670
van den Bent MJ (2006) Adjuvant treatment of high grade gliomas. Ann Oncol 17(Suppl 10):x186–x190
Schiff D (2007) Temozolomide and radiation in low-grade and anaplastic gliomas: temoradiation. Cancer Invest 25:776–784
Garkavtsev I, Kozin SV, Chernova O, Xu L, Winkler F, Brown E, Barnett GH, Jain RK (2004) The candidate tumour suppressor protein ING4 regulates brain tumour growth and angiogenesis. Nature 428:328–332
Nozell S, Laver T, Moseley D, Nowoslawski L, De Vos M, Atkinson GP, Harrison K, Nabors LB, Benveniste EN (2008) The ING4 tumor suppressor attenuates NF-κB activity at the promoters of target genes. Mol Cell Biol 28:6632–6645
Wang H, Zhang W, Huang HJ, Liao WS, Fuller GN (2004) Analysis of the activation status of Akt, NFκB, and Stat3 in human diffuse gliomas. Lab Invest 84:941–951
Angileri FF, Aguennouz M, Conti A, La Torre D, Cardali S, Crupi R, Tomasello C, Germano A, Vita G, Tomasello F (2008) NF-κB activation and differential expression of survivin and Bcl-2 in human grade 2–4 astrocytomas. Cancer 112:2258–2266
Korkolopoulou P, Levidou G, Saetta AA, El-Habr E, Eftichiadis C, Demenagas P, Thymara I, Xiromeritis K, Boviatsis E, Thomas-Tsagli E, Panayotidis I, Patsouris E (2008) Expression of nuclear factor-κB in human astrocytomas: relation to pI kappa Bα, vascular endothelial growth factor, Cox-2, microvascular characteristics, and survival. Hum Pathol 39:1143–1152
Robe PA, Bentires-Alj M, Bonif M, Rogister B, Deprez M, Haddada H, Khac MT, Jolois O, Erkmen K, Merville MP, Black PM, Bours V (2004) In vitro and in vivo activity of the nuclear factor-κB inhibitor sulfasalazine in human glioblastomas. Clin Cancer Res 10:5595–5603
Otsuka G, Nagaya T, Saito K, Mizuno M, Yoshida J, Seo H (1999) Inhibition of NF-κB activation confers sensitivity to tumor necrosis factor-alpha by impairment of cell cycle progression in human glioma cells. Cancer Res 59:4446–4452
Moriuchi S, Glorioso JC, Maruno M, Izumoto S, Wolfe D, Huang S, Cohen JB, Yoshimine T (2005) Combination gene therapy for glioblastoma involving herpes simplex virus vector-mediated codelivery of mutant IκBα and HSV thymidine kinase. Cancer Gene Ther 12:487–496
Weaver KD, Yeyeodu S, Cusack JC Jr, Baldwin AS Jr, Ewend MG (2003) Potentiation of chemotherapeutic agents following antagonism of NF-κB in human gliomas. J Neurooncol 61:187–196
Dhandapani KM, Mahesh VB, Brann DW (2007) Curcumin suppresses growth and chemoresistance of human glioblastoma cells via AP-1 and NF-κB transcription factors. J Neurochem 102:522–538
Yamini B, Yu X, Dolan ME, Wu MH, Kufe DW, Weichselbaum RR (2007) Inhibition of NF-κB activity by temozolomide involves O6-methylguanine induced inhibition of p65 DNA binding. Cancer Res 67:6889–6898
Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS Jr (1998) NF-κB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science 281:1680–1683
Chu ZL, McKinsey TA, Liu L, Gentry JJ, Malim MH, Ballard DW (1997) Suppression of tumor necrosis factor-induced cell death by inhibitor of apoptosis c-IAP2 is under NF-κB control. Proc Natl Acad Sci USA 94:10057–10062
Varfolomeev E, Vucic D (2008) (Un)expected roles of c-IAPs in apoptotic and NFκB signaling pathways. Cell Cycle 7:1511–1521
Cao L, Wang Z, Yang X, Xie L, Yu L (2008) The evolution of BIR domain and its containing proteins. FEBS Lett 582:3817–3822
Eckelman BP, Salvesen GS (2006) The human anti-apoptotic proteins cIAP1 and cIAP2 bind but do not inhibit caspases. J Biol Chem 281:3254–3260
Shiozaki EN, Chai J, Rigotti DJ, Riedl SJ, Li P, Srinivasula SM, Alnemri ES, Fairman R, Shi Y (2003) Mechanism of XIAP-mediated inhibition of caspase-9. Mol Cell 11:519–527
Srinivasula SM, Hegde R, Saleh A, Datta P, Shiozaki E, Chai J, Lee RA, Robbins PD, Fernandes-Alnemri T, Shi Y, Alnemri ES (2001) A conserved XIAP-interaction motif in caspase-9 and Smac/DIABLO regulates caspase activity and apoptosis. Nature 410:112–116
Bertrand MJ, Milutinovic S, Dickson KM, Ho WC, Boudreault A, Durkin J, Gillard JW, Jaquith JB, Morris SJ, Barker PA (2008) cIAP1 and cIAP2 facilitate cancer cell survival by functioning as E3 ligases that promote RIP1 ubiquitination. Mol Cell 30:689–700
Declercq W, Vanden Berghe T, Vandenabeele P (2009) RIP kinases at the crossroads of cell death and survival. Cell 138:229–232
Varfolomeev E, Goncharov T, Fedorova AV, Dynek JN, Zobel K, Deshayes K, Fairbrother WJ, Vucic D (2008) c-IAP1 and c-IAP2 are critical mediators of tumor necrosis factor alpha (TNFalpha)-induced NF-κB activation. J Biol Chem 283:24295–24299
Choi C, Kutsch O, Park J, Zhou T, Seol DW, Benveniste EN (2002) Tumor necrosis factor-related apoptosis-inducing ligand induces caspase-dependent interleukin-8 expression and apoptosis in human astroglioma cells. Mol Cell Biol 22:724–736
Nozell S, Laver T, Patel K, Benveniste EN (2006) Mechanism of IFN-beta-mediated inhibition of IL-8 gene expression in astroglioma cells. J Immunol 177:822–830
Ma Z, Qin H, Benveniste EN (2001) Transcriptional suppression of matrix metalloproteinase-9 gene expression by IFN-gamma and IFN-beta: critical role of STAT-1alpha. J Immunol 167:5150–5159
Nozell S, Chen X (2002) p21B, a variant of p21(Waf1/Cip1), is induced by the p53 family. Oncogene 21:1285–1294
Nozell S, Wu Y, McNaughton K, Liu G, Willis A, Paik JC, Chen X (2003) Characterization of p73 functional domains necessary for transactivation and growth suppression. Oncogene 22:4333–4347
Hayden MS, Ghosh S (2008) Shared principles in NF-κB signaling. Cell 132:344–362
Catz SD, Johnson JL (2001) Transcriptional regulation of bcl-2 by nuclear factor κB and its significance in prostate cancer. Oncogene 20:7342–7351
Chen C, Edelstein LC, Gelinas C (2000) The Rel/NF-κB family directly activates expression of the apoptosis inhibitor Bcl-x(L). Mol Cell Biol 20:2687–2695
Chen F, Demers LM, Vallyathan V, Lu Y, Castranova V, Shi X (1999) Involvement of 5′-flanking κB-like sites within bcl-x gene in silica-induced Bcl-x expression. J Biol Chem 274:35591–35595
Tracey L, Perez-Rosado A, Artiga MJ, Camacho FI, Rodriguez A, Martinez N, Ruiz-Ballesteros E, Mollejo M, Martinez B, Cuadros M, Garcia JF, Lawler M, Piris MA (2005) Expression of the NF-κB targets BCL2 and BIRC5/Survivin characterizes small B-cell and aggressive B-cell lymphomas, respectively. J Pathol 206:123–134
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, Barrette T, Pandey A, Chinnaiyan AM (2004) ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6:1–6
Rhodes DR, Kalyana-Sundaram S, Mahavisno V, Varambally R, Yu J, Briggs BB, Barrette TR, Anstet MJ, Kincead-Beal C, Kulkarni P, Varambally S, Ghosh D, Chinnaiyan AM (2007) Oncomine 3.0: genes, pathways, and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia 9:166–180
Loveland BE, Johns TG, Mackay IR, Vaillant F, Wang ZX, Hertzog PJ (1992) Validation of the MTT dye assay for enumeration of cells in proliferative and antiproliferative assays. Biochem Int 27:501–510
Lin WW, Karin M (2007) A cytokine-mediated link between innate immunity, inflammation, and cancer. J Clin Invest 117:1175–1183
Li X, Cai L, Chen H, Zhang Q, Zhang S, Wang Y, Dong Y, Cheng H, Qi J (2009) Inhibitor of growth 4 induces growth suppression and apoptosis in glioma U87MG. Pathobiology 76:181–192
McCoy MK, Tansey MG (2008) TNF signaling inhibition in the CNS: implications for normal brain function and neurodegenerative disease. J Neuroinflammation 5:45.13–45.41
Hayashi S, Yamamoto M, Ueno Y, Ikeda K, Ohshima K, Soma G, Fukushima T (2001) Expression of nuclear factor-κB, tumor necrosis factor receptor type 1, and c-Myc in human astrocytomas. Neurol Med Chir (Tokyo) 41:187–195
Kargiotis O, Rao JS, Kyritsis AP (2006) Mechanisms of angiogenesis in gliomas. J Neurooncol 78:281–293
Moore PA, Belvedere O, Orr A, Pieri K, LaFleur DW, Feng P, Soppet D, Charters M, Gentz R, Parmelee D, Li Y, Galperina O, Giri J, Roschke V, Nardelli B, Carrell J, Sosnovtseva S, Greenfield W, Ruben SM, Olsen HS, Fikes J, Hilbert DM (1999) BLyS: member of the tumor necrosis factor family and B lymphocyte stimulator. Science 285:260–263
Sudheerkumar P, Shiras A, Das G, Jagtap JC, Prasad V, Shastry P (2008) Independent activation of Akt and NF-κB pathways and their role in resistance to TNF-alpha mediated cytotoxicity in gliomas. Mol Carcinog 47:126–136
Wilson J, Balkwill F (2002) The role of cytokines in the epithelial cancer microenvironment. Semin Cancer Biol 12:113–120
Atkinson GP, Nozell SE, Harrison DK, Stonecypher MS, Chen D, Benveniste EN (2009) The prolyl isomerase Pin1 regulates the NF-κB signaling pathway and interleukin-8 expression in glioblastoma. Oncogene 28:3735–3745
Bao L, Kimzey A, Sauter G, Sowadski JM, Lu KP, Wang DG (2004) Prevalent overexpression of prolyl isomerase Pin1 in human cancers. Am J Pathol 164:1727–1737
Atkinson GP, Nozell SE, Benveniste ET (2010) NF-κB and STAT3 signaling in glioma: targets for future therapies. Expert Rev Neurother 10:575–586
Li L, Thomas RM, Suzuki H, De Brabander JK, Wang X, Harran PG (2004) A small molecule Smac mimic potentiates TRAIL- and TNF-α-mediated cell death. Science 305:1471–1474
Li L, Gondi CS, Dinh DH, Olivero WC, Gujrati M, Rao JS (2007) Transfection with anti-p65 intrabody suppresses invasion and angiogenesis in glioma cells by blocking NF-κB transcriptional activity. Clin Cancer Res 13:2178–2190
Tsuboi Y, Kurimoto M, Nagai S, Hayakawa Y, Kamiyama H, Hayashi N, Kitajima I, Endo S (2009) Induction of autophagic cell death and radiosensitization by the pharmacological inhibition of nuclear factor-κB activation in human glioma cell lines. J Neurosurg 110:594–604
Kuwayama K, Matsuzaki K, Mizobuchi Y, Mure H, Kitazato KT, Kageji T, Nakao M, Nagahiro S (2009) Promyelocytic leukemia protein induces apoptosis due to caspase-8 activation via the repression of NFκB activation in glioblastoma. Neuro Oncol 11:132–141
Zheng M, Morgan-Lappe SE, Yang J, Bockbrader KM, Pamarthy D, Thomas D, Fesik SW, Sun Y (2008) Growth inhibition and radiosensitization of glioblastoma and lung cancer cells by small interfering RNA silencing of tumor necrosis factor receptor-associated factor 2. Cancer Res 68:7570–7578
Sharma V, Tewari R, Sk UH, Joseph C, Sen E (2008) Ebselen sensitizes glioblastoma cells to Tumor Necrosis Factor (TNFalpha)-induced apoptosis through two distinct pathways involving NF-κB downregulation and Fas-mediated formation of death inducing signaling complex. Int J Cancer 123:2204–2212
Jiang W, Cazacu S, Xiang C, Zenklusen JC, Fine HA, Berens M, Armstrong B, Brodie C, Mikkelsen T (2008) FK506 binding protein mediates glioma cell growth and sensitivity to rapamycin treatment by regulating NF-κB signaling pathway. Neoplasia 10:235–243
Ichiyama T, Nishikawa M, Lipton JM, Matsubara T, Takashi H, Furukawa S (2001) Thiopental inhibits NF-κB activation in human glioma cells and experimental brain inflammation. Brain Res 911:56–61
LaCasse EC, Mahoney DJ, Cheung HH, Plenchette S, Baird S, Korneluk RG (2008) IAP-targeted therapies for cancer. Oncogene 27:6252–6275
Acknowledgments
This work was supported in part by Public Service Grants CA-97247 from the National Cancer Institute (E. N. B.), NS-50665 from the National Institute of Neurological Disorders and Stroke (E. N. B.), CA-13148-35 from the National Cancer Institute (E. N. B.), 5P30CA013149-38 from the UAB Comprehensive Cancer Center (E. N. B.), IRG-60-001-47 from the American Cancer Society (S. N.), CA-13148-31 from the National Cancer Institute (S. N.), and funding from the Southeastern Brain Tumor Foundation (S. N.). We thank Dr. Xinbin Chen for providing the pBABE-HI-TetO plasmid, Dr. Tong Zhou for providing antibodies specific for Bcl-2 and Bcl-xL, and Dr. G. Yancey Gillespie and the UAB Brain Tumor Tissue Core for providing the brain tissue samples used herein. We also thank Maria G. Salazar of the CFAR/CCC DNA Sequencing Core at UAB for expert advice and technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xueyan Zhao and Travis Laver contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, X., Laver, T., Hong, S.W. et al. An NF-κB p65-cIAP2 link is necessary for mediating resistance to TNF-α induced cell death in gliomas. J Neurooncol 102, 367–381 (2011). https://doi.org/10.1007/s11060-010-0346-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-010-0346-y