Abstract
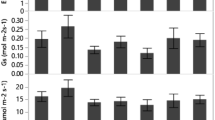
Drought is often an important constraint on plant regeneration in the Mediterranean. Pistacia lentiscus is of particular interest in restoration actions in fire-prone areas. This study addressed the current knowledge gap on the physiological response of this species to drought during its initial development. We hypothesized that the seedlings would have a high resistance to drought. In addition, we expected that seedlings from two sources with contrasting climate conditions would perform distinctly under drought. Seeds were collected from two populations in north and south Portugal. Seedlings were subjected to a progressively lower osmotic potential during 25 days, through increasing PEG concentration in the hydroponic solution. The physiological response of the seedlings was assessed by measuring gas exchange and chlorophyll fluorescence after 15 and 25 days of drought exposure, when the osmotic potential was −2.47 and −5.17 MPa, respectively. Shoot growth, net CO2 assimilation rate, stomatal conductance and transpiration were significantly reduced under drought conditions, whilst intrinsic water-use efficiency (IWUE) remained unaffected. This response partially agreed with the known strategy of P. lentiscus to cope with drought, typically maintaining high IWUE, however the observed reduction in stomatal conductance was not as pronounced as expected. Drought induced a decrease in leaf pigments that could be linked to photo-protective mechanisms. Seed source did not have a significant effect on drought response. P. lentiscus showed a high resistance to drought, which clearly supported its use in restoration actions in the Mediterranean, but these findings require further testing under field conditions.

Similar content being viewed by others
Abbreviations
- A:
-
Net CO2 assimilation rate
- E:
-
Transpiration rate
- gs :
-
Stomatal conductance
- Ci/Ca :
-
Ratio of intercellular to atmospheric CO2 concentration
- IWUE:
-
Intrinsic water use efficiency
- Fv/Fm :
-
Maximum photochemical efficiency of PSII
- qP :
-
Photochemical quenching
- ΦPSII :
-
Effective quantum efficiency of PSII
- NPQ:
-
Non-photochemical quenching
- WC:
-
Water content
- CT:
-
Control
- D:
-
Drought
- OP:
-
Osmotic potential
- MW:
-
Mann–Whitney
- KW:
-
Kruskal–Wallis
References
Bacelar EA, Moutinho-Pereira JM, Gonçalves B, Ferreira H, Correia CM (2007) Changes in growth, gas exchange, xylem hydraulic properties and water use efficiency of three olive cultivars under contrasting water availability regimes. Environ Exp Bot 60:183–192
Bacelar E, Moutinho-Pereira JM, Gonçalves B, Brito C, Gomes-Laranjo J, Ferreira H, Correia CM (2012) Water use strategies of plants under drought conditions. In: Aroca Ricardo (ed) Plant responses to drought stress: from morphological to molecular features. Springer, Berlin, pp 145–170
Baeza MJ, Santana VM, Pausas JG, Vallejo VR (2011) Successional trends in standing dead biomass in Mediterranean basin species. J Veg Sci 22:467–474
Björkman O, Demmig-Adams B (1994) Regulation of photosynthetic light energy capture, conversion, and dissipation in leaves of higher plants. In: Schulze E-D, Caldwell MM (eds) Ecophysiology of Photosynthesis. Springer, Berlin, p 1747
Correia O, Barradas MD (2000) Ecophysiological differences between male and female plants of Pistacia lentiscus L. Plant Ecol 149:131–142
Cortina J, Green JJ, Baddeley JA, Watson CA (2008) Root morphology and water transport of Pistacia lentiscus seedlings under contrasting water supply: a test of the pipe stem theory. Environ Exp Bot 62:343–350
Filella I, Lusia J, Piñol J, Peñuelas J (1998) Pistacia lentiscus and Quercus ilex saplings in severe drought and high temperature conditions. Environ Exp Bot 39:213–220
Flexas J, Gulías J, Jonasson S, Medrano H, Mus M (2001) Seasonal patterns and control of gas exchange in local populations of the Mediterranean evergreen shrub Pistacia lentiscus L. Acta Oecol 22:33–43
Galmés J, Abadía A, Cifre J, Medrano H, Flexas J (2007) Photoprotection processes under water stress and recovery in Mediterranean plants with different growth forms and leaf habits. Physiol Plant 130:495–510
García-Fayos P (2001) Bases ecológicas para la recolección, almacenamiento y germinación de semillas de especies de uso forestal en la Comunidad Valenciana. Generalitat Valenciana, Valencia (Book ID: 6170)
García-Fayos P, Verdú M (1998) Soil seed bank factors controlling germination and establishment of a Mediterranean shrub: Pistacia lentiscus L. Acta Oecol 19:357–366
Gratani L, Varone L (2004) Adaptive photosynthetic strategies of the Mediterranean maquis species according to their origin. Photosynthetica 42:551–558
Gúlias J, Flexas J, Abadía A, Madrano H (2002) Photosynthetic responses to water deficit in six Mediterranean sclerophyll species: possible factors explaining the declining distribution of Rhamnus ludovici-salvatoris, an endemic Balearic species. Tree Physiol 22:687–697
IPCC (2013) Working group I: the physical basis of climate change. Cambridge University Press, Cambridge
IPMA (Portuguese Institute of the Sea and Atmosphere). https://www.ipma.pt/pt/oclima/normais.clima/ (Last accessed Aug 2013)
Jordano P (1989) Pre-dispersal biology of Pistacia lentiscus (Anacardiaceae): cumulative effects on seed removal by birds. Oikos 55:375–386
Keeley JE, Bond WJ, Bradstock RA, Pausas JG, Rundel PW (2012) Fire in Mediterranean ecosystems. Cambridge University Press, New York, p 515
Kyparissis A, Petropoulou Y, Manetas Y (1995) Summer survival of leaves in a soft-leaved shrub (Phlomis fruticosa L., Labiatae) under Mediterranean field conditions: avoidance of photoinhibitory damage through decreased chlorophyll contents. J Exp Bot 46:1825–1831
Liodakis S, Kakardakis T (2008) Measuring the relative particle foliar combustibility of WUI forest species located near Athens. J Therm Anal Calorim 93:627–635
Maestre FT, Cortina J, Bautista S, Bellot J, Vallejo R (2003) Small-scale environmental heterogeneity and spatiotemporal dynamics of seedling establishment in a semiarid degraded ecosystem. Ecosystems 6:630–643
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Michel BE, Kaufmann MR (1973) The osmotic potential of polyethylene glycol 6000. Plant Physiol 51:914–916
Monteiro M, Santos C, Mann RM, Soares AM, Lopes T (2007) Evaluation of cadmium genotoxicity in Lactuca sativa L. using nuclear microsatellites. Environ Exp Bot 60:421–427
Moreno-Gutiérrez C, Dawson TE, Nicolás E, Querejeta JI (2012) Isotopes reveal contrasting water use strategies among coexisting plant species in a Mediterranean ecosystem. New Phytol 196:489–496
Moutinho-Pereira JM, Correia CM, Gonçalves B, Bacelar EL, Torres-Pereira JM (2004) Leaf gas-exchange and water relations of grapevines grown in three different conditions. Photosynthetica 42:81–86
Murata N, Takahashi S, Nishiyama Y, Allakhverdiev SI (2007) Photoinhibition of photosystem II under environmental stress. Biochim et Biophys Acta (BBA)-Bioenerg, 1767(6):414-421
Osmond CB, Anderson JM, Ball MC, & Egerton JJG (1999) Compromising efficiency: the molecular ecology of light-resource utilization in plants. Physiol plant ecol. Blackwell Science Ltd, Oxford, 1–25.
Paula S, Pausas JG (2008) Burning seeds: germinative response to heat treatments in relation to resprouting ability. J Ecol 96:543–552
Pausas J, Llovet J, Rodrigo A, Vallejo R (2008) Are wildfires a disaster in the Mediterranean basin? A review. Int J Wildland Fire 17:713–723
Santos C, Fragoeiro S, Phillips A (2005) Physiological response of grapevine cultivars and a rootstock to infection with Phaeoacremonium and Phaeomoniella isolates: an in vitro approach using plants and calluses. Sci Hortic 103:187–198
Sims DA, Gamon JÁ (2002) Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens Environ 81:337–354
Tsakaldimi M, Ganatsas P (2001) Treatments improving seed germination of two Mediterranean sclerophyll species: Ceratonia siliqua and Pistacia lentiscus. In: Proceedings of the 3rd Balcan scientific conference, vol II, pp 119–127. ISBN: 954-90896-3-0
Valladares F, Sánchez-Gómez D (2006) Ecophysiological traits associated with drought in Mediterranean tree seedlings: individual responses versus interspecific trends in eleven species. Plant Biol 8:688–697
Vallejo VR, Smanis A, Chirino E, Fuentes D, Valdecantos A, Vilagrosa A (2012) Perspectives in dryland restoration: approaches for climate change adaptation. New For 43:561–579
Vasques A, Chirino E, Vilagrosa A, Vallejo VR, Keizer JJ (2013) The role of seed provenance in the early development of Arbutus unedo seedlings under contrasting watering conditions. Environ Exp Bot 96:11–19
Vasques A, Vallejo VR, Santos C, Keizer JJ (2014) The role of cold storage and seed source in the germination of three Mediterranean shrub species with contrasting dormancy types. Ann For Sci. doi:10.1007/s13595-014-0395-z
Verdú M, García-Fayos P (1998a) Female biased sex ratios in Pistacia lentiscus L. (Anacardiaceae). Plant Ecol 135:95–101
Verdú M, García-Fayos P (1998b) Ecological causes, function, and evolution of abortion and parthenocarpy in Pistacia lentiscus (Anacardiaceae). Can J Bot 76:134–141
Verdú M, García-Fayos P (2000) The effect of deceptive fruits on predispersal seed predation by birds in Pistacia lentiscus. Plant Ecol 156(2):245–248
Vilagrosa A, Morales F, Abadía A, Bellot J, Cochard H, Gil-Pelegrin E (2010) Are symplast tolerance to intense drought conditions and xylem vulnerability to cavitation coordinated? An integrated analysis of photosynthetic, hydraulic and leaf level processes in two Mediterranean drought-resistant species. Environ Exp Bot 69:233–242
Vilagrosa A, Hernández EI, Luis VC, Cochard H, Pausas JG (2014) Physiological differences explain the co-existence of different regeneration strategies in Mediterranean ecosystems. New Phytol 201:1277–1288
Zohary M (1952) A monographical study of the genus Pistacia. Palest J Bot Jerusalem Ser 5:187–228
Zunzunegui M, Barradas MCD, Ain-Lhout F, Alvarez-Cansino L, Esquivias MP, Novo FG (2011) Seasonal physiological plasticity and recovery capacity after summer stress in Mediterranean scrub communities. Plant Ecol 212:127–142
Acknowledgments
The authors would like to thank Diana Vieira, Xufeng Shen, Carlota Vaz and Mariana Pedro for their kind help provided in seed harvesting and to Jingxian Tao, Armando Costa, Cristina Monteiro, Paula Maia and Nelson Abrantes for their laboratory support. We are also very grateful to Banc de Llavors Forestalls de Valencia (Spain) for the indications on the seed handling protocols and to the reviewers and editors for their valuable comments. This study was funded by the Portuguese Foundation for Science and Technology (FCT) and QREN through the PhD grant of the first author (SFRH/BD/47522/2008) and the post-doctoral fellowship of M.C. Dias (SFRH/BPD/41700/2007). The post-doctoral fellowship of G. Pinto (BPD/UI88/2154/2014) was funded by Centro de Estudos do Ambiente e Mar-CESAM- INCENTIVO. The review process of this manuscript received funding from the European Union Seventh Framework Programme (FP7/2007-2013) under Grant Agreement No. 283068 (CASCADE project).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vasques, A.R., Pinto, G., Dias, M.C. et al. Physiological response to drought in seedlings of Pistacia lentiscus (mastic tree). New Forests 47, 119–130 (2016). https://doi.org/10.1007/s11056-015-9497-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-015-9497-1