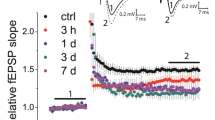
The aim of the present work was to study the dynamics of presynaptic modification in conditions of blockade of GABA receptor NR2B subunits, which are key for consolidation of LTP. This was addressed by studying the developmental features of NMDA-dependent long-term potentiation (LTP) in field CA1 of living rat hippocampal slices incubated with ifenprodil by testing with paired-pulse stimuli throughout the recording period before and for 1 h after high-frequency stimulation of Schaffer collaterals (100 Hz, 1 sec). Perfusion of slices with ifenprodil had no effect on the baseline characteristics of CA3–CA1 synapses in terms of the relationship between the amplitudes of the first and second responses and stimulus intensity, i.e., paired-pulse facilitation. LTP was signifi cantly less marked in slices incubated with ifenprodil, while potentiation of the second response in the pair was essentially the same in the control and experimental groups. In control slices, potentiation was accompanied by a significant decrease in PPF immediately after tetanization. This effect was not seen in slices incubated with ifenprodil; furthermore, a transient increase in PPF was seen 15 min after tetanization. In both groups, early LTP was increased proportionately to the decrease in PPF. In the absence of presynaptic modifications, LTP was weakened, including in control slices, and an increase in PPF in experimental slices was accompanied by temporary depression of focal potentials. Ifenprodil had no obvious effect on the maintenance of LTP. It is suggested that detection of the adverse influence of ifenprodil on the process of consolidation requires complex analysis taking account of all the factors operating. The results obtained here support a tight link between LTP and modifications of pairedpulse plasticity and provide grounds for suggesting that NMDA receptor NR2B subunits are directly related to the involvement of the presynaptic component in long-term synaptic potentiation.
Similar content being viewed by others
References
Barcomb, K., Hell, J. W., Benke, T. A., and Bayer, K. U., “The CaMKII/GluN2B protein interaction maintains synaptic strength,” J. Biol. Chem., 291, No. 31, 16,082–16,089 (2016).
Barria, A. and Malinow, R., “NMDA receptor subunit composition controls synaptic plasticity by regulating binding to CaMKII,” Neuron, 48, 289–301 (2005).
Bartlett, T. E., Bannister, N. J., Collett, V. J., et al., “Differential roles of NR2A and NR2B-containing NMDA receptors in LTP and LTD in the CA1 region of two-week old rat hippocampus,” Neuropharmacology, 52, 60–70 (2007).
Bayazitov, I. T., Richardson, R. J., Fricke, R. G., and Zakharenko, S. S., “Slow presynaptic and fast postsynaptic components of compound long-term potentiation,” J. Neurosci., 27, No. 43, 11,510–11,521 (2007).
Blundon, J. A. and Zakharenko, S. S., “Dissecting the components of longterm potentiation,” Neuroscientist, 14, No. 6, 598–608 (2008).
Chavis, P. and Westbrook, G., “Integrins mediate functional pre- and postsynaptic maturation at a hippocampal synapse,” Nature, 411, 317– 321 (2001).
Coultrap, S. J. and Bayer, K. U., “CaMKII regulation in information processing and storage,” Trends Neurosci., 35, No. 10, 607–618 (2012).
Emptage, N. J., Reid, C. A., Fine, A., and Bliss, T. V., “Optical quantal analysis reveals a presynaptic component of LTP at hippocampal Schaffer-associational synapses,” Neuron, 38, 797–804 (2003).
Enoki, R., Hu, Y.-L., Hamilton, D., and Fine, A., “Expression of long-term plasticity at individual synapses in hippocampus is graded, bidirectional, and mainly presynaptic: optical quantal analysis,” Neuron, 62, 242–253 (2009).
Fioravante, D. and Regehr, W. G., “Short-term forms of presynaptic plasticity,” Curr. Opin. Neurobiol., 21, 269–274 (2011).
Foster, K. A., McLaughlin, N., Edbauer, D., et al., “Distinct roles of NR2A and NR2B cytoplasmic tails in long-term potentiation,” J. Neurosci., 30, No. 7, 2676–2685 (2010).
Fox, C. J., Russell, K. I., Wang, Y. T., and Christie, B. R., “Contribution of NR2A and NR2B NMDA subunits to bidirectional synaptic plasticity in the hippocampus in vivo,” Hippocampus, 16, No. 11, 907–915 (2006).
Gambrill, A. C. and Barria, A., “NMDA receptor subunit composition controls synaptogenesis and synapse stabilization,” Proc. Natl. Acad. Sci. USA, 108, No. 14, 5855–5860 (2011).
Gardoni, F., Mauceri, D., Malinverno, M., et al., “Decreased NR2B subunit synaptic levels cause impaired long-term potentiation but not longterm depression,” J. Neurosci., 29, 669–677 (2009).
Ge, Y., Dong, Z., Bagot, R. C., et al., “Hippocampal long-term depression is required for the consolidation of spatial memory,” Proc. Natl. Acad. Sci. USA, 107, No. 38, 16,697–16,702 (2010).
Halt, A. R., Dallapiazza, R. F., Zhou, Y., et al., “CaMKII binding to GluN2B is critical during memory consolidation,” EMBO J., 31, No. 5, 1203–1216 (2012).
Hendricson, A. W., Miao, C. L., Lippmann, M. J., and Morrisett, R. A., “Ifenprodil and ethanol enhance NMDA receptor-dependent longterm depression,” J. Pharmacol. Exp. Ther., 301, 938–944 (2002).
Hestrin, S., “Developmental regulation of NMDA receptor-mediated synaptic currents at a central synapse,” Nature, 357, 686–689 (1992).
Ivanova, O. Ya., Dobryakova, Yu. V., and Markevich, V. A., “Dickkopf-1 and norrin suppress long-term potentiation in vivo in the rat hippocampus,” Zh. Vyssh. Nerv. Deyat., 69, No. 4, 514–521 (2019).
Kudryashova, I. V., “Molecular bases of synapse destabilization as a factor in structural plasticity,” Neirokhimiya, 36, No. 1, 3–13 (2019).
Kudryashova, I. V., Onufriev, M. V., and Gulyaeva, N. V., “Caspase-3 and calpain: differently directed roles in presynaptic long-term plasticity,” Neirokhimiya, 31, No. 3, 200–206 (2014).
Li, R., Huang, F.-S., Abbas, A.-K., and Wigström, H., “Role of NMDA receptor subtypes in different forms of NMDA-dependent synaptic plasticity,” BMC Neurosci., 8, 55 (2007).
Liu, L., Wong, T. P., Pozza, M. F., et al., “Role of NMDA receptor subtypes in governing the direction of hippocampal synaptic plasticity,” Science, 304, 1021–1024 (2004).
Malenka, R. C., “Synaptic plasticity in the hippocampus: LTP and LTD,” Cell, 78, No. 4, 535–538 (1994).
Morishita, W., Lu, W., Smith, G. B., et al., “Activation of NR2B-containing NMDA receptors is not required for NMDA receptor-dependent long-term depression,” Neuropharmacology, 52, No. 1, 71–76 (2007).
Paoletti, P. and Neyton, J., “NMDA receptor subunits: function and pharmacology,” Curr. Opin. Pharmacol., 7, 39–47 (2007).
Sachser, R. M., Santana, F., Crestani, A. P., et al., “Forgetting of long-term memory requires activation of NMDA receptors, L-type voltage-dependent Ca2+ channels, and calcineurin,” Sci. Rep., 6, 22,771 (2016).
Sanhuez, M. and Lisman, J., “The CaMKII/NMDAR complex as a molecular memory,” Mol. Brain, 6, 10 (2013).
Santschi, L. A. and Stanton, P. K., “A paired-pulse facilitation analysis of long-term synaptic depression at excitatory synapses in rat hippocampal CA1 and CA3 regions,” Brain Res., 962, No. 1–2, 78–91 (2003).
Schulz, P. E., “Long-term potentiation involves increases in the probability of neurotransmitter release,” Proc. Natl. Acad. Sci. USA, 94, No. 11, 5888–5893 (1997).
Scullin, C. S., Tafoya, L. C., Wilson, M. C., and Partridge, L. D., “Presynaptic residual calcium and synaptic facilitation at hippocampal synapses of mice with altered expression of SNAP-25,” Brain Res., 1431, 1–12 (2012).
Shipton, O. A. and Paulsen, O., “GluN2A and GluN2B subunit-containing NMDA receptors in hippocampal plasticity,” Philos. Trans. R. Soc. Lond. B Biol. Sci., 369, No. 1633, 20130163 (2014).
Sokolov, M. V., Rossokhin, A. V., Astrelin, A. V., et al., “Quantal analysis suggests strong involvement of presynaptic mechanisms during the initial maintenance of long-term potentiation in rat hippocampal CA1 area in vitro,” Brain Res., 957, 61–75 (2002).
Stanton, P. K., “LTD, LTP, and the sliding threshold for long-term synaptic plasticity,” Hippocampus, 6, No. 1, 35–42 (1996).
Stanton, P. K., Winterer, J., Zhang, X. L., and Muller, W., “Imaging LTP of presynaptic release of FM1-43 from the rapidly recycling vesicle pool of Schaffer collateral-CA1 synapses in rat hippocampal slices,” Eur. J. Neurosci., 22, 2451–2461 (2005).
Stein, I. S., Donaldson, M. S., and Hell, J. W., “CaMKII binding to GluN2B is important for massed spatial learning in the Morris water maze,” F1000Res, 3, 193 (2014).
Valiullina-Rakhmatullina, F. F., Bol’shakov, A. P., and Rozov, A. V., “Three modalities of synaptic neurotransmitter release: rapid synchronous, multivesicular, and asynchronous. Similarities and differences in mechanism,” Zh. Vyssh. Nerv. Deyat., 69, No. 1, 3–13 (2019).
Volgushev, M., Voronin, L. L., Chistiakova, M., and Singer, W., “Relations between long-term synaptic modifi cations and paired-pulse interactions in the rat neocortex,” Eur. J. Neurosci., 9, No. 8, 1656–1665 (1997).
Williams, K., Russell, S. L., Shen, Y. M., and Molinoff, P. B., “Developmental switch in the expression of NMDA receptors occurs in vivo and in vitro,” Neuron, 10, 267–278 (1993).
Wong, J. M. and Gray, J. A., “Long-term depression is independent of GluN2 subunit composition,” J. Neurosci., 38, No. 19, 4462–4470(2018).
Xiao, X., Levy, A. D., Rosenberg, B. J., et al., “Disruption of coordinated presynaptic and postsynaptic maturation underlies the defects in hippocampal synapse stability and plasticity in Abl2/Arg-deficient mice,” J. Neurosci., 36, No. 25, 6778–6791 (2016).
Yasuda, H. and Mukai, H., “Turning off of GluN2B subunits and turning on of CICR in hippocampal LTD induction after developmental GluN2 subunit switch,” Hippocampus, 25, 1274–1284 (2015).
Zucker, R. S. and Regehr, W. G., “Short-term synaptic plasticity,” Annu. Rev. Physiol., 64, 355–405 (2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 70, No. 1, pp. 115–124, January–February, 2020.
Rights and permissions
About this article
Cite this article
Kudryashova, I.V. Presynaptic LTP in Blockade of NMDA2B Receptors. Neurosci Behav Physi 50, 1005–1011 (2020). https://doi.org/10.1007/s11055-020-00999-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-020-00999-8