Abstract
A completely green method is reported here for the preparation of ethyl cellulose submicrospherical capsules wrapped with graphene. Graphene preparation is done by the sonication-assisted graphite exfoliation with the use of ethyl cellulose in an ethanol medium. Edge functionalization of graphene with ethyl cellulose under solvothermal treatment, as suggested from the XRD patterns, can be the driving force for the graphene wrapping of ethyl cellulose capsules that are formed during the addition of excess water to the stable graphene dispersion. Material morphology and structure are analyzed using various characterization techniques, and the novel nanocomposite is effectively used in Cr (VI) removal from aqueous solution, where maximum adsorption is observed in acidic pH indicating the dominance of electrostatic adsorption. The equilibrium data fitted more to Langmuir isotherm than the Freundlich isotherm.
Graphical abstract

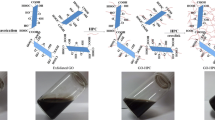
Green one-pot method is reported here for the fabrication of ethyl cellulose submicrospherical capsules wrapped with graphene and is effectively used for Cr (VI) removal.




Similar content being viewed by others
Data Availability
All data associated with the manuscript will be made available to the readers and reviewers upon demand.
Abbreviations
- EC:
-
ethyl cellulose
- GO:
-
graphene oxide
- rGO:
-
reduced graphene oxide
- graphene/EC:
-
ethyl cellulose submicrospherical capsules wrapped with graphene
- XRD:
-
X-ray diffraction
- FTIR:
-
Fourier transform infrared
- SEM:
-
scanning electron microscopy
- TEM:
-
transmission electron spectroscopy
References
Alemu A, Lemma B, Gabbiye N, Alula MT, Desta MT (2018) Removal of chromium (VI) from aqueous solution using vesicular basalt: a potential low cost wastewater treatment system. Heliyon 4:e00682. https://doi.org/10.1016/j.heliyon.2018.e00682
Božič M, Elschner T, Tkaučič D, Bračič M, Hribernik S, Stana Kleinschek K, Kargl R (2018) Effect of different surface active polysaccharide derivatives on the formation of ethyl cellulose particles by the emulsion-solvent evaporation method. Cellulose 25:6901–6922. https://doi.org/10.1007/s10570-018-2062-2
Brodie BC (1859) XIII. On the atomic weight of graphite. Phil Trans R Soc 149:249–259. https://doi.org/10.1098/rstl.1859.0013
Brownson DAC, Kampouris DK, Banks CE (2012) Graphene electrochemistry: fundamental concepts through to prominent applications. Chem Soc Rev 41:6944–6976. https://doi.org/10.1039/c2cs35105f
Ciesielski A, Samorì P (2014) Graphene via sonication assisted liquid-phase exfoliation. Chem Soc Rev 43:381–398. https://doi.org/10.1039/C3CS60217F
Djerafi R, Swanepoel A, Crampon C, Kalombo L, Labuschagne P, Badens E, Masmoudi Y (2017) Supercritical antisolvent co-precipitation of rifampicin and ethyl cellulose. Eur J Pharm Sci 102:161–171. https://doi.org/10.1016/j.ejps.2017.03.016
Gao H, Sun Y, Zhou J, Xu R, Duan H (2013) Mussel-inspired synthesis of polydopamine-functionalized graphene hydrogel as reusable adsorbents for water purification. ACS Appl Mater Interfaces 5:425–432. https://doi.org/10.1021/am302500v
Geim AK, Novoselov KS (2007) The rise of graphene. Nat Mater 6:183–191. https://doi.org/10.1038/nmat1849
Geleta WS, Alemayehu E, Lennartz B (2021) Volcanic rock materials for defluoridation of water in fixed-bed column systems. Molecules 26:977. https://doi.org/10.3390/molecules26040977
Goswami S, Ghosh UC (2006) Studies on adsorption behaviour of Cr(VI) onto synthetic hydrous stannic oxide. WSA 31:597–602. https://doi.org/10.4314/wsa.v31i4.5150
Guardia L, Fernández-Merino MJ, Paredes JI, Solís-Fernández P, Villar-Rodil S, Martínez-Alonso A, Tascón JMD (2011) High-throughput production of pristine graphene in an aqueous dispersion assisted by non-ionic surfactants. Carbon 49:1653–1662. https://doi.org/10.1016/j.carbon.2010.12.049
Haridas V, Yaakob Z, Renuka NK et al (2021) Selective electrochemical determination of paracetamol using hematite/graphene nanocomposite modified electrode prepared in a green chemical route. Mater Chem Phys 263:124379. https://doi.org/10.1016/j.matchemphys.2021.124379
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339–1339. https://doi.org/10.1021/ja01539a017
Khan SA, Riaz-ur-Rehman, Khan MA (1995) Adsorption of chromium (III), chromium (VI) and silver (I) on bentonite. Waste Manag 15:271–282. https://doi.org/10.1016/0956-053X(95)00025-U
Kilic M, Apaydin-Varol E, Pütün AE (2011) Adsorptive removal of phenol from aqueous solutions on activated carbon prepared from tobacco residues: equilibrium, kinetics and thermodynamics. J Hazard Mater 189:397–403. https://doi.org/10.1016/j.jhazmat.2011.02.051
Kim T-H, Jeon EK, Ko Y, Jang BY, Kim BS, Song HK (2014) Enlarging the d-spacing of graphite and polarizing its surface charge for driving lithium ions fast. J Mater Chem A 2:7600–7605. https://doi.org/10.1039/C3TA15360F
Li D, Müller MB, Gilje S, Kaner RB, Wallace GG (2008) Processable aqueous dispersions of graphene nanosheets. Nat Nanotech 3:101–105. https://doi.org/10.1038/nnano.2007.451
Li J, Liu J, Tan G, Jiang J, Peng S, Deng M, Qian D, Feng Y, Liu Y (2014) High-sensitivity paracetamol sensor based on Pd/graphene oxide nanocomposite as an enhanced electrochemical sensing platform. Biosens Bioelectron 54:468–475. https://doi.org/10.1016/j.bios.2013.11.001
Liang YT, Hersam MC (2010) Highly concentrated graphene solutions via polymer enhanced solvent exfoliation and iterative solvent exchange. J Am Chem Soc 132:17661–17663. https://doi.org/10.1021/ja107661g
Malipeddi VR, Awasthi R, Dua K (2016) Formulation and evaluation of controlled release ethylcellulose and polyethylene glycol microspheres containing metoprolol tartrate. Interv Med Appl Sci 8:60–67. https://doi.org/10.1556/1646.8.2016.2.6
Mardi S, Risi Ambrogioni M, Reale A (2020) Developing printable thermoelectric materials based on graphene nanoplatelet/ethyl cellulose nanocomposites. Mater Res Express 7:085101. https://doi.org/10.1088/2053-1591/ababc0
McAllister MJ, Li J-L, Adamson DH et al (2007) Single sheet functionalized graphene by oxidation and thermal expansion of graphite. Chem Mater 19:4396–4404. https://doi.org/10.1021/cm0630800
Melitas N, Chuffe-Moscoso O, Farrell J (2001) Kinetics of soluble chromium removal from contaminated water by zerovalent iron media: corrosion inhibition and passive oxide effects. Environ Sci Technol 35:3948–3953. https://doi.org/10.1021/es001923x
Mondal NK, Chakraborty S (2020) Adsorption of Cr(VI) from aqueous solution on graphene oxide (GO) prepared from graphite: equilibrium, kinetic and thermodynamic studies. Appl Water Sci 10:61. https://doi.org/10.1007/s13201-020-1142-2
Olad A, Nabavi R (2007) Application of polyaniline for the reduction of toxic Cr(VI) in water. J Hazard Mater 147:845–851. https://doi.org/10.1016/j.jhazmat.2007.01.083
Parida P, Mishra SC, Sahoo S, Behera A, Nayak BP (2016) Development and characterization of ethylcellulose based microsphere for sustained release of nifedipine. J Pharm Anal 6:341–344. https://doi.org/10.1016/j.jpha.2014.02.001
Pérez-Martínez JI, Morillo E, Maqueda C, Ginés JM (2001) Ethyl cellulose polymer microspheres for controlled release of norfluazon: microencapsulation and controlled release of norfluazon. Pest Manag Sci 57:688–694. https://doi.org/10.1002/ps.339
Ranjan P, Agrawal S, Sinha A, Rao TR, Balakrishnan J, Thakur AD (2018) A low-cost non-explosive synthesis of graphene oxide for scalable applications. Sci Rep 8:12007. https://doi.org/10.1038/s41598-018-30613-4
Selvi K (2001) Removal of Cr(VI) from aqueous solution by adsorption onto activated carbon. Bioresour Technol 80:87–89. https://doi.org/10.1016/S0960-8524(01)00068-2
Sharma YC (2001) Effect of temperature on interfacial adsorption of Cr(VI) on wollastonite. J Colloid Interface Sci 233:265–270. https://doi.org/10.1006/jcis.2000.7232
Sharma YC, Weng CH (2007) Removal of chromium(VI) from water and wastewater by using riverbed sand: kinetic and equilibrium studies. J Hazard Mater 142:449–454. https://doi.org/10.1016/j.jhazmat.2006.08.078
Stankovich S, Piner RD, Chen X, Wu N, Nguyen SBT, Ruoff RS (2006) Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly(sodium 4-styrenesulfonate). J Mater Chem 16:155–158. https://doi.org/10.1039/B512799H
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen SBT, Ruoff RS (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45:1558–1565. https://doi.org/10.1016/j.carbon.2007.02.034
Staudenmaier L (1898) Verfahren zur Darstellung der Graphitsäure. Ber Dtsch Chem Ges 31:1481–1487. https://doi.org/10.1002/cber.18980310237
Sun X-F, Ma Y, Liu X-W, Wang SG, Gao BY, Li XM (2010) Sorption and detoxification of chromium(VI) by aerobic granules functionalized with polyethylenimine. Water Res 44:2517–2524. https://doi.org/10.1016/j.watres.2010.01.027
Sun P, Kuga S, Wu M, Huang Y (2014) Exfoliation of graphite by dry ball milling with cellulose. Cellulose 21:2469–2478. https://doi.org/10.1007/s10570-014-0264-9
Wang S, Yi M, Shen Z, Zhang X, Ma S (2014) Adding ethanol can effectively enhance the graphene concentration in water–surfactant solutions. RSC Adv 4:25374–25378. https://doi.org/10.1039/C4RA03345K
Weng C-H, Sharma YC, Chu S-H (2008) Adsorption of Cr(VI) from aqueous solutions by spent activated clay. J Hazard Mater 155:65–75. https://doi.org/10.1016/j.jhazmat.2007.11.029
Yamuna A, Sundaresan P, Chen S-M (2019) Ethylcellulose assisted exfoliation of graphite by the ultrasound emulsification: an application in electrochemical acebutolol sensor. Ultrason Sonochem 59:104720. https://doi.org/10.1016/j.ultsonch.2019.104720
Yi M, Shen Z (2015) A review on mechanical exfoliation for the scalable production of graphene. J Mater Chem A 3:11700–11715. https://doi.org/10.1039/C5TA00252D
Zhang LL, Zhou R, Zhao XS (2010) Graphene-based materials as supercapacitor electrodes. J Mater Chem 20:5983. https://doi.org/10.1039/c000417k
Zhang Q, An C, Fan S, Shi S, Zhang R, Zhang J, Li Q, Zhang D, Hu X, Liu J (2018) Flexible gas sensor based on graphene/ethyl cellulose nanocomposite with ultra-low strain response for volatile organic compounds rapid detection. Nanotechnology 29:285501. https://doi.org/10.1088/1361-6528/aabf2f
Zheng Q, Kim J-K (2015) Synthesis, structure, and properties of graphene and graphene oxide. In: Graphene for transparent conductors. Springer New York, New York, pp 29–94
Acknowledgements
The authors thank Sree Neelakanta Govt. Sanskrit College Pattambi and the University of Calicut for providing the facilities for carrying out the research work. SAIF Kochi, India is acknowledged for TEM, XRD, and FTIR spectral analyses. CSIF, University of Calicut is acknowledged for FESEM–EDAX analysis. Mar Athanasius College (Autonomous), Kothamangalam is acknowledged for surface area analysis.
Funding
UGC, New Delhi, India had provided a research fellowship in the form of UGC-MANF to Shemeena Mullakkattuthodi.
Author information
Authors and Affiliations
Contributions
Sumayya P K and Shemeena Mullakkattuthodi had done the experimental part; Suvarna K. Subrahmanian had done the experimental part and manuscript editing; Sankaran Sugunan contributed in manuscript writing and discussions of the work; and Binitha N. Narayanan made the scientific contribution, experimental part, interpretation of data, and preparation of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sumayya, P.K., Subrahmanian, S.K., Mullakkattuthodi, S. et al. Green in situ preparation of novel graphene-wrapped ethyl cellulose submicrospherical capsules and its effective use in Cr (VI) removal. J Nanopart Res 23, 104 (2021). https://doi.org/10.1007/s11051-021-05212-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-021-05212-6