Abstract
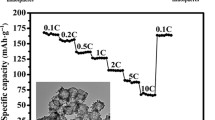
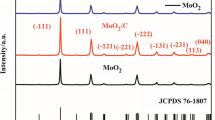
In this work, hollow FeS2 nanospheres were successfully synthesized via a simple hydrolysis method followed by annealing and a sulfidation process. These nanospheres have diameters of approximately range from 300 to 400 nm and shells about 50 nm thick. Due to the unique hollow structures, these FeS2 reveal excellent long-cycling performance through improving the ratio of the pseudocapacitance effects in total charge storage. At a current density of 1 A g−1, the FeS2 anode delivered a high capacity of 541.5 mAh g−1 after 100 cycles. More importantly, these FeS2 could retain a capacity of 313.5 mAh g−1 after 500 cycles at a high current density of 5 A g−1, and still achieve a capacity of 147.2 mAh g−1 after 400 cycles at a higher current density of 10 A g−1.






Similar content being viewed by others
References
Armand M, Tarascon JM (2008) Building better batteries. Nature 451:652–657. https://doi.org/10.1038/451652a
Augustyn V, Come J, Lowe MA, Kim JW, Taberna PL, Tolbert S, Abruña HD, Simon P, Dunn B (2013) High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance. Nat Mater 12:518–522. https://doi.org/10.1038/nmat3601
Bai ZC, Zhang YW, Zhang YH, Guo CL, Tang B (2015) A large-scale, green route to synthesize of leaf-like mesoporous CuO as high-performance anode materials for lithium ion batteries. Electrochim Acta 159:29–34. https://doi.org/10.1016/j.electacta.2015.01.188
Chen X, Zhang N, Sun K (2012) Facile fabrication of CuO mesoporous nanosheet cluster array electrodes with super lithium-storage properties. J Mater Chem 22:13637–13642. https://doi.org/10.1039/C2JM32014B
Chen CJ, Wen YW, Hu XL, Ji XL, Yan MY, Mai LQ, Hu P, Shan B, Huang YH (2015) Na+ intercalation pseudocapacitance in graphene-coupled titanium oxide enabling ultra-fast sodium storage and long-term cycling. Nat Commun 6:6929. https://doi.org/10.1038/ncomms7929
Chen YY, Hu XD, Evanko B, Sun XH, Li X, Hou TY, Cai S, Zheng CM, Hu WB, Stucky G (2018) High-rate FeS2/CNT neural network nanostructure composite anodes for stable, high-capacity sodium-ion batteries. Nano Energy 46:117–127. https://doi.org/10.1016/j.nanoen.2018.01.039
Choi JW, Cheruvally G, Ahn HJ, Kim KW, Ahn JH (2006) Electrochemical characteristics of room temperature Li/FeS2 batteries with natural pyrite cathode. J Power Sources 163:158–165. https://doi.org/10.1016/j.jpowsour.2006.04.075
Cui J, Yao SS, Kim JK (2017) Recent progress in rational design of anode materials for high-performance Na-ion batteries. Energy Storage Mater 7:64–114. https://doi.org/10.1016/j.ensm.2016.12.005
Douglas A, Carter R, Oakes L, Share K, Cohn AP, Pint CL (2015) Ultrafine iron pyrite (FeS2) nanocrystals improve sodium–sulfur and lithium–sulfur conversion reactions for efficient batteries. ACS Nano 9:11156–11165. https://doi.org/10.1021/acsnano.5b04700
Evans T, Piper D, Kim S, Han S, Bhat V, Oh K, Lee S (2014) Ionic liquid enabled FeS2 for high-energy-density lithium-ion batteries. Adv Mater 26:7386–7392. https://doi.org/10.1002/adma.201402103
Goodenough JB, Kim Y (2010) Challenges for rechargeable Li batteries. Chem Mater 22:587–603. https://doi.org/10.1039/10.1021/cm901452z
Hu Z, Zhu ZQ, Cheng FY, Zhang K, Wang JB, Chen CC, Chen J (2015) Pyrite FeS2 for high-rate and long-life rechargeable sodium batteries. Energy Environ Sci 8:1309–1316. https://doi.org/10.1039/C4EE03759F
Huang SY, Liu XY, Li QY, Chen J (2009) Pyrite film synthesized for lithium-ion batteries. J Alloys Compd 472:L9–L12. https://doi.org/10.1016/j.jallcom.2008.04.058
Kim TB, Jung WH, Ryu HS, Kim KW, Ahn JH, Cho KK, Cho GB, Nam TH, Ahn IS, Ahn HJ (2008) Electrochemical characteristics of Na/FeS2 battery by mechanical alloying. J Alloys Compd 449:304–307. https://doi.org/10.1016/j.jallcom.2006.02.113
Kitajou A, Yamaguchi J, Hara S, Okada S (2014) Discharge/charge reaction mechanism of a pyrite-type FeS2 cathode for sodium secondary batteries. J Power Sources 247:391–395. https://doi.org/10.1016/j.jpowsour.2013.08.123
Li TT, Guo ZX, Li XY, Wu ZN, Zhang K, Liu HW, Sun HZ, Liu Y, Zhang H (2015) Colloidal systhesis of marcasite of FeS2 ananoparticles with improved electrochemical performance. RSC Adv 5:98967–98970. https://doi.org/10.1039/C5RA22610D
Liu C, Li F, Ma L, Cheng H (2010) Advanced materials for energy storage. Adv Mater 22:E28–E62. https://doi.org/10.1002/adma.200903328
Liu J, Wen Y, Wang Y, Aken P, Maier J, Yu Y (2014) Carbon-encapsulated pyrite as stable and earth-abundant high energy cathode material for rechargeable lithium batteries. Adv Mater 26:6025–6030. https://doi.org/10.1002/adma.201401496
Liu ZM, Lu TC, Song T, Yu X, Lou XW, Paik U (2017) Structure-designed synthesis of FeS2@C yolk–shell nanoboxes as a high-performance anode for sodium-ion batteries. Energy Environ Sci 10:1576–1580. https://doi.org/10.1039/C7EE01100H
Lu ZX, Wang NN, Zhang YH, Xue P, Guo MQ, Tang B, Bai ZC, Dou SX (2017) Pyrite FeS2@C nanorods as smart cathode for sodium ion battery with ultra-long lifespan and notable rate performance from tunable pseudocapacitance. Electrochim Acta 260:755–761. https://doi.org/10.1016/j.electacta.2017.12.031
Shao-Horn Y, Osmialowski S, Horn QC (2002) Nano-FeS2 for commercial Li/FeS2 primary batteries. J Electrochem Soc 149:1499–1502. https://doi.org/10.1149/1.1513558
Wang J, Polleux J, Lim J, Dunn B (2007) Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles. J Phys Chem C 111:14925–14931. https://doi.org/10.1021/jp074464w
Wang L, Zhao JS, He XM, Wan CR (2011) Kinetic investigation of sulfurized polyacrylonitrile cathode material by electrochemical impedance spectroscopy. Electrochim Acta 56:5252–5256. https://doi.org/10.1016/j.electacta.2011.03.009
Wang ZY, Zhou L, Lou XW (2012) Metal oxide hollow nanostructures for lithium-ion batteries. Adv Mater 24:1903–1911. https://doi.org/10.1002/adma.201200469
Wang YX, Yang JP, Chou SL, Liu HK, Zhang WX, Zhao DY, Dou SX (2015) Uniform yolk-shell iron sulfide–carbon nanospheres for superior sodium–iron sulfide batteries. Nat Commun 6:8689. https://doi.org/10.1038/ncomms9689
Wang FB, Li GD, Meng XG, Li YX, Gao QF, Xu YQ, Cui WF (2018a) FeS2 nanosheets encapsulated in 3D porous carbon spheres for excellent Na storage in sodium-ion batteries. Inorg Chem Front 5:2462–2471. https://doi.org/10.1039/C8QI00679B
Wang NN, Xu X, Liao T, Du Y, Bai ZC, Dou SX (2018b) Boosting sodium storage of double-Shell sodium titanate microspheres constructed from 2D ultrathin nanosheets via sulfur doping. Adv Mater 30:1804157. https://doi.org/10.1002/adma.201804157
Whittingham MS (2004) Lithium batteries and cathode materials. Chem Rev 104:4271–4302. https://doi.org/10.1002/chin.200450266
Zhamu A, Chen GR, Liu CG, Neff D, Fang Q, Yu ZN, Xiong W, Wang YB, Wang XB, Jang BZ (2012) Reviving rechargeable lithium metal batteries: enabling next-generation high-energy and high-power cells. Energy Environ Sci 5:5701–5707. https://doi.org/10.1039/C2EE02911A
Zhang D, Tu JP, Xiang JY, Qiao YQ, Xia XH, Wang XL, Gu CD (2011) Influence of particle size on electrochemical performances of pyrite FeS2 for Li-ion batteries. Electrochim Acta 56:9980–9985. https://doi.org/10.1016/j.electacta.2011.08.119
Zhang K, Hu Z, Liu X, Tao ZL, Chen J (2015) FeSe2 microspheres as a high-performance anode material for Na-ion batteries. Adv Mater 27:3305–3309. https://doi.org/10.1002/adma.201500196
Zhao WX, Guo CX, Li CM (2017) Lychee-like FeS2@FeSe2 core–shell microspheres anode in sodium ion batteries for large capacity and ultralong cycle life. J Mater Chem A 5:19195–19202. https://doi.org/10.1039/C7TA05931K
Zhou MJ, Liu YC, Chen J, Yang XL (2015) Double shelled hollow SnO2/polymer microsphere as a high-capacity anode material for superior reversible lithium ion storage. J Mater Chem A 3:1068–1076. https://doi.org/10.1039/C4TA05235H
Zhu YJ, Suo LM, Gao T, Fan XL, Han FD, Wang CS (2015) Ether-based electrolyte enabled Na/FeS2 rechargeable batteries. Electrochem Commun 54:18–22. https://doi.org/10.1016/j.elecom.2015.02.006
Funding
This work was supported by the National Natural Science Foundation of China (No. 11774188), the Incubation Program of University’ Preponderant Discipline of Shandong Province (No. 03010304), Mountain Tai Young Scholarship (No. 23170504), and the Natural Science Foundation of Shandong Province (No. ZR2018QB003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, F., Li, G. & Cui, W. FeS2 hollow nanospheres as high-performance anode for sodium ion battery and their surface pseudocapacitive properties. J Nanopart Res 21, 121 (2019). https://doi.org/10.1007/s11051-019-4565-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-019-4565-7