Abstract
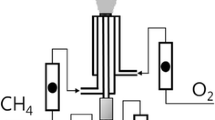
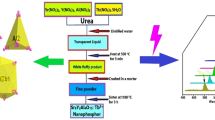
In flame spray pyrolysis (FSP), the generation of uniform nanoparticles can be quite challenging due to difficulties in controlling droplet sizes during liquid spraying and uneven flame temperature. Here, we report a method to produce relatively uniform nanocrystals of a Tb3+ doped Y2O3 phosphor. In ethanol, metal nitrate precursors were simply mixed with organic surfactants to form a homogeneous solution which was then subjected to FSP. Depending on relative concentrations of the surfactant (oleic acid) to the metal precursors (yttrium and terbium nitrates), different sizes and morphologies of Y2O3:Tb3+ particles were obtained. By adjusting the surfactant concentration, Y2O3:Tb3+ crystals as small as 20~25 nm were acquired. X-ray diffraction and transmittance electron microscopy were used to prove that as-synthesized nanoparticles were highly crystalline due to the high temperature of FSP. X-ray photoelectron spectroscopy revealed that terbium dopants were well distributed throughout Y2O3 particles and a small portion of carbonate impurities were remained on the surface of particles, presumably originated from incomplete combustion of the organic surfactants. Photoluminescence (PL) spectra of Y2O3:Tb3+ nanocrystals exhibited a green light emission ensuring that the terbium doping was successfully occurred. However, when post-annealing was performed on the nanocrystals, their PL was dramatically enhanced indicating that quenching centers such as carbonate impurities and surface defects may have been removed by the annealing process. Owing to the continuous processability of FSP, this current method can be a practical way to produce nanoparticles in a large quantity. The obtained Y2O3:Tb3+ nanocrystals were used to fabricate a transparent film with poly-ethylene-co-vinyl acetate (poly-EVA) polymer, which was suitable for a spectral converting layer for a solar cell.

ᅟ










Similar content being viewed by others
References
Barr TL (1977) Esca studies of naturally passivated metal foils. J Vac Sci Technol 14:660–665. https://doi.org/10.1116/1.569174
Basavegowda N, Mishra K, Thombal RS, Kaliraj K, Lee YR (2017) Sonogreen synthesis of yttium oxide (Y2O3) nanoparticles as a novel heterogenous catalyst for the construction of biologically interesting 1,3-thioazolidin-4-ones. Catal Lett 147:2630–2639. https://doi.org/10.1007/s10562-017-2168-4
Boukerika A, Guerbous L (2014) Annealing effects on structural and luminescence properties of red Eu3+-doped Y2O3 nanophosphors prepared by sol-gel method. J Lumin 145:148–153. https://doi.org/10.1016/j.jlumin.2013.07.037
Camenzind A, Strobel R, Krumeich F, Pratsinis SE (2007) Luminescence and crystallinity of flame-made Y2O3:Eu3+ nanoparticles. Adv Powder Technol 18:5–22. https://doi.org/10.1163/156855207779768142
Camenzind A, Strobel R, Pratsinis SE (2005) Cubic or monoclinic Y2O3:Eu3+ nanoparticles by one step flame spray pyrolysis. Chem Phys Lett 415:193–197. https://doi.org/10.1016/j.cplett.2005.09.002
Chen N, Ji L, Du GP (2014) Luminescent properties of hybrid nanostructures of ion-exchanged Y2O3:Eu3+ nanoparticles with 2-thenoyltrifluoroacetone ligands. J Lumin 153:259–263. https://doi.org/10.1016/j.jlumin.2014.03.042
Chui WS, Khiew PS, Isa D, Cloke M, Radiman S, Abd-Shukor R, Abdullah MH, Huang NM (2008) Synthesis of two-dimensional ZnO nanopellets by pyrolysis of zinc oleate. Chem Eng J 142:337–343. https://doi.org/10.1016/j.cej.2008.04.034
Cullity BD, Stock SR (2001) Elements of x-ray diffraction, 3rd edn. Prentice Hall, Upper Saddle River
Dickenko YT, Suslick KS (2005) Chemical aerosol flow synthesis of semiconductor nanoparticles. J Am Chem Soc 127:12196–125197
Dosev D, Guo B, Kennedy IM (2006) Photoluminescence of as an indication of crystal structure and particle size in nanoparticles synthesized by flame spray pyrolysis. J Aerosol Sci 37:402–412. https://doi.org/10.1016/j.jaerosci.2005.08.009
Ghazaghi M, Mousavi HZ, Rashidi AM, Shirkhanloo H, Ghaffarian HR (2017) Preparation of graphene-nickel nanoparticles hybrid by spray pyrolysis using nickel oleate precursor and its application as a ferrofluid Inorg Nano-Met Chem 47:558–564. https://doi.org/10.1080/15533174.2016.1186084
Glassman I, Yetter RA (2008) Combustion, fourth edition. Elsevier Inc, USA, pp 186–189
Grohn AJ, Pratsinis SE, Sanchez-Ferrer A, Mezzenga R, Wegner K (2014) Scale-up of nanoparticle synthesis by flame spray pyrolysis: the high-temperature particle residence time. Ind Eng Chem Res 53:10734–10742. https://doi.org/10.1021/Ie501709s
Guo B, Luo ZP (2008) Particle size effect on the crystal structure of Y2O3 particles formed in a flame aerosol process. J Am Ceram Soc 91:1653–1658. https://doi.org/10.1111/j.1551-2916.2008.02341.x
Guo B, Yim H, Hwang W, Nowell M, Luo Z (2011) Crystalline phase of Y2O3:Eu particles generated in a substrate-free flame process. Particuology 9:24–31. https://doi.org/10.1016/j.partic.2010.07.020
Huang XY, Han SY, Huang W, Liu XG (2013) Enhancing solar cell efficiency: the search for luminescent materials as spectral converters. Chem Soc Rev 42:173–201. https://doi.org/10.1039/C2cs35288e
Jadhav AP, Khan S, Kim SJ, Cho S-H (2014) Near-IR quantum cutting phosphors: a step towards enhancing solar cell efficiency. Appl Sci Converg Technol 23:221–239. https://doi.org/10.5757/ASCT.2014.23.5.289
Kang YC, Seo DJ, Park SB, Park HD (2001) Morphological and optical characteristics of Y2O3:Eu phosphor particles prepared by flame spray pyrolysis. Jpn J Appl Phys 1(40):4083–4086. https://doi.org/10.1143/Jjap.40.4083
Khan S, Park B-I, Han JS, Lee SY, Cho SH (2018) Flame synthesized Y2O3:Tb3+–Yb3+ phosphors as spectral convertors for solar cells. Res Chem Intermed 44:619–4632. https://doi.org/10.1007/s11164-018-3270-y
Mahajan SV, Dickerson JH (2007) Synthesis of monodisperse sub-3 nm RE2O3 and Gd2O3: RE3+ nanocrystals. Nanotechnology 18:325605. https://doi.org/10.1088/0957-4484/18/32/325605
Majumdar D, Chatterjee D (1991) X-ray photoelectron spectroscopic studies on yttria, zirconia, and yttria-stabilized zirconia. J Appl Phys 70:988–992. https://doi.org/10.1063/1.349611
Milosevic OB, Mancic L, Rabanal ME, Gomez LS, Marinkovic K (2009) Aerosol route in processing of nanostructured functional materials. KONA Powder Part J 27:84–106. https://doi.org/10.14356/kona.2009010
Packiyaraj P, Thangadurai P (2014) Structural and photoluminescence studies of Eu3+ doped cubic Y2O3 nanophosphors. J Lumin 145:997–1003. https://doi.org/10.1016/j.jlumin.2013.07.074
Pandey AR, Rai VK, Kumar KH (2014) Influence of Li+ codoping on visible emission of Y2O3:Tb3+, Yb3+ phosphor. Spectrochim Acta A 118:619–623. https://doi.org/10.1016/j.saa.2013.08.109
Qin X, Ju YG, Bernhard S, Yao N (2005) Flame synthesis of Y2O3:Eu nanophosphors using ethanol as precursor solvents. J Mater Res 20:2960–2968. https://doi.org/10.1557/Jmr.2005.0364
Reichl R, Gaukler KH (1986) An investigation of air-grown yttrium oxide and experimental determination of the sputtering yield and the inelastic mean free path. Appl Surf Sci 26:196–210. https://doi.org/10.1016/0169-4332(86)90005-X
Rowan BC, Wilson LR, Richards BS (2008) Advanced material concepts for luminescent solar concentrators. IEEE J Sel Top Quantum Electron 14:1312–1322. https://doi.org/10.1109/Jstqe.2008.920282
Si R, Zhang YW, Zhou HP, Sun LD, Yan CH (2007) Controlled-synthesis, self-assembly behavior, and surface-dependent optical properties of high-quality rare-earth oxide nanocrystals. Chem Mater 19:18–27. https://doi.org/10.1021/Cm0618392
Singh AK, Singh SK, Rai SB (2014) Role of Li+ ion in the luminescence enhancement of lanthanide ions: favorable modifications in host matrices. RSC Adv 4:27039–27061. https://doi.org/10.1039/C4ra01055h
Sotiriou GA, Schneider M, Pratsinis SE (2011) Color-tunable nanophosphors by codoping flame-made Y2O3 with Tb and Eu. J Phys Chem C 115:1084–1089. https://doi.org/10.1021/Jp106137u
Sotiriou GA, Schneider M, Pratsinis SE (2012) Green, silica-coated monoclinic Y2O3:Tb3+ nanophosphors: flame synthesis and characterization. J Phys Chem C 116:4493–4499. https://doi.org/10.1021/jp211722z
Strobel R, Pratsinis SE (2007) Flame aerosol synthesis of smart nanostructured materials. J Mater Chem 17:4743–4756. https://doi.org/10.1039/B711652G
Teoh WY, Amal R, Madler L (2010) Flame spray pyrolysis: an enabling technology for nanoparticles design and fabrication. Nanoscale 2:1324–1347. https://doi.org/10.1039/C0NR00017E
Yuan JL, Zeng XY, Zhao JT, Zhang ZJ, Chen HH, Yang XX (2008) Energy transfer mechanisms in Tb3+, Yb3+ codoped Y2O3 downconversion phosphor. J Phys D Appl Phys 41:105406. https://doi.org/10.1088/0022-3727/41/10/105406
Zhang QY, Huang XY (2010) Recent progress in quantum cutting phosphors. Prog Mater Sci 55:353–427. https://doi.org/10.1016/j.pmatsci.2009.10.001
Zhao Y, Li J-G, Guo M, Yang X (2013) Structural and photoluminescent investigation of LTbH/LEuH nanosheets and their color-tunable colloidal hybrids. J Mater Chem C 1:3584–3592. https://doi.org/10.1039/C3TC30205A
Funding
We would like to acknowledge the financial support from the R&D Convergence Program of NST (National Research Council of Science & Technology) of Republic of Korea (project No. CAP-16-10-KIMS) and KIST institutional funding (project No. 2E28020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest in this work.
Electronic supplementary material
ESM 1
The following are available online at www.mdpi.com/link, Fig. S1: Representative SEM images for measuring particle size distributions of Y2O3:Tb3+ nanocrystals obtained with 0.1-M (left) and 0.5-M oleic acid (right), Fig. S2: XRD patterns of Y2O3:Tb3+ nanocrystals obtained with 0.1-M oleic acid: (a) as-synthesized product; heat-treated (b) 500 °C; (c) 700 °C; (d) 900 °C, and Fig. S3: TEM images of Y2O3:Tb3+ nanocrystals obtained with 0.1-M oleic acid: (a) as-synthesized product; heat-treated at (b) 500 °C; (c) 700 °C; (d) 800 °C; and (e) 900 °C. (DOCX 2828 kb)
Rights and permissions
About this article
Cite this article
Khan, S., Han, J.S., Lee, S.Y. et al. Flame-synthesized Y2O3:Tb3+ nanocrystals as spectral converting materials. J Nanopart Res 20, 241 (2018). https://doi.org/10.1007/s11051-018-4347-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-018-4347-7