Abstract
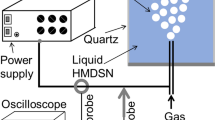
This study demonstrates a novel way of hydrogen extraction from toluene and direct storage in plasma generated palladium (Pd) nanoparticles in one-step. Monodispersed Pd nanoparticles with a narrow particle-size distribution have been successfully synthesized, for the first time, by a plasma discharge between Pd electrodes in the cavitation field of toluene. The resulting well-dispersed Pd nanoparticles embedded in carbon are stabilized against agglomeration. The effect of experiment time on the particle size and the expansion of lattice spacing due to hydrogen are investigated by experimental and computational studies using density functional theory (DFT). Using X-ray diffraction and high-resolution transmission electron microscopy measurements, it was found that particle size and lattice expansion increases with experiment time. Pd nanoparticle synthesis for in situ hydrogen storage in one step using plasma discharge in an appropriate solvent emphasizes the importance of adopting this methodology which offers several advantages. These include rapid reaction rate, ability to form very small nanoparticles with narrow size distribution and hydrogen extraction from the solvent for direct storage in the Pd nanoparticle lattice.











Similar content being viewed by others
References
Adams BD, Wu G, Nigro S, Chen A (2009) Facile synthesis of Pd−Cd nanostructures with high capacity for hydrogen storage. J Am Chem Soc 131:6930–6931. https://doi.org/10.1021/ja901798u
Amin-Ahmadi B et al (2013) Effect of deposition rate on the microstructure of electron beam evaporated nanocrystalline palladium thin films. Thin Solid Films 539:145–150. https://doi.org/10.1016/j.tsf.2013.05.083
Blöchl PE (1994) Projector augmented-wave method. Phys Rev B 50:17953–17979
Blöchl PE, Jepsen O, Andersen OK (1994) Improved tetrahedron method for Brillouin-zone integrations. Phys Rev B 49:16223–16233
Campesi R, Cuevas F, Gadiou R, Leroy E, Hirscher M, Vix-Guterl C, Latroche M (2008) Hydrogen storage properties of Pd nanoparticle/carbon template composites. Carbon 46:206–214. https://doi.org/10.1016/j.carbon.2007.11.006
Caputo R, Alavi ALI (2003) Where do the H atoms reside in PdH x systems? Mol Phys 101:1781–1787. https://doi.org/10.1080/0026897031000094489
Chan CT, Louie SG (1983) Self-consistent pseudopotential calculation of the electronic structure of PdH and Pd 4 H. Phys Rev B 27:3325
Chaudhary RP, Koymen AR (2015) ynthesis of magnetic GdC2 nanoparticles using cavitation plasma. Mater Lett 158:194–197. https://doi.org/10.1016/j.matlet.2015.05.156
Chaudhary RP, Mohanty SK, Koymen AR (2011) New method for synthesis of Pt nanoparticles embedded in a carbon matrix. J Nanosci Nanotechnol 11:10396–10400. https://doi.org/10.1166/jnn.2011.5138
Chaudhary RP, Mohanty SK, Koymen AR (2014) Novel method for synthesis of Fe core and C shell magnetic nanoparticles. Carbon 79:67–73. https://doi.org/10.1016/j.carbon.2014.07.043
Chen A, Ostrom C (2015) Palladium-based nanomaterials: synthesis and electrochemical applications. Chem Rev 115:11999–12044. https://doi.org/10.1021/acs.chemrev.5b00324
Eichenauer W (1967) F. A. Lewis: the palladium hydrogen system. Academic Press, London and New York 1967. 178 Seiten, 87 Abbildungen. Preis: 45 s.. Berichte der Bunsengesellschaft für physikalische Chemie 71:1160–1161. https://doi.org/10.1002/bbpc.19670710933
Ellner M (1981) Zusammenhang zwischen strukturellen und thermo-dynamischen eigenschaften bei phasen der kupfer-familie in T10-B4-systemen. J Less Common Met 78:21–32. https://doi.org/10.1016/0022-5088(81)90140-5
Elsässer C, Fähnle M, Ho K, Chan CT (1991) Ab initio pseudopotential calculations of total energies and forces for hydrogen in palladium. Phys B Condens Matter 172:217–224
Ferguson G Jr, Schindler A, Tanaka T, Morita T (1965) Neutron diffraction study of temperature-dependent properties of palladium containing absorbed hydrogen. Phys Rev 137:A483
Flanagan TB, Oates W (1991) The palladium-hydrogen system. Annu Rev Mater Sci 21:269–304
Fukai Y (2005) The metal-hydrogen system basic bulk properties, 2nd ed, vol 21. Springer-Verlag Berlin Heidelberg. https://doi.org/10.1007/3-540-28883-X
Gajdoš M, Hummer K, Kresse G, Furthmüller J, Bechstedt F (2006) Linear optical properties in the projector-augmented wave methodology. Phys Rev B 73:045112
Gelatt C Jr, Ehrenreich H, Weiss JA (1978) Transition-metal hydrides: electronic structure and the heats of formation. Phys Rev B 17:1940
Gupta M, Freeman A (1978) Electronic structure and proton spin-lattice relaxation in PdH. Phys Rev B 17:3029
Hohenberg P, Kohn W (1964) Inhomogeneous electron gas. Phys Rev 136:B864–B871
Houari A, Matar SF, Eyert V (2014) Electronic structure and crystal phase stability of palladium hydrides. J Appl Phys 116:173706. https://doi.org/10.1063/1.4901004
Huang H et al (2015) Novel hydrogen storage properties of palladium nanocrystals activated by a pentagonal cyclic twinned structure. Nano Res 8:2698–2705. https://doi.org/10.1007/s12274-015-0776-0
Isaeva L, Bazhanov D, Isaev E, Eremeev S, Kulkova S, Abrikosov I (2011) Dynamic stability of palladium hydride: an ab initio study. Int J Hydrog Energy 36:1254–1258
Khanuja M, Mehta BR, Agar P, Kulriya PK, Avasthi DK (2009) Hydrogen induced lattice expansion and crystallinity degradation in palladium nanoparticles: effect of hydrogen concentration, pressure, and temperature. J Appl Phys 106:093515. https://doi.org/10.1063/1.3253733
Kobayashi H, Yamauchi M, Kitagawa H, Kubota Y, Kato K, Takata M (2008) On the nature of strong hydrogen atom trapping inside Pd nanoparticles. J Am Chem Soc 130:1828–1829. https://doi.org/10.1021/ja7102372
Kohn W, Sham LJ (1965) Self-consistent equations including exchange and correlation effects. Phys Rev 140:A1133–A1138
Kresse G, Furthmüller J (1996a) Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput Mater Sci 6:15–50. https://doi.org/10.1016/0927-0256(96)00008-0
Kresse G, Furthmüller J (1996b) Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B 54:11169–11186
Ludwig W, Savara A, Madix RJ, Schauermann S, Freund H-J (2012) Subsurface hydrogen diffusion into Pd nanoparticles: role of low-coordinated surface sites and facilitation by carbon. J Phys Chem C 116:3539–3544. https://doi.org/10.1021/jp209033s
McKeehan L (1923) The crystal structures of the system palladium-hydrogen. Phys Rev 21:334
Methfessel M, Kubler J (1982) Bond analysis of heats of formation: application to some group VIII and IB hydrides. J Phys F: Met Phys 12:141
Methfessel M, Paxton AT (1989) High-precision sampling for Brillouin-zone integration in metals. Phys Rev B 40:3616–3621
Muniz-Miranda M, Gellini C, Canton P, Marsili P, Giorgetti E (2014) SERS and catalytically active Ag/Pd nanoparticles obtained by combining laser ablation and galvanic replacement. J Alloys Compd 615(supplement 1):S352–S356. https://doi.org/10.1016/j.jallcom.2013.12.064
Parambhath VB, Nagar R, Sethupathi K, Ramaprabhu S (2011) Investigation of spillover mechanism in palladium decorated hydrogen exfoliated functionalized graphene. J Phys Chem C 115:15679–15685. https://doi.org/10.1021/jp202797q
Perdew JP, Burke K, Wang Y (1996) Generalized gradient approximation for the exchange-correlation hole of a many-electron system. Phys Rev B 54:16533–16539
Pitt M, Gray EM (2003) Tetrahedral occupancy in the Pd-D system observed by in situ neutron powder diffraction EPL. Europhys Lett 64:344
Raimondi F, Scherer GG, Kötz R, Wokaun A (2005) Nanoparticles in energy technology: examples from electrochemistry and catalysis. Angew Chem Int Ed 44:2190–2209. https://doi.org/10.1002/anie.200460466
Savadogo O, Lee K, Oishi K, Mitsushima S, Kamiya N, Ota KI (2004) New palladium alloys catalyst for the oxygen reduction reaction in an acid medium. Electrochem Commun 6:105–109. https://doi.org/10.1016/j.elecom.2003.10.020
Semaltianos NG, Petkov P, Scholz S, Guetaz L (2013) Palladium or palladium hydride nanoparticles synthesized by laser ablation of a bulk palladium target in liquids. J Colloid Interface Sci 402:307–311. https://doi.org/10.1016/j.jcis.2013.03.062
Suslick K (1997) Handbook of heterogeneous catalysis vol 3. Wiley-VCH, Weinheim
Switendick A (1970) Electronic band structures of metal hydrides. Solid State Commun 8:1463–1467
Switendick A (1971) Metal hydrides—structure and band structure. Int J Quantum Chem 5:459–470
van den Berg AWC, Arean CO (2008) Materials for hydrogen storage: current research trends and perspectives. Chem Commun 6:668–681. https://doi.org/10.1039/B712576N
Williams A, Kübler J, Gelatt C Jr (1979) Cohesive properties of metallic compounds: augmented-spherical-wave calculations. Phys Rev B 19:6094
Worsham J Jr, Wilkinson M, Shull C (1957) Neutron-diffraction observations on the palladium-hydrogen and palladium-deuterium systems. J Phys Chem Solids 3:303–310
Zhao Y, Qin S-J, Li Y, Deng F-X, Liu Y-Q, Pan G-B (2014) Electrodeposition of dendritic Pd nanoarchitectures on n-GaN(0001): nucleation and electrocatalysis for direct formic acid fuel cells. Electrochim Acta 145:148–153. https://doi.org/10.1016/j.electacta.2014.09.008
Acknowledgements
The authors would like to thank Characterization Center for Materials and Biology at the University of Texas at Arlington for allowing the use of their facility. The computation was carried out in the High-Performance Computing Center at the University of Texas at Arlington and Stampede at TACC, TX.
Funding
MNH is partially supported by the National Science Foundation award no. 1609811.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chaudhary, R.P., Barman, S.K., Huda, M.N. et al. One-step hydrogen extraction and storage in plasma generated palladium nanoparticles. J Nanopart Res 20, 227 (2018). https://doi.org/10.1007/s11051-018-4322-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-018-4322-3