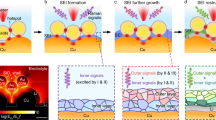
Abstract
Silicon, an attractive candidate for high-energy lithium-ion batteries (LIBs), displays an alloying mechanism with lithium and presents several unique characteristics which make it an interesting scientific topic and also a technological challenge. In situ local probe measurements have been recently developed to understand the lithiation process and propose an effective remedy to the failure mechanisms. One of the most specific techniques, which is able to follow the phase changes in poorly crystallized electrode materials, makes use of Raman spectroscopy within the battery, i.e., in operando mode. Such an approach has been successful but is still limited by the rather signal-to-noise ratio of the spectroscopy. Herein, the operando Raman signal from the silicon anodes is enhanced by plasmonic nanoparticles following the known surface-enhanced Raman spectroscopy (SERS). Coinage metals (Ag and Au) display a surface plasmon resonance in the visible and allow the SERS effect to take place. We have found that the as-prepared materials reach high specific capacities over 1000 mAh/g with stability over more than 1000 cycles at 1C rate and can be suitable to perform as anodes in LIB. Moreover, the incorporation of coinage metals enables SERS to take place specifically on the surface of silicon. Consequently, by using a specially designed Raman cell, it is possible to follow the processes in a silicon-coinage metal-based battery trough operando SERS measurements.









Similar content being viewed by others
References
Ashuri M, He Q, Shaw LL (2016) Silicon as a potential anode material for Li-ion batteries: where size, geometry and structure matter. Nanoscale 8:74–103. https://doi.org/10.1039/c5nr05116a
Bach P, Valencia-Jaime I, Rütt U et al (2016) Electrochemical lithiation cycles of gold anodes observed by in situ high-energy X-ray diffraction. Chem Mater 28:2941–2948. https://doi.org/10.1021/acs.chemmater.5b04719
Bok T, Choi S, Lee J, Park S (2014) Effective strategies for improving the electrochemical properties of highly porous Si foam anodes in lithium-ion batteries. J Mater Chem A 2:14195–14200. https://doi.org/10.1039/C4ta02392g
Bruce PGG, Scrosati B, Tarascon J-M (2008) Nanomaterials for rechargeable lithium batteries. Angew Chem Int Ed Engl 47:2930–2946. https://doi.org/10.1002/anie.200702505
Chai J, Buriak JM (2008) Using cylindrical domains of block copolymers to self-assemble and align metallic nanowires. ACS Nano 2:489–501. https://doi.org/10.1021/nn700341s
Chan CK, Ruffo R, Hong SS, Cui Y (2009) Surface chemistry and morphology of the solid electrolyte interphase on silicon nanowire lithium-ion battery anodes. J Power Sources 189:1132–1140. https://doi.org/10.1016/j.jpowsour.2009.01.007
Chockla AM, Bogart TD, Hessel CM et al (2012) Influences of gold, binder and electrolyte on silicon nanowire performance in Li-ion batteries. J Phys Chem C 116:18079–18086. https://doi.org/10.1021/jp305371v
Chou CY, Seo JH, Tsai YH et al (2015) Anomalous stagewise lithiation of gold-coated silicon nanowires: a combined in situ characterization and first-principles study. ACS Appl Mater Interfaces 7:16976–16983. https://doi.org/10.1021/acsami.5b01930
Ferraresi G, Czornomaz L, Villevieille C et al (2016) Elucidating the surface reactions of an amorphous Si thin film as a model electrode for Li-ion batteries. ACS Appl Mater Interfaces 8:29791–29798. https://doi.org/10.1021/acsami.6b10929
Gabudean AM, Biro D, Astilean S (2011) Localized surface plasmon resonance (LSPR) and surface-enhanced Raman scattering (SERS) studies of 4-aminothiophenol adsorption on gold nanorods. J Mol Struct 993:420–424. https://doi.org/10.1016/j.molstruc.2010.11.045
Gershinsky G, Bar E, Monconduit L, Zitoun D (2014) Operando electron magnetic measurements of Li-ion batteries. Energy Environ Sci 7:2012–2016. https://doi.org/10.1039/c4ee00490f
Guillot N, Shen H, Frémaux B et al (2010) Surface enhanced Raman scattering optimization of gold nanocylinder arrays: influence of the localized surface plasmon resonance and excitation wavelength. Appl Phys Lett 97:023113. https://doi.org/10.1063/1.3462068
Han Q, Zhang C, Gao W et al (2016) Ag-Au alloy nanoparticles: synthesis and in situ monitoring SERS of plasmonic catalysis. Sensors Actuators B Chem 231:609–614. https://doi.org/10.1016/j.snb.2016.03.068
Hochbaum AI, Gargas D, Hwang YJ, Yang P (2009) Single crystalline mesoporous silicon nanowires. Nano Lett 9:3550–3554. https://doi.org/10.1021/nl9017594
Hossain MK, Kitahama Y, Huang GG et al (2009) Surface-enhanced Raman scattering: realization of localized surface plasmon resonance using unique substrates and methods. Anal Bioanal Chem 394:1747–1760. https://doi.org/10.1007/s00216-009-2762-4
Huang J, Chiam SY, Tan HH et al (2010) Fabrication of silicon nanowires with precise diameter control using metal nanodot arrays as a hard mask blocking material in chemical etching. Chem Mater 22(13):4111–4116. https://doi.org/10.1021/cm101121c
Huang Z, Geyer N, Werner P et al (2011) Metal-assisted chemical etching of silicon: a review. Adv Mater 23:285–308. https://doi.org/10.1002/adma.201001784
Hy S, Chen YH, Liu J et al (2014) In situ surface enhanced Raman spectroscopic studies of solid electrolyte interphase formation in lithium ion battery electrodes. J Power Sources 256:324–328. https://doi.org/10.1016/j.jpowsour.2014.01.092
Jiao LS, Liu JY, Li HY et al (2016) Facile synthesis of reduced graphene oxide-porous silicon composite as superior anode material for lithium-ion battery anodes. J Power Sources 315:9–15. https://doi.org/10.1016/j.jpowsour.2016.03.025
Key B, Bhattacharyya R, Morcrette M et al (2009) Real-time NMR investigations of structural changes in silicon electrodes for lithium-ion batteries. J Am Chem Soc 131:9239–9249. https://doi.org/10.1021/ja8086278
Ko M, Chae S, Ma J et al (2016) Scalable synthesis of silicon-nanolayer-embedded graphite for high-energy lithium-ion batteries. Nat Energy 1:16113. https://doi.org/10.1038/nenergy.2016.113
Ma S, Cai Q, Lu K et al (2016) Bi-functional Au/FeS (Au/Co3O4) composite for in situ SERS monitoring and degradation of organic pollutants. J Nanopart Res 18:1–13. https://doi.org/10.1007/s11051-016-3335-z
Nanda J, Datta MK, Remillard JT et al (2009) In situ Raman microscopy during discharge of a high capacity silicon-carbon composite Li-ion battery negative electrode. Electrochem Commun 11:235–237. https://doi.org/10.1016/j.elecom.2008.11.006
Okamoto H (1990) The Li-Si (lithium-silicon) system. Bull Alloy Phase Diagr 11:306–312. https://doi.org/10.1007/BF03029305
Oumellal Y, Delpuech N, Mazouzi D et al (2011) The failure mechanism of nano-sized Si-based negative electrodes for lithium ion batteries. J Mater Chem 21:6201. https://doi.org/10.1039/c1jm10213c
Qiu L, Wang W, Zhang A et al (2016) Core-shell nanorod columnar array combined with gold nanoplate-nanosphere assemblies enable powerful in-situ SERS detection of bacteria. ACS Appl Mater Interfaces 8:24394–24403. https://doi.org/10.1021/acsami.6b06674
Rochow EG, Bailar JC Jr, Emeléus HJ, Nyholm R (1973) Comprehensive inorganic chemistry, vol 1. Pergamon, Oxford, p 1323
Sayed SY, Wang F, Malac M et al (2009) Heteroepitaxial growth of gold nanostructures on silicon by galvanic displacement. ACS Nano 3:2809–2817. https://doi.org/10.1021/nn900685a
Sharma B, Frontiera RR, Henry A-I et al (2012) SERS: materials, applications, and the future. Mater Today 15:16–25. https://doi.org/10.1016/S1369-7021(12)70017-2
Shi L, Wang W, Wang A et al (2016) Understanding the impact mechanism of the thermal effect on the porous silicon anode material preparation via magnesiothermic reduction. J Alloys Compd 661:27–37. https://doi.org/10.1016/j.jallcom.2015.11.196
Stiles PL, Dieringer JA, Shah NC, Van Duyne RP (2008) Surface-enhanced Raman spectroscopy. Annu Rev Anal Chem 1:601–626. https://doi.org/10.1146/annurev.anchem.1.031207.112814
Tan T, Tian C, Ren Z et al (2013) LSPR-dependent SERS performance of silver nanoplates with highly stable and broad tunable LSPRs prepared through an improved seed-mediated strategy. Phys Chem Chem Phys 15:21034–21042. https://doi.org/10.1039/c3cp52236a
Teki R, Datta MK, Krishnan R et al (2009) Nanostructured silicon anodes for lithium ion rechargeable batteries. Small 5:2236–2242. https://doi.org/10.1002/smll.200900382
Wilson AM, Zank G, Eguchi K et al (1997) Pyrolysed silicon-containing polymers as high capacity anodes for lithium-ion batteries. J Power Sources 68:195–200. https://doi.org/10.1016/S0378-7753(96)02551-7
Xie J, Tong L, Su L et al (2017) Core-shell yolk-shell Si@C@Void@C nanohybrids as advanced lithium ion battery anodes with good electronic conductivity and corrosion resistance. J Power Sources 342:529–536. https://doi.org/10.1016/j.jpowsour.2016.12.094
Yang J, Kraytsberg A, Ein-Eli Y (2015) In-situ Raman spectroscopy mapping of Si based anode material lithiation. J Power Sources 282:294–298. https://doi.org/10.1016/j.jpowsour.2015.02.044
Zheng MS, Dong QF, Cai HQ et al (2005) Formation and influence factors of solid electrolyte interphase film on the negative electrode surface in lithium-ion batteries. J Electrochem Soc 152:A2207–A2210. https://doi.org/10.1149/1.2060669
Acknowledgments
The authors acknowledge Dr. Hagit Aviv for her kind assistance with the Raman and Dr. Luba Burlaka for the TEM.
Funding
This work received partial funding from the Israel Committee of High Education and Prime Minister office within the framework of the INREP project. Y. M. would like to thank the Israeli Ministry of Science, Technology and Space for the financial support.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Miroshnikov, Y., Zitoun, D. Operando plasmon-enhanced Raman spectroscopy in silicon anodes for Li-ion battery. J Nanopart Res 19, 372 (2017). https://doi.org/10.1007/s11051-017-4063-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-017-4063-8