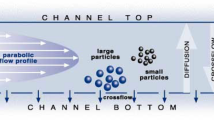
Abstract
Asymmetric flow field-flow fractionation (AF4) coupled with dynamic light scattering or multiangle light scattering detectors is a promising technique for the size-based separation of colloidal particles (nano- and submicron scale) and the online determination of the particle size of the separated fractions in aqueous suspensions. In most cases, the applications of these detectors are problematic due to the material-specific properties of the analyte that results in erroneous calculations, and as an alternative, different nanoparticle size standards are required to properly calibrate the size-based retention in AF4. The availability of nanoparticle size standards in different materials is limited, and this deviation from ideal conditions of retention is mainly due to material-specific and particle coating-specific membrane–particle interactions. Here, we present an experimental method on the applicability of polystyrene nanoparticles (PS NP) as standard for AF4 calibration and compare with gold nanoparticle (Au NP) standards having different nominal sizes and surface functionalities.


Similar content being viewed by others
References
Ariza MJ, Benavente J (2001) Streaming potential along the surface of polysulfone membranes: a comparative study between two different experimental systems and determination of electrokinetic and adsorption parameters. J Membrane Sci 190:119–132
Bendixen N, Losert S, Adlhart C, Lattuada M, Ulrich A (2014) Membrane–particle interactions in an asymmetric flow field flow fractionation channel studied with titanium dioxide nanoparticles. J Chromatogr A 1334:92–100
Chithrani BD, Ghazani AA, Chan WCW (2006) Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett 6:662–668. doi:10.1021/nl052396o
De M, Ghosh PS, Rotello VM (2008) Applications of nanoparticles in biology. Adv Mater 20:4225–4241
Du Q, Schimpf ME (2002) Correction for particle-wall interactions in the separation of colloids by flow field-flow fractionation. Anal Chem 74:2478–2485
Elimelech M, Gregory J, Jia X, Williams RA (1995) Particle deposition and aggregation—measurement, modelling and simulation. Elsevier, New York
Gallego-Urrea JA, Tuoriniemi J, Hassellöv M (2011) Applications of particle-tracking analysis to the determination of size distributions and concentrations of nanoparticles in environmental, biological and food samples TrAC. Trends Anal Chem 30:473–483
Gigault J, Hackley V (2013) Observation of size-independent effects in nanoparticle retention behavior during asymmetric-flow field-flow fractionation. Anal Bioanal Chem 405:6251–6258
Hakkinen H (2012) The gold-sulfur interface at the nanoscale. Nat Chem 4:443–455
Hotze EM, Phenrat T, Lowry GV (2010) Nanoparticle aggregation: challenges to understanding transport and reactivity in the environment. J Environ Qual 39:1909–1924
Lewinski N, Colvin V, Drezek R (2008) Cytotoxicity of nanoparticles. Small 4:26–49
Lin SY, Tsai YT, Chen CC, Lin CM, Chen Ch (2004) Two-step functionalization of neutral and positively charged thiols onto citrate-stabilized Au nanoparticles. J Phys Chem B 108:2134–2139
Litzen A, Wahlund KG (1991) Effects of temperature, carrier composition and sample load in asymmetrical flow field-flow fractionation. J Chromatogr A 548:393–406
Loeschner K et al (2013) Optimization and evaluation of asymmetric flow field-flow fractionation of silver nanoparticles. J Chromatogr A 1272:116–125
Lopez-Serrano A, Olivas RM, Landaluze JS, Camara C (2014) Nanoparticles: a global vision. Characterization, separation, and quantification methods. Potential environmental and health impact. Anal Methods 6:38–56
Mitrano DM, Barber A, Bednar A, Westerhoff P, Higgins CP, Ranville JF (2012) Silver nanoparticle characterization using single particle ICP-MS (SP-ICP-MS) and asymmetrical flow field flow fractionation ICP-MS (AF4-ICP-MS). J Anal Atom Spectrom 27:1131–1142
Mudalige TK, Qu H, Linder SW (2015a) Asymmetric flow field flow fractionation hyphenated ICP-MS as an alternative to cloud point extraction for quantification of silver nanoparticles and silver speciation: application for nanoparticles with protein corona. Anal Chem 87:7395–7401
Mudalige TK, Qu H, Linder SW (2015b) An improved methodology of asymmetric flow field flow fractionation hyphenated with inductively coupled mass spectrometry for the determination of size distribution of gold nanoparticles in dietary supplements. J Chromatogr A 1420:92–97
Mudalige TK, Qu H, Sánchez-Pomales G, Sisco PN, Linder SW (2015c) Simple functionalization strategies for enhancing nanoparticle separation and recovery with asymmetric flow field flow fractionation. Anal Chem 87:1764–1772
Petosa AR, Jaisi DP, Quevedo IR, Elimelech M, Tufenkji N (2010) Aggregation and deposition of engineered nanomaterials in aquatic environments: role of physicochemical interactions. Environ Sci Technol 44:6532–6549
Qu H, Mudalige TK, Linder SW (2014) Capillary electrophoresis/inductively-coupled plasma-mass spectrometry: development and optimization of a high resolution analytical tool for the size-based characterization of nanomaterials in dietary supplements. Anal Chem 86:11620–11627
Salgýn S, Salgýn U, Soyer N (2013) Streaming potential measurements of polyethersulfone ultrafiltration membranes to determine salt effects on membrane zeta potential. Int J Electrochem Sc 8:4073–4084
Sozer N, Kokini JL (2009) Nanotechnology and its applications in the food sector. Trends Biotechnol 27:82–89
Sze A, Erickson D, Ren L, Li D (2003) Zeta-potential measurement using the Smoluchowski equation and the slope of the current-time relationship in electroosmotic flow. J Colloid Interf Sci 261:402–410
Ulrich A et al (2012) Critical aspects of sample handling for direct nanoparticle analysis and analytical challenges using asymmetric field flow fractionation in a multi-detector approach. J Anal Atom Spectrom 27:1120–1130
Wahlund KG (2013) Flow field-flow fractionation: critical overview. J Chromatogr A 1287:97–112
Westermark G, Kariis H, Persson I, Liedberg B (1999) An infrared study on the chemisorption of tertiary phosphines on coinage and platinum group metal surfaces. Colloid Surface A 150:31–43
Acknowledgments
These studies were conducted using the Nanotechnology Core Facility (NanoCore) located on the US Food and Drug Administration’s Jefferson Laboratories campus (Jefferson, AR), which houses the FDA National Center for Toxicological Research and the FDA Office of Regulatory Affairs Arkansas Regional Laboratory. We thank Dr. Marilyn Khanna, Crystal Ford, Dr. Venu Gopal Bairi, Dr. Jin-hee Lim, and Dr. Yasith Nanayakkara for their support and valuable comments on the draft manuscript. The views expressed in this document are those of the researchers and should not be interpreted as the official opinion or policy of the US Food and Drug Administration, Department of Health and Human Services, or any other agency or component of the US Government. The mention of trade names, commercial products, or organizations is for clarification of the methods used and should not be interpreted as an endorsement of a product or manufacturer.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qu, H., Quevedo, I.R., Linder, S.W. et al. Importance of material matching in the calibration of asymmetric flow field-flow fractionation: material specificity and nanoparticle surface coating effects on retention time. J Nanopart Res 18, 292 (2016). https://doi.org/10.1007/s11051-016-3601-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-016-3601-0