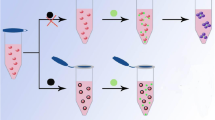
Abstract
Gold nanostars (AuNS) with a mean hydrodynamic size of 40 nm, obtained with a seed-growth approach using a zwitterionic surfactant (laurylsulfobetaine, LSB), were successfully coated with glutathione (GSH), obtaining a stable and purified solid product which can be easily stored and re-dissolved on need in 0.1 M aqueous solution of Hepes buffered at pH 7. Upon exposure to micromolar concentrations of Pb2+ cation, the GSH-coated nano-objects undergo a fast aggregation followed by sedimentation leading to complete precipitation in about an hour. The subsequent disappearing of the intense LSPR extinction can of course be followed spectrophotometrically but, most importantly, can be easily detected with the naked eye. No signs of this event are noticed when other divalent cations are added to the colloidal suspension in the same condition. A careful investigation was performed to study this selectivity and the behaviour of aggregation as a function of time and Pb2+ cation concentration. We demonstrate that an easy, rapid, instrument-free, visual detection of micromolar levels of Pb2+ is thus possible with this assay, showing a good selectivity towards other investigated metal cations.








Similar content being viewed by others
References
Amato E, Diaz-Fernandez YA, Taglietti A, Pallavicini P, Pasotti L, Cucca L, Milanese C, Grisoli P, Dacarro C, Fernandez-Hechavarria JM et al (2011) Synthesis, characterization and antibacterial activity against gram positive and gram negative bacteria of biomimetically coated silver nanoparticles. Langmuir 27(15):9165–9173
Arduini A, Demuru D, Pochini A, Secchi A (2005) Recognition of quaternary ammonium cations by calix 4 arene derivatives supported on gold nanoparticles. Chem Commun 5:645–647
Beqa L, Singh AK, Khan SA, Senapati D, Arumugam SR, Ray PC (2011) Gold nanoparticle-based simple colorimetric and ultrasensitive dynamic light scattering assay for the selective detection of Pb(II) from paints, plastics, and water samples. Acs Appl Mater Interfaces 3(3):668–673
Casu A, Cabrini E, Dona A, Falqui A, Diaz-Fernandez Y, Milanese C, Taglietti A, Pallavicini P (2012) Controlled synthesis of gold nanostars by using a Zwitterionic surfactant. Chem A Eur J 18(30):9381–9390
Cavallaro G, Triolo D, Licciardi M, Giammona G, Chirico G, Sironi L, Dacarro G, Dona A, Milanese C, Pallavicini P (2013) Amphiphilic copolymers based on poly (hydroxyethyl)-D, L-aspartamide : a suitable functional coating for biocompatible gold nanostars. Biomacromolecules 14(12):4260–4270
Chai F, Wang CA, Wang TT, Li L, Su ZM (2010) Colorimetric detection of Pb2+ using glutathione functionalized gold nanoparticles. Acs Appl Mater Interfaces 2(5):1466–1470
Daniel MC, Astruc D (2004) Gold nanoparticles: Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev 104(1):293–346
Durgadas CV, Lakshmi VN, Sharma CP, Sreenivasan K (2011) Sensing of lead ions using glutathione mediated end to end assembled gold nanorod chains. Sens Actuators B Chem 156(2):791–797
Edwards M, Triantafyllidou S, Best D (2009) Elevated blood lead in young children due to lead-contaminated drinking water: Washington, DC, 2001–2004. Environ Sci Technol 43(5):1618–1623
Elghanian R, Storhoff JJ, Mucic RC, Letsinger RL, Mirkin CA (1997) Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 277(5329):1078–1081
El-Sayed MA (2001) Some interesting properties of metals confined in time and nanometer space of different shapes. Acc Chem Res 34(4):257–264
Fabbrizzi L, Pallavicini P, Parodi L, Perotti A, Taglietti A (1995) Molecular recognition of the imidazolo residue by a dicopper(II) complex with a bisdien macrocycle bearing 2 pendant arms. J Chem Soc Chem Commun 23:2439–2440
Fabbrizzi L, Licchelli M, Rabaioli G, Taglietti A (2000) The design of luminescent sensors for anions and ionisable analytes. Coord Chem Rev 205:85–108
Fu R, Li J, Yang W (2012) Aggregation of glutathione-functionalized Au nanoparticles induced by Ni2+ ions. J Nanopart Res 14:929
Guan J, Jiang L, Li J, Yang W (2008) pH-dependent aggregation of histidine-functionalized Au nanoparticles induced by Fe3+ ions. J of Phys Chem C 112(9):3267–3271
Guidotti TL, Calhoun T, Davies-Cole JO, Knuckles ME, Stokes L, Glymph C, Lum G, Moses, Goldsmith DF, Ragain L (2009) Elevated lead in drinking water in Washington, DC, 2003-2004: The public health response (vol 115, pg 695, 2007). Environ Health Perspect 117(8):A342–A342
Hutter E, Pileni MP (2003) Detection of DNA hybridization by gold nanoparticle enhanced transmission surface plasmon resonance spectroscopy. J Phys Chem B 107(27):6497–6499
Kim YJ, Johnson RC, Hupp JT (2001) Gold nanoparticle-based sensing of “spectroscopically silent” heavy metal ions. Nano Lett 1(4):165–167
Lee J-S, Han MS, Mirkin CA (2007) Colorimetric detection of mercuric ion (Hg2+) in aqueous media using DNA-functionalized gold nanoparticles. Angew Chem Int Edition 46(22):4093–4096
Li H, Cui Z, Han C (2009) Glutathione-stabilized silver nanoparticles as colorimetric sensor for Ni(2 +) ion. Sens Actuators B Chem 143(1):87–92
Li JL, Chen LX, Lou TT, Wang YQ (2011) Highly sensitive SERS detection of As3 + ions in aqueous media using glutathione functionalized silver nanoparticles. Acs Appl Mater Interfaces 3(10):3936–3941
Liu C-W, Huang C-C, Chang H-T (2008) Control over surface DNA density on gold nanoparticles allows selective and sensitive detection of mercury(II). Langmuir 24(15):8346–8350
Marcotte N, Taglietti A (2003) Transition-metal-based chemosensing ensembles: ATP sensing in physiological conditions. Supramol Chem 15(7–8):617–625
Pallavicini P, Chirico G, Collini M, Dacarro G, Dona A, D’Alfonso L, Falqui A, Diaz-Fernandez Y, Freddi S, Garofalo B et al (2011) Synthesis of branched Au nanoparticles with tunable near-infrared LSPR using a zwitterionic surfactant. Chem Commun 47(4):1315–1317
Plascencia-Villa G, Bahena D, Rodriguez AR, Ponce A, Jose-Yacaman M (2013) Advanced microscopy of star-shaped gold nanoparticles and their adsorption-uptake by macrophages. Metallomics 5(3):242–250
Saha K, Agasti SS, Kim C, Li XN, Rotello VM (2012) Gold nanoparticles in chemical and biological sensing. Chem Rev 112(5):2739–2779
Sokolowska M, Bal W (2005) Cu(II) complexation by “non-coordinating” N-2-hydroxyethylpiperazine-N’-2-ethanesulfonic acid (Hepes buffer). J Inorg Biochem 99(8):1653–1660
Sung HK, Oh SY, Park C, Kim Y (2013) Colorimetric detection of Co2+ ion using silver nanoparticles with spherical, plate, and rod shapes. Langmuir 29(28):8978–8982
Taglietti A, Fernandez YAD, Cucca L, Dacarro G, Grisoli P, Necchi V, Pallavicini P, Pasotti L, Patrini M (2012) Antibacterial activity of glutathione-coated silver nanoparticles against gram positive and gram negative bacteria. Langmuir 28:8140–8148
Taglietti A, Fernandez YAD, Galinetto P, Grisoli P, Milanese C, Pallavicini P (2013) Mixing thiols on the surface of silver nanoparticles: preserving antibacterial properties while introducing SERS activity. J Nanoparticle Res 15(11):1–13
Tokareva I, Minko S, Fendler JH, Hutter E (2004) Nanosensors based on responsive polymer brushes and gold nanoparticle enhanced transmission surface plasmon resonance spectroscopy. J Am Chem Soc 126(49):15950–15951
Yang WR, Gooding JJ, He ZC, Li Q, Chen GN (2007) Fast colorimetric detection of copper ions using l-cysteine functionalized gold nanoparticles. J Nanosci Nanotechnol 7(2):712–716
Acknowledgments
Funding had been granted by Fondazione Cariplo (Bandi Ricerca Scientifica e Tecnologica sui Materiali Avanzati, 2010-0454), the Ministero dell’Istruzione, Università e Ricerca (MIUR). Authors also want to thank Vittorio Necchi and Centro Grandi Strumenti, for TEM images.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
D’Agostino, A., Taglietti, A., Bassi, B. et al. A naked eye aggregation assay for Pb2+ detection based on glutathione-coated gold nanostars. J Nanopart Res 16, 2683 (2014). https://doi.org/10.1007/s11051-014-2683-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-014-2683-9