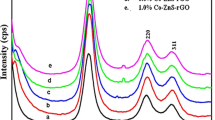
Abstract
The present work deals with syntheses of CdS/graphene and ZnS/graphene nanocomposites by hydrothermal reaction of graphene oxide and morpholine-4-carbodithioate complex of Cd and Zn, respectively. The corresponding nanocomposites has been investigated separately as photocatalyst in the decomposition of methylene blue in the presence of UV light and also as adsorbents in the removal of Cd(II) and Pb(II) ions in contaminated water. These studies have established that CdS/graphene and ZnS/graphene are effective photocatalyst as well as effective adsorbents in the removal of Cd(II) and Pb(II) ions to an extent of 97 and 99 % by ZnS/graphene and CdS/graphene nanocomposite, respectively, under 1 g L−1 of adsorption dose and at pH ~7. Further studies also established Langmuir model befitting for the adsorption of Pb(II) and Cd(II) ions on CdS/graphene and ZnS/graphene, respectively. The presence of interfering ions on extent of Cd(II) and Pb(II) removal has also been reported.



















Similar content being viewed by others
References
Aravamudan G, Brown DH, Venkappayya D (1971) Metal complexes of morpholine-4-carbodithioate. J Chem Soc A 17:2744–2747
Barrelet CJ, Wu Y, Bell DC, Lieber CM (2003) Synthesis of CdS and ZnS nanowires using single-source molecular precursors. J Am Chem Soc 125:11498–11499
Bhattacharya R, Saha S (2008) Growth of CdS nanoparticles by chemical method and its characterization. Pranama J Phys 71:187–192
Bourlinos AB, Gournis D, Petridis D, Szabo T, Szeri A, Dekany I (2003) Graphite oxide: chemical reduction to graphite and surface modification with primary aliphatic amines and amino acids. Langmuir 19:6050–6055
Budhraja J, Singh M (2004) Spectrophotometric study of adsorption of bromo thymol blue on charcoal and phosphates of FeIII, AlIII, CrIII and ZnII. J Indian Chem Soc 81:573–575
Chakrabarty S, Sarma HP (2011) Heavy metal contamination of drinking water in Kamprup district, Assam, India. Environ Monit Assess 179:479–486
Chen PC, Mwakwari SC, Oyelere AK (2008) Gold nanoparticles: from nanomedicine to nanosensing. Nanotechnol Sci Appl 1:45–66
Chen D, Huang F, Ren G, Li D, Zheng M, Wang Y, Lin Z (2010a) ZnS nano-architectures: photocatalysis, deactivation and regeneration. Nanoscale 10:2062–2064
Chen G, Zhang W, Zhong X (2010b) Single-source precursor route for overcoating CdS and ZnS shells around CdSe core nanocrystals. Front Chem China 5:214–220
Feng M, Sun R, Zhan H, Chen Y (2010) Lossless synthesis of graphene nanosheets decorated with tiny cadmium sulfide quantum dots with excellent nonlinear optical properties. Nanotecnology 21:1–7
Ge T, Kuai L, Geng B (2011) Solution-phase chemical route to branched single-crystalline CdS nanoarchitectures and their field emission property. J Alloy Compd 509:L353–L358
Han MY, Quek CH (2000) Photochemical synthesis in formamide and room-temperature coulomb staircase behavior of size-controlled gold nanoparticles. Langmuir 16:362–367
Hu H, Wang X, Liu F, Wang J, Xu C (2011) Rapid microwave-assisted synthesis of graphene nanosheets-zinc sulfide nanocomposites: optical and photocatalytic properties. Synth Met 161:404–410
Hudlikar M, Jogleker S, Dhaygude M, Kodam K (2012) Latex-mediated synthesis of ZnS nanoparticles: green synthesis approach. J Nanopart Res 14:865/1–865/6
Kaveri S, Thirugnanam L, Dutta M, Ramasamy J, Fukata N (2013) Thiourea assisted one-pot easy synthesis of CdS/rGO composite by the wet chemical method: structural, optical and photocatalytic properties. Ceram Int. doi:10.1016/j.ceramint.2013.05.025
Kole AK, Kumbhakar P (2012) Effect of manganese doping on the photoluminescence characteristics of chemically synthesized zinc sulfide nanoparticles. Appl Nanosci 2:15–23
Kuila T, Bose S, Mishra AK, Khanra P, Kim NH, Lee JH (2012) Chemical functionalization of graphene and its applications. Prog Mater Sci 57:1061–1105
Lee HS, Woo CS, Youn BK, Kim SY, Oh ST, Sung YE, Lee HI (2005) Bandgap modulation of TiO2 and its effect on the activity in photocatalytic oxidation of 2-isopropyl-6-methyl-4-pyrimidinol. Top Catal 35:255–260
Liu F, Shao X, Wang J, Yang S, Li H, Meng X, Liu X, Wang M (2013) Solvothermal synthesis of graphene-CdS nanocomposites for highly efficient visible-light photocatalyst. J Alloys Comp 551:327–332
Lv XJ, Chang WF, Fu HX, Zhang H, Cheng JS, Zhang GJ, Song Y, Hu CY, Li JH (2012) Hydrogen evolution from water using semiconductor nanoparticle/graphene composite photocatalysts without noble metals. J Mater Chem 22:1539–1546
Malik MA, Revaprasadu N, O’Brien P (2001) Air-stable single-source precursors for the synthesis of chalcogenide semiconductor nanoparticles. Chem Mater 13:913–920
Mandal S, Sahu MK, Patel RK (2013) Adsorption studies of arsenic (III) removal from water by zirconium polyacrylamide hybrid material (ZrPACM-43). Water Res Ind. doi:10.1016/j.wri.2013.09.003
Mazumder DNG, Ghosh A, Majumdar KK, Ghosh N, Saha C, Mazumder RNG (2010) Arsenic contamination of ground water and its health impact on population of district of Nadia, West Bengal, India. Indian J Community Med 35:331–338
Mehta SK, Khushboo UA, Kumar S (2012) A mechanistic study of photoluminescence quenching of cetyl trimethyl ammonium bromide stabilized SnS nanoparticles with β-cyclodextrin. J Nanosci Nanotechnol 12:1760–1764
Memon GZ, Bhanger MI, Akhtar M (2009) Peach-nut shells-an effective and low cost adsorbent for the removal of endosulfan from aqueous solutions. Pak J Anal Environ Chem 10:14–18
Miranda G, Quiroz A, Salazar M (2000) Cadmium and lead removal from water by duck week. Lemna gibba L. (Lemnaceae). Hidrobiologica 10:7–12
Nair PST, Radhakrishanan N, Revaprasadu GA, Kolawole, Brien PO’ (2003) Cd(NH2CSNHNHCSNH2)Cl2: a new single-source precursor for the preparation of CdS nanoparticles. Polyhedron 22:3129–3135
Nethravathi C, Nisha T, Ravishankar N, Shivakumara C, Rajamathi M (2009) Grapene-nanocrystalline metal sulphide composites produced by one-pot reaction starting from graphite oxide. Carbon 47:2054–2059
Onwudiwe DC, Strydom C, Oluwafemi OS, Songca SP (2012) Effect of temperature on the optical and structural properties of hexadecylamine capped ZnS nanoparticles using zinc(II) N-ethyl-N-phenyldithiocarbamate as single source precursor. Mater Res Bull 47:4445–4451
Paalman MAA, Van der Waeijden CH, Loch JPG (1994) Sorption of cadmium on suspended matter under estuarine conditions; competition and complexation with major seawater ions. Water Air Soil Poll 73:49–60
Pan S, Liu X (2012) ZnS-Graphene nanocomposites: synthesis, Characterization and optical properties. J Solid State Chem 191:51–56
Park S, Ruoff RS (2009) Chemical methods for the production of graphenes. Nat Nanotechnol 4:217–224
Pickett NL, Brien PO’ (2001) Syntheses of semiconductor nanoparticles using single-molecular precursors. Chem Rec 1:467–469
Raul PK, Devi RR, Umlong LM, Banerjee S, Singh L, Purkait M (2012) Removal of fluoride from water using iron oxide-hydroxide nanoparticles. J Nanosci Nanotechnol 12:1–9
Reindle E, Troll WJ (1975) Metabolic reduction of benzidine azo dyes to benzidine in the Rhesus monkey. J Natl Cancer Inst 55:181–182
Sahoo AK, Srivastava SK (2013) Controllable architecture of CdS and CuS by single-source precursor-mediated approach and their photocatalytic activity. J Nanopart Res 15:1591–1605
Sahu MK, Mandal S, Dash SS, Badhai P, Patel RK (2013) Removal of Pb(II) from aqueous solution by acid activated red mud. J Environ Chem Eng. doi:10.1016/j.jece.2013.09.027
Shihabudheen MM, Kinattukara PL, Pradeep T (2010) A novel cellulose-manganese oxide hybrid material by in situ soft chemical synthesis and its application for the removal of Pb(II) from water. J Hazard Mater 181:955–986
Singanan M (2011) Removal of Lead (II) and cadmium (II) ions from waste water using activated biocarbon. Sci Asia 37:115–119
Singh AK (2004) Arsenic contamination in ground of North Eastern India. In: Proceedings of National seminar on hydrology with focal theme on “Water Quality” held at National Institute of Hydrology, Roorkee during November 22–23
Singh N, Rastogi K, Kumar R (1981) Separation and determination of selenium (IV) and tellurium (IV) carbodithioates by thin-layer chromatography and visible spectrophotometry using morpholine-4-carbodithioate. Analyst 106:599–602
Soltani N, Saion E, Erfani M, Rezaee K, Bahmanorokh G, Drummen GPC, Bahrami A, Hussein MZ (2012) Influence of the polyvinylpyrrolidone concentration on particle size and dispersion of ZnS nanoparticles synthesized by microwave irradiation. Int J Mol Sci 13:12412–12427
Tembhurkar AR, Shilpa D (2006) Studies on fluoride removal using adsorption process. J Environ Sci Eng 48:151–156
Tong H, Zhu YJ (2006) Synthesis of CdS nanocrystals based on low-temperature thermolysis of one single-source organometallic precursor. Nanotechnology 17:845–851
Ushakov NM, Yurkov G, Yu KV, Zaapsis DA, Kataeva NA, Kosobudskii ID, Gubin SP (2006) Optical properties of cadmium sulfide nanoparticles on the surface of polytetrafluoroethylene nanogranules. Opt Spectrosc 100:414–418
Vazquez A, Lopez I, Gomez I (2012) Cadmium sulfide and zinc sulfide nanostructures formed by electrophoretic deposition. Key Eng Mat 507:101–105
Venkatram N, Rao DN, Akundi MA (2005) Nonlinear absorption, scattering and optical limiting studies of CdS nanoparticles. Opt Express 13:867–872
Wang P, Jiang T, Zhu C, Zhai Y, Wang D, Dong S (2010) One-step, solvothermal synthesis of Graphene-CdS and Graphene-ZnS quantum dot nanocomposites and their interesting photovoltaic properties. Nanoscale 3:794–799
Wang L, Li J, Jianga Q, Zhao L (2012) Water-soluble Fe3O4 nanoparticles with high solubility for removal of heavy-metal ions from waste water. Dalton Trans 41:4544–4551
Wang Y, Wang Q, Zhan X, Wang F, Safdar M, He J (2013a) Visible light driven type II heterostructures and their enhanced photocatalysis properties: a review. Nanoscale 5:8326–8339
Wang Y, Wang F, Jun He (2013b) Controlled fabrication and photocatalytic properties of a three-dimensional ZnO nanowire/reduced graphene oxide/CdS heterostructure on carbon cloth. Nanoscale 5:11291–11297
Wang F, Wang Y, Zhan X, Safdar M, Gong J, He J (2014) Pt nanoparticle and CdS quantum dot assisted WO3 nanowires grown on flexible carbon fibers for efficient oxygen production. Cryst Eng Commun 16:1389–1394
WHO (2011) Arsenic in drinking-water: background document for development of WHO guidelines for drinking-water quality. World Health Organization
Williams G, Kamat PV (2009) Graphene–semiconductor nanocomposites: excited-state interactions between ZnO nanoparticles and graphene oxide. Langmuir 25:13869–13873
Xu Y, Yang L, Yang J (2010) Removal of cadmium (II) from aqueous solution by two kinds of manganese coagulants. Int J Eng Sci Technol 2:1–8
Xue L, Shen C, Zheng M, Lu H, Li N, Ji G, Pan L, Cao J (2011) Hydrothermal synthesis of graphene-ZnS quantum dot nanocomposites. Mater Lett 65:198–200
Yadav AK, Kaushik CP, Haritash AK, Kansal A, Rani N (2006) Defluoridation of ground water using brick powder as an adsorbent. J Hazard Mater 128:289–293
Yang MQ, Xu YJ (2013) Selective photoredox using graphene-based composite photocatalysts. Phys Chem Chem Phys 15:19102–19118
Ye A, Fan W, Zhang Q, Deng W, Wang Y (2012) CdS-graphene and CdS-CNT nanocomposites as visible-light photocatalysts for hydrogen evolution and organic dye degradation. Catal Sci Technol 2:969–978
Yin Y, Ling X, Ge C, Xia C, Zhang Z (1998) Synthesis of cadmium sulfide nanoparticles in situ using γ-radiation. Chem Commun 16:1641–1642
Yu L, Ruan H, Zheng Y, Li D (2013) A facile solvothermal method to produce ZnS quantum dots-decorated graphene nanosheets with superior photoactivity. Nanotechnology. doi:10.1088/0957-4484/24/37/375601
Yuan-yuan S, Yang J, Ke-qiang Q (2010) Synthesis of ZnS nanoparticles by solid-liquid chemical reaction with ZnO and Na2S under ultrasonic. T Nonferr Metal Soc 20:s215–s221
Zhang K, Liu X (2011) One step synthesis and characterization of CdS nanorod/graphene nanosheet composite. Appl Surf Sci 257:10379–10383
Zhang YC, Chen WW, Hu XY (2007) Controllable synthesis and optical properties of Zn-doped CdS nanorods from single-source molecular precursors. Cryst Growth Des 7:580–586
Zhang Y, Tang ZR, Fu X, Xu YJ (2010) TiO2-graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: is TiO2-graphene truly different from other TiO2-carbon composite materials. ACS Nano 4:7303–7314
Zhang N, Zhang Y, Pan X, Fu X, Liu S, Xu YJ (2011) Assembly of CdS nanoparticles on the two-dimensional graphene scaffold as visible-light-driven photocatalyst for selective organic transformation under ambient conditions. J Phys Chem C 115:23501–23511
Zhang Y, Liu W, Wang R (2012a) From ZnS nanoparticles, nanobelts to nanotetrapods: the ethylenediamine modulated anisotropic growth of ZnS nanostructures. Nanoscale 4:2394–2399
Zhang Y, Zhang N, Tang ZR, Xu YJ (2012b) Graphene transforms wide band gap ZnS to a visible light photocatalyst. The new role of graphene as a macromolecular photosensitizer. ACS Nano 6:9777–9789
Zhang N, Zhang Y, Xu YJ (2012c) Recent progress on graphene-based photocatalysts: current status and future perspectives. Nanoscale 4:5492–5813
Zhang N, Yang MQ, Tang Z, Xu YJ (2014) Toward improving the graphene semiconductor composite photoactivity via the addition of metal ions as generic interfacial mediator. ACS Nano 8:623–633
Zhiyong G, Liu N, Wu D, Tao W, Xu F, Jiang K (2012) Graphene-CdS composite, synthesis and enhanced photocatalytic activity. App Surf Sci 258:2473–2478
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahoo, A.K., Srivastava, S.K., Raul, P.K. et al. Graphene nanocomposites of CdS and ZnS in effective water purification. J Nanopart Res 16, 2473 (2014). https://doi.org/10.1007/s11051-014-2473-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-014-2473-4