Abstract
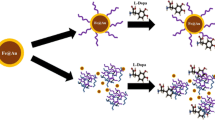
Polymer-coated inorganic nanoparticles are widely studied as carrier systems for various biomedical applications. For their translation into clinical applications, it is critical that they remain in a stable, non-agglomerated state and are also produced at a high yield with ease. Herein, we introduce a novel methodology called the isolate-functionalize-release (ISOFURE) strategy to synthesize stable hydrogel-coated nanoparticles in the absence of a stabilizing reagent. As a model platform, we isolated and functionalized gold nanoparticles (GNPs) in a degradable poly(beta amino ester) (PBAE) hydrogel composite. Isolation was done either by adding GNPs to the PBAE hydrogel precursor solution or via in situ precipitation of GNPs inside the pre-synthesized PBAE hydrogel. Atom transfer radical polymerization (ATRP) was used as the model reaction to grow a temperature responsive poly(ethylene glycol) 600 dimethacrylate crosslinked poly(N-isopropyl acrylamide) hydrogel shell over the GNP core. Release of the temperature responsive hydrogel-coated GNPs was done by degrading the PBAE ISOFURE matrix in water. Characterization of the various nanoparticles using UV–Vis spectroscopy confirmed in situ precipitation of GNPs as well as ATRP over the GNPs. Light scattering confirmed the presence of temperature sensitive hydrogel shell over the GNP surface. Aging studies showed that the ISOFURE method based crosslinked hydrogel-coated GNPs showed higher stability and yield than that of the solution-based functionalized GNPs. The use of ISOFURE strategy shows promise as a simple tool for enhanced functionalization and stabilization over various nanocarriers under flexible experimental conditions.





Similar content being viewed by others
References
Anderson DG, Tweedie CA, Hossain N, Navarro SM, Brey DM, Vliet KJV, Langer R, Burdick JA (2006) A combinatorial library of photocrosslinkable and degradable materials. Adv Mater 18:2614–2618
Boisselier E, Diallo AK, Salmon L, Ornelas C, Ruiz J, Astruc D (2010) Encapsulation and stabilization of gold nanoparticles with “click” polyethyleneglycol dendrimers. J Am Chem Soc 132:2729–2742
Chakraborty S, Bishnoi SW, Perez-Luna VH (2010) Gold nanoparticles with poly(N-isopropylacrylamide) formed via surface initiated atom transfer free radical polymerization exhibit unusually slow aggregation kinetics. J Phys Chem C 114:5947–5955
Chirra HD, Hilt JZ (2010) Nanoscale characterization of the equilibrium and kinetic response of hydrogel structures. Langmuir 26:11249–11257
Chirra HD, Biswal D, Hilt JZ (2009) In: Pathak Y, Thassu D (eds) Nanoparticulate drug delivery systems (NPDDS) II: formulation and characterization. Informa Healthcare USA Inc., New York, pp 90–114
Chirra HD, Biswal D, Hilt JZ (2011) Controlled synthesis of responsive hydrogel nanostructures via microcontact printing and ATRP. Polym Adv Technol 22:773–780
De M, Ghosh PS, Rotello VM (2008) Applications of nanoparticles in biology. Adv Mater 20:4225–4241
Dong H, Zhu M, Yoon JA, Gao H, Jin R, Matyjaszewski K (2008) One-pot synthesis of robust core/shell gold nanoparticles. J Am Chem Soc 130:12852–12853
Dzidic I (1972) Relative gas-phase basicities of some amines, anilines, and pyridines. J Am Chem Soc 94:8333–8335
El-Sayed IH, Huang X, El-Sayed MA (2006) Selective laser photo-thermal therapy of epithelial carcinoma using anti-EGFR antibody conjugated gold nanoparticles. Cancer Lett 239:129–135
Hawkins AM, Satarkar NS, Hilt JZ (2009) Demonstration of remote-controlled drug release from degradable nanocomposite hydrogels. Pharm Res 26:667–673
Houbenov N, Minko S, Stamm M (2003) Mixed polyelectrolyte brush from oppositely charged polymers for switching of surface charge and composition in aqueous environment. Macromolecules 36:5897–5901
Jordan R, West N, Ulman A, Chou Y-M, Nuyken O (2001) Nanocomposites by surface-initiated living cationic polymerization of 2-oxazolines on functionalized gold nanoparticles. Macromolecules 34:1606–1611
Ke YC, Stroeve P (2005) Characterization and techniques: polymer-layered silicate and silica nanocomposites. Elsevier Science, Amsterdam, pp 275–327
Kim JH, Lee TR (2004) Thermo- and pH-responsive hydrogel-coated gold nanoparticles. Chem Mater 16:3647–3651
Kim JH, Lee TR (2006) Discrete thermally responsive hydrogel-coated gold nanoparticles for use as drug-delivery vehicles. Drug Dev Res 67:61–69
Krsko P, Libera M (2005) Biointeractive hydrogels. Mater Today 8:36–44
Li DX, He Q, Cui Y, Li J (2007) Thermosensitive copolymer networks modified gold nanoparticles for nanocomposite entrapment. Chem Eur J 13:2224–2229
Moghimi SM, Hunter AC, Murray JC (2005) Nanomedicine: current status and future prospects. FASEB J 19:311–330
Ohno K, Koh K-M, Tsuji Y, Fukuda T (2002) Synthesis of gold nanoparticles coated with welldefined, high-density polymer brushes by surface-initiated living radical polymerization. Macromolecules 35:8989–8993
Sanvicens N, Marco MP (2008) Multifunctional nanoparticles—properties and prospects for their use in human medicine. Trends Biotechnol 26:425–433
Satarkar NS, Hilt JZ (2008) Magnetic hydrogel nanocomposites for remote controlled pulsatile drug release. J Control Release 3:246–251
Sidorenko A, Krupenkin T, Taylor A, Fratzl P, Aizenberg J (2007) Reversible switching of hydrogel-actuated nanostructures into complex micropatterns. Science 315:487–490
Slaughter BV, Khurshid SS, Fisher OZ, Khademhosseini A, Peppas NA (2009) Hydrogels in regenerative medicine. Adv Mater 21:3307–3329
Zhao M, Sun L, Crooks RM (1998) Preparation of Cu nanoclusters within dendrimer templates. J Am Chem Soc 120:4877–4878
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chirra, H.D., Spencer, D. & Hilt, J.Z. Isolate, functionalize, and release: the ISOFURE platform for the functionalization of nanoparticles. J Nanopart Res 14, 1230 (2012). https://doi.org/10.1007/s11051-012-1230-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-012-1230-9