Abstract
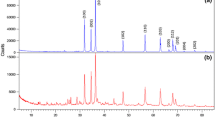
In this study, we report the synthesis of well-aligned nanocrystalline hexagonal zinc oxide (ZnO) nanoparticles by facile solid-state and co-precipitation method. The co-precipitation reactions were performed using aqueous and ethylene glycol (EG) medium using zinc acetate and adipic acid to obtain zinc adipate and further decomposition at 450 °C to confer nanocrystalline ZnO hexagons. XRD shows the hexagonal wurtzite structure of the ZnO. Thermal study reveals complete formation of ZnO at 430 °C in case of solid-state method, whereas in case of co-precipitation method complete formation was observed at 400 °C. Field emission scanning electron microscope shows spherical morphology for ZnO synthesized by solid-state method. The aqueous-mediated ZnO by co-precipitation method shows rod-like morphology. These rods are formed via self assembling of spherical nanoparticles, however, uniformly dispersed spherical crystallites were seen in EG-mediated ZnO. Transmission electron microscope (TEM) investigations clearly show well aligned and highly crystalline transparent and thin hexagonal ZnO. The particle size was measured using TEM and was observed to be 50–60 nm in case of solid-state method and aqueous-mediated co-precipitation method, while 25–50 nm in case of EG-mediated co-precipitation method. UV absorption spectra showed sharp absorption peaks with a blue shift for EG-mediated ZnO, which demonstrate the mono-dispersed lower particle size. The band gap of the ZnO was observed to be 3.4 eV which is higher than the bulk, implies nanocrystalline nature of the ZnO. The photoluminescence studies clearly indicate the strong violet and weak blue emission in ZnO nanoparticles which is quite unique. The process investigated may be useful to synthesize other oxide semiconductors and transition metal oxides.











Similar content being viewed by others
References
Bougrine A, El Hichou A, Addou M, Ebothe J, Kachouane A, Troyon M (2003) Structural, optical and cathodoluminescence characteristics of undoped and tin-doped ZnO thin films prepared by spray pyrolysis. Mater Chem Phys 80:438
Chen Y, Shen C, Chen H, Wei Y, Wu L (1991) Grain growth and electrical properties in ZnO varistors with various valence states of additions. Jpn J Appl Phys 30:84–90
Cullity B (1977) Elements of X-ray diffraction. Addison-Wesley, Reading MA, p 102
Dosch R, Tuttle B, Brooks R (1986) Chemical preparation and properties of high-field zinc oxide varistors. J Mater Res 1:90–99
Haile S, Johnson D, Wiseman G, Bowen H (1989) Aqueous precipitation of spherical zinc oxide powders for varistor applications. J Am Ceram Soc 72:2004–2008
Hembram K, Sivaprahasam D, Rao T (2011) Combustion synthesis of doped nanocrystalline ZnO powders for varistors applications. J Eur Ceram Soc 31(10):1905–1913
Hingorani S, Pillai V, Kumar P, Multani M, Shah D (1993) Microemulsion mediated synthesis of zinc oxide nanoparticles for varistor studies. Mater Res Bull 28:1303
Hu X, Zhu Y, Wang S (2004) Sonochemical and microwave-assisted synthesis of linked single-crystalline ZnO rods. Mater Chem Phys 88:421
Huang M, Mao S, Feick H, Yan H, Wu Y, Kind H, Weber E, Russo R, Yang P (2001) Room-temperature ultraviolet nanowire nanolasers. Science 292:1897–1899
JCPDS card No. 36.1451
Kanade K, Kale B, Aiyer R, Das B (2006) Effect of solvents on the synthesis of nano-size zinc oxide and its properties. Mater Res Bull 41:590–600
Kanade K, Kale B, Baeg JO, Lee S, Lee C, Moon SJ, Chang H (2007) Self-assembled aligned Cu doped ZnO nanoparticles for photocatalytic hydrogen production under visible light irradiation. Mater Chem Phys 102:98–104
Khan Y, Durrani S, Mehmood M, Ahmad J, Riaz Khan M, Firdous S (2010) Low temperature synthesis of fluorescent ZnO nanoparticles. Appl Surf Sci 257(5):1756–1761
Lauf R, Bond W (1984) Fabrication of high-field zinc oxide varistors by sol–gel processing. Ceram Bull 63:278
Lee N, Kim M, Chung I, Oh M (1991) Electrical characteristics and reheat-treatment effects in a ZnO varistor fabricated by two-stage heat-treatment. J Mater Sci 26:1126
Manjula G, Nair M, Nirmala, Rekha K, Anukaliani A (2011) Structural, optical, photo catalytic and antibacterial activity of ZnO and Co doped ZnO nanoparticles. Mater Lett 65(12):1797–1800
Mousavi H, Haratizadeh H, Minaee H (2011) The effect of morphology and doping on photoluminescence of ZnO nanostructures. Opt Commun 284(14):3558–3561
Ohya Y, Niwa T, Ban T, Takahashi Y (2001) Thin film transistor of ZnO fabricated by chemical solution deposition. Jpn J Appl Phys 40:297
Okuyama K, Lenggoro I, Tagami N, Tamaki S, Tohge N (1997) Preparation of ZnS and CdS fine particles with different particle sizes by a spray-pyrolysis method. J Mater Sci 32:1229
Silverstein R, Webster F (1997) Spectroscopic identification of organic compound, 6th edn. Wiley, New York, pp 81–97
Thorat J, Kanade K, Nikam L, Chaudhari P, Kale B (2010) Nanostructured ZnO hexagons and optical properties. J Mater Sci 22(4):394–399
Vanheusden K, Warren W, Seager C, Tallant D, Voigt JA, Gnade B (1996) Mechanisms behind green photoluminescence in ZnO phosphor powders. J Appl Phys 79:7983
Wang J, Shu D, Xiao M, Meng Y (2006) Co-polymerization of carbon dioxide and propylene oxide using zinc adipate as catalyst. J Appl Polym Sci 99(1):200–206
Xu J, Ji W, Lin J, Tang S, Du Y (1998) Preparation of ZnS nanoparticles by ultrasonic radiation method. Appl Phys A 66:639
Yang J, An S, Park W, Yi G, Choi W (2004) Photocatalysis using ZnO thin films and nanoneedles grown by metal–organic chemical vapor deposition. Adv Mater 16:1661–1664
Zhang J, Sun L, Yin J, Su H, Liao C, Yan C (2002) Control of ZnO morphology via a simple solution route. Chem Mater 14:4172
Zhao Y, Kwon Y (2004) Templateless hydrothermal synthesis of aligned ZnO nanorods. Chem Lett 33:1578
Zou B, Volkov V, Wang Z (1999) Optical properties of amorphous ZnO, CdO, and PbO nanoclusters in solution. Chem Mater 11:3037
Acknowledgments
The authors were grateful to University of Pune and UGC, Government of India for the financial assistance and Dr. Arun Andhale, Principal, Mahatama Phule Mavidyalaya, Pimpri Pune-411017 for providing facilities to carry out the above research. The authors also thank staff of Nanocrystalline Material laboratory of C-MET, Pune for many stimulating discussions and providing characterization facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thorat, J.H., Kanade, K.G., Nikam, L.K. et al. Self-aligned nanocrystalline ZnO hexagons by facile solid-state and co-precipitation route. J Nanopart Res 14, 657 (2012). https://doi.org/10.1007/s11051-011-0657-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-011-0657-8