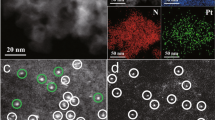
Abstract
We have investigated the catalysis of Pt nanocrystals for olefin hydrogenation in liquid phase by using several kinds of Pt nanocrystals with different shapes, whose results suggest that Pt cube shows the highest catalytic activity without significant shape change after the repeated hydrogenation. We prepared four nanocrystals according to our original methods: four kinds of Pt cubes (6.7, 8.2, 9.5, and 10.1 nm), Pt tetrahedron (4.6 nm), Pt nanowire (2.0 nm), and Pt cuboctahedron (7.5 nm) with high shape selectivity and narrow size distributions. To avoid the effect of different surface protecting agent on hydrogenation, we use the same polymer (polyacrylic acid) to stabilize four kinds of Pt nanocrystals. We adopted olefins with different structural characteristics, such as hexene, cyclohexene, cis-, and trans-stilbene. Turnover frequency (TOF) values for the hydrogenation of both cis- and trans-stilbene exhibit the following order: Pt cube > Pt cuboctahedron > Pt tetrahedron ≈ Pt nanowire, in accord with the ratio of Pt(100) on the surface of Pt nanocrystals. Raman spectroscopy of chemically adsorbed trans-stilbene on Pt cube and Pt tetrahedron indicates that the C–C double bonds of olefin group and phenyl group prefer to interact with Pt(100) facet, coinciding with its highest activity for olefin hydrogenation. The hydrogenation of cyclohexene as well as 1-hexene over Pt cubes with different sizes gave similar TOF values, suggesting that the hydrogenation takes place preferentially on the planes of Pt(100) facet in liquid phase.







Similar content being viewed by others
References
Ahmadi TS, Wang ZL, Green TC, Henglein A, El-Sayed MA (1996) Shape-controlled synthesis of colloidal platinum nanoparticles. Science 272:1924–1926
Anderson JR (1975) Structure of metallic catalysts. Academic Press, London
Bernardo CGPM, Gomes JANF (2001) The adsorption of ethylene on the (100) surfaces of platinum, palladium and nickel: a DFT study. J Mol Struct (Theochem) 542:263–271
Bratlie KM, Flores LD, Somorjai GA (2005) Hydrogenation and dehydrogenation of cyclohexene on Pt(100): a sum frequency generation vibrational spectroscopic and kinetic study. Surf Sci 599:93–106
Bratlie KM, Lee H, Komvopoulos K, Yang P, Somorjai GA (2007) Platinum nanoparticle shape effects on benzene hydrogenation selectivity. Nano Lett 7:3097–3101
Bürgi T, Atamny F, Schlögl R, Baiker A (2000) Adsorption of ethyl pyruvate on Pt(111) studied by XPS and UPS. J Phys Chem B 104:5953–5960
Chen JH, Yau SL, Chang SC (2002) An in situ scanning tunneling microscopy and cyclic voltammetric study of iodobenzene and iodoheptane molecules adsorbed on a Pt(111) electrode. J. Phys Chem B 106:9079–9085
Chen J, Xiong Y, Yin Y, Xia Y (2006) Pt nanoparticles surfactant-directed assembled into colloidal spheres and used as substrates in forming Pt nanorods and nanowires. Small 2:1340–1343
Chen J, Lim B, Lee EP, Xia Y (2009) Shape-controlled synthesis of platinum nanocrystals for catalytic and electrocatalytic applications. Nano Today 4:81–95
Fu X, Wang Y, Wu N, Gui L, Tang Y (2002) Shape-selective preparation and properties of oxalate-stabilized Pt colloid. Langmuir 18:4619–4624
Ge Q, King DA (1999) The chemisorption and dissociation of ethylene on Pt{111} from first principles. J Chem Phys 110:4699–4702
Gottlieb HE, Kotlyar V, Nudelman A (1997) NMR chemical shifts of common laboratory solvents as trace impurities. J Org Chem 62:7512–7515
Grass ME, Yue Y, Habas SE, Rioux RM, Teall CI, Yang P, Somorjai GA (2008) Silver ion mediated shape control of platinum nanoparticles: removal of silver by selective etching leads to increased catalytic activity. J Phys Chem C 112:4797–4804
Hardeveld RV, Hartog F (1969) The statistics of surface atoms and surface sites on metal crystals. Surf Sci 15:189–230
Herricks T, Chen J, Xia Y (2004) Polyol synthesis of platinum nanoparticles: control of morphology with sodium nitrate. Nano Lett 4:2367–2371
Ikeda S, Ishino S, Harada T, Okamoto N, Sakata T, Mori H, Kuwabata S, Torimoto T, Matsumura M (2006) Ligand-free platinum nanoparticles encapsulated in a hollow porous carbon shell as a highly active heterogeneous hydrogenation catalyst. Angew Chem Int Ed 45:7063–7066
Kinge S, Bönnemann H (2006) One-pot dual size- and shape selective synthesis of tetrahedral Pt nanoparticles. Appl Organomet Chem 20:784–787
Kweskin SJ, Rioux RM, Habas SE, Komvopoulos K, Yang P, Somorjai GA (2006) Carbon monoxide adsorption and oxidation on monolayer films of cubic platinum nanoparticles investigated by infrared-visible sum frequency generation vibrational spectroscopy. J Phys Chem B 110:15920–15925
Lee H, Habas SE, Kweskin S, Butcher D, Somorjai GA, Yang P (2006) Morphological control of catalytically active platinum nanocrystals. Angew Chem Int Ed 45:7824–7828
Louette P, Bodino F, Pireaux J-J (2005) Poly(acrylic acid) (PAA) XPS reference core level and energy loss spectra. Surf Sci Spectra 12:22–26
Mittendorfer F, Thomazeau C, Raybaud P, Toulhoat H (2003) Adsorption of unsaturated hydrocarbons on Pd(111) and Pt(111): a DFT study. J Phys Chem B 107:12287–12295
Narayanan R, El-Sayed MA (2004a) Changing catalytic activity during colloidal platinum nanocatalysis due to shape changes: electron-transfer reaction. J Am Chem Soc 126:7194–7195
Narayanan R, El-Sayed MA (2004b) Shape-dependent catalytic activity of platinum nanoparticles in colloidal solution. Nano Lett 4:1343–1348
Narayanan R, El-Sayed MA (2005a) Catalysis with transition metal nanoparticles in colloidal solution: nanoparticle shape dependence and stability. J Phys Chem B 109:12663–12676
Narayanan R, El-Sayed MA (2005b) Effect of colloidal nanocatalysis on the metallic nanoparticle shape: the Suzuki reaction. Langmuir 21:2027–2033
Narayanan R, El-Sayed MA (2008) Some aspects of colloidal nanoparticle stability, catalytic activity, and recycling potential. Top Catal 47:15–21
Narayanan R, Tabor C, El-Sayed MA (2008) Can the observed changes in the size or shape of a colloidal nanocatalyst reveal the nanocatalysis mechanism type: homogeneous or heterogeneous? Top Catal 48:60–74
Ng YH, Ikeda S, Harada T, Higashida S, Sakata T, Mori H, Matsumura M (2007) Fabrication of hollow carbon nanospheres encapsulating platinum nanoparticles using a photocatalytic reaction. Adv Mater 19:597–601
Okamoto K, Akiyama R, Yoshida H, Yoshida T, Kobayashi S (2005) Formation of nanoarchitectures including subnanometer palladium clusters and their use as highly active catalysts. J Am Chem Soc 127:2125–2135
Peng ZM, You HJ, Yang H (2010) Composition-dependent formation of platinum silver nanowires. ACS Nano 4:1501–1510
Petroski J, El-Sayed MA (2003) FTIR study of the adsorption of the capping material to different platinum nanoparticle shapes. J Phys Chem A 107:8371–8375
Rioux RM, Song H, Grass M, Habas S, Niesz K, Hoefelmeyer JD, Yang P, Somorjai GA (2006) Monodisperse platinum nanoparticles of well-defined shape: synthesis, characterization, catalytic properties and future prospects. Top Catal 39:167–174
Sánchez-Sánchez CM, Solla-Gullón J, Vidal-Iglesias FJ, Aldaz A, Montiel V, Herrero E (2010) Imaging structure sensitive catalysis on different shape-controlled platinum nanoparticles. J Am Chem Soc 132:5622–5624
Serrano-Ruiz JC, López-Cudero A, Solla-Gullón J, Sepúlveda-Escribano A, Aldaz A, Rodríguez-Reinoso F (2008) Hydrogenation of α, β unsaturated aldehydes over polycrystalline, (111) and (100) preferentially oriented Pt nanoparticles supported on carbon. J Catal 253:159–166
Shen Z, Yamada M, Miyake M (2007) Preparation of single-crystalline platinum nanowires with small diameters under mild conditions. Chem Commun 245–247
Somorjai GA (2008) The 13th international symposium on relations between homogeneous and heterogeneous catalysis—an introduction. Top Catal 48:1–7
Somorjai GA, Park JY (2008) Colloid science of metal nanoparticle catalysts in 2D and 3D structures. Challenges of nucleation, growth, composition, particle shape, size control and their influence on activity and selectivity. Top Catal 49:126–135
Song H, Kim F, Connor S, Somorjai GA, Yang P (2005) Pt nanocrystals: shape control and langmuir-Blodgett Monolayer formation. J Phys Chem B 109:188–193
Teranishi T, Kurita R, Miyake M (2000) Shape control of Pt nanoparticles. J Inorg Organomet Polym 10:145–156
Tian N, Zhou ZY, Sun SG, Ding Y, Wang ZL (2007) Electro-oxidation activity nanocrystals with high-index facets and high synthesis of tetrahexahedral platinum. Science 316:732–735
Tsung CK, Kuhn JN, Huang W, Aliaga C, Hung L, Somorjai GA, Yang P (2009) Sub-10 nm platinum nanocrystals with size and shape control: catalytic study for ethylene and pyrrole hydrogenation. J Am Chem Soc 131:5816–5822
Watanabe H, Okamoto Y, Furuya K, Sakamoto A, Tasumi M (2002) Vibrational analysis of trans-stilbene in the ground and excited singlet electronic states revisited. J Phys Chem A 106:3318–3324
Yamada M, Kon S, Miyake M (2005) Synthesis and size control of Pt nanocubes with high selectivity using the additive effect of NaI. Chem Lett 34:1050–1051
Yu Y, Xu B (2006) Shape-controlled synthesis of Pt nanocrystals: an evolution of the tetrahedral shape. Appl Organomet Chem 20:638–647
Acknowledgments
This work was supported by Fuel Cell Cutting-Edge Science Research Project (No. 08001242-0) and Development of PEFC Technologies aiming for Practical Application/Base technology/Analysis of Morphology, Electrochemical reaction and Mass transfer for MEA materials (No. 10000806-0) from NEDO, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cao, M., Miyabayashi, K., Shen, Z. et al. Olefin hydrogenation catalysis of platinum nanocrystals with different shapes. J Nanopart Res 13, 5147–5156 (2011). https://doi.org/10.1007/s11051-011-0497-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-011-0497-6